Embryology and Phylogeny (see dedicated Neurovascular Evolution and Vascular Neurombryology pages for details)
The PCA is an old vessel, in fact emerging in the lower species prior to development of the MCA. This makes sense because some of the areas it supplies — occipital and mesial temporal lobes (besides the tectum) — are phylogenetically more well established than the bulk of frontoparieral areas now served by the MCA. The PCA originally belongs to the anterior, carotid circulation, arising as the carotid terminates into the cranial and caudal rami — the future ACA and PCA, respectively. Transfer of the PCA territory to the vertebrobasilar circulation is a process which seems to be necessitated, from the phylogenetic standpoint, by the relatively large volume of brain supplied in the human and other “higher species” by the carotid system. In many mammals the vertebrobasilar system does not prominently figure in PCA supply, being confined to the brainstem and cerebellum. This is the simple phylogenetic explanation for the most common circle of Willis variant — the “fetal” PCA — named as such when early “fetal” arrangement of PCA origin from the ICA persists in the adult form, which is about 20-25% of the time. The variability in number is due to haggling over the semantics of what exactly constitutes a “fetal” PCA. Is it “complete” absence of the P1 segment of the PCA? Is it a state where P1 is felt to be insufficient to provide adequate PCA perfusion should the PCOM be closed? Is it simply when the PCOM is larger than the P1? Whatever the case, you have every right to confidently pick your own definition. The important point to appreciate is that there is no such thing as an “absent” PCOM or P1. There is certainly an angiographic or MRA or CTA absence of these vessels, which means nothing except that your equipment is not good enough to see it. They are always present in fact, as embryologically required.
Early development of the PCA is dominated by its supply of the lateral and third ventricular choroidal territory (the choroidal stage, as best described in Surgical Neuroangiography), together with the Anterior Choroidal Artery. In fact, from a phylogenetic standpoint, it is the Anterior Choroidal and not the PCA that serves as the artery to the occipital and temporal lobar areas. In the human, the Anterior Choroidal parenchymal supply is typically restricted to its segment proximal to its plexal point (again see dedicated page for details), with hemispheric territory transferred to the PCA. However, on occasion the anterior choroidal retains some of its formerly extensive cortical possessions, and as such might even be mistaken for a fetal PCA. In the vast majority of cases, however, the PCA is responsible for the supply of the mesial Occipital, inferomesial Parietal, and inferior Temporal lobes, as well as the choroid plexus of the lateral (together with the anterior choroidal) and third ventricles. Importantly, it also contributes to the supply of the cerebral peduncles and the collicular plate, its phylogenetically older territories.
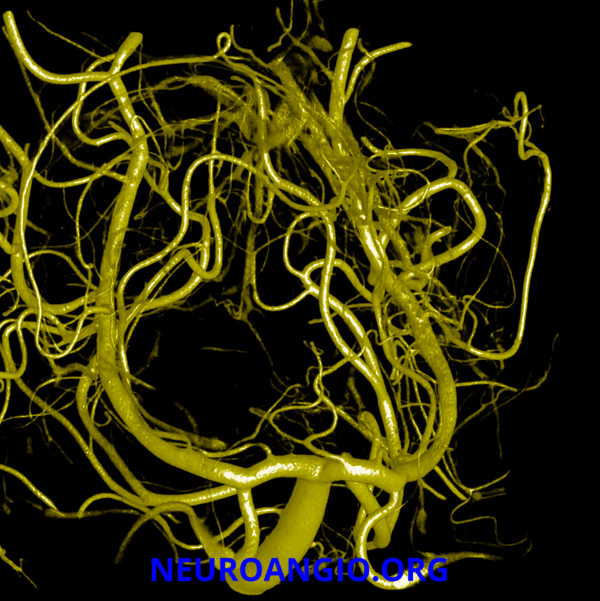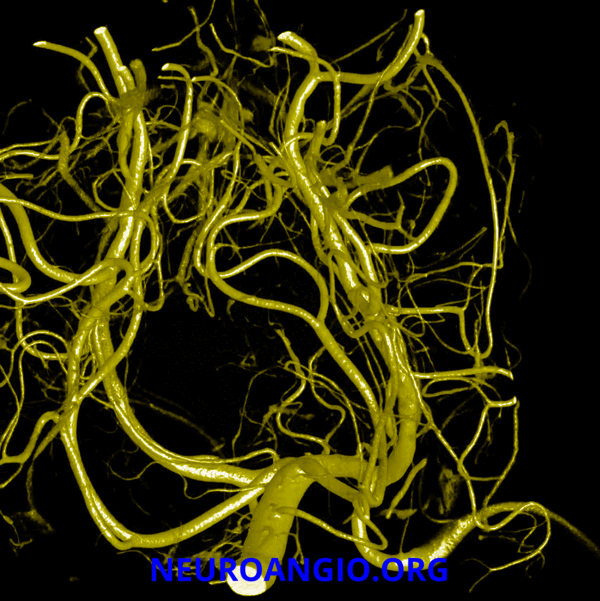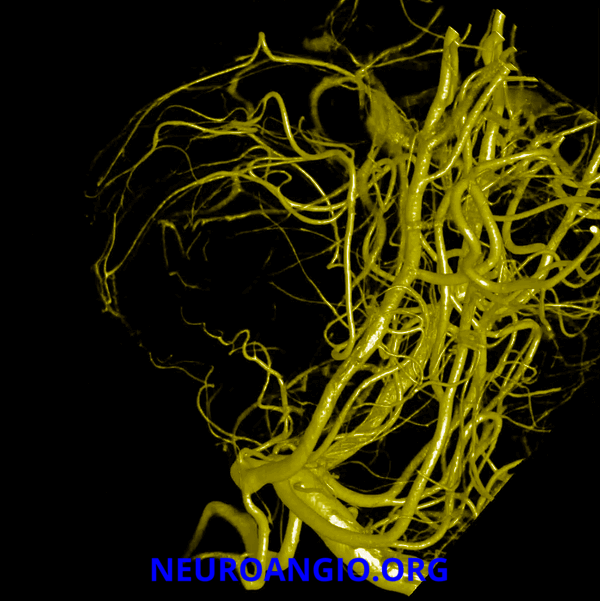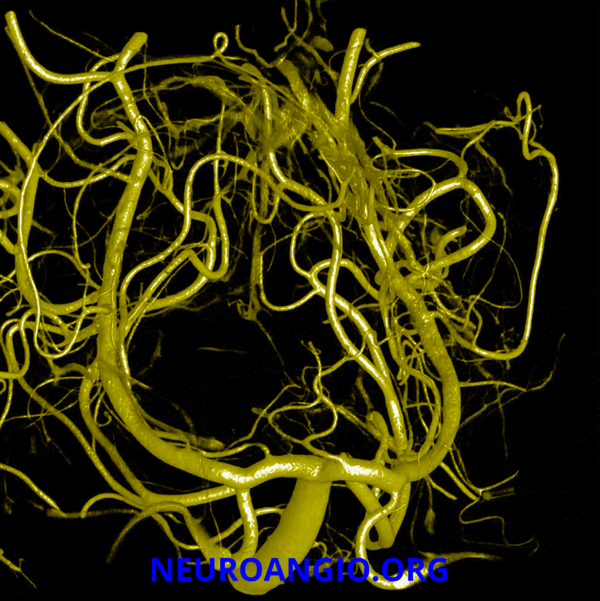
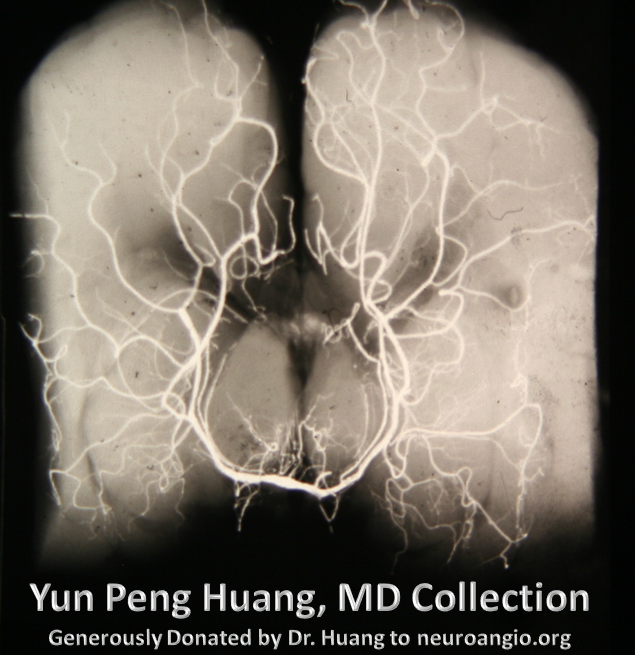
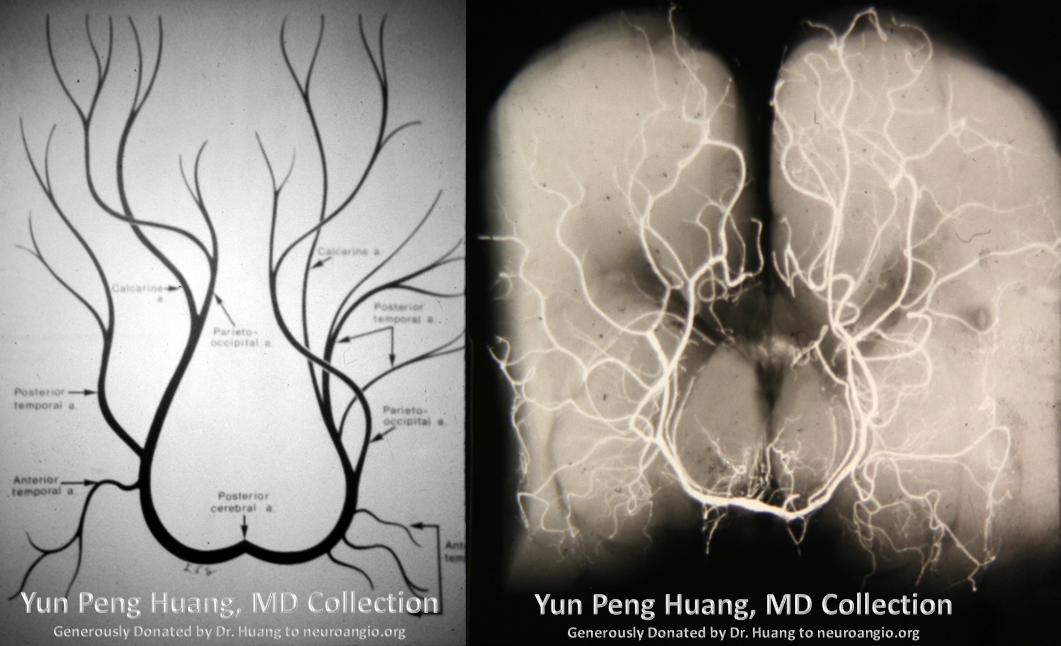
Here is a beautiful image of a PCA specimen x-ray from the Yung Peng Huang collection
Also from the collection, a specimen photograph
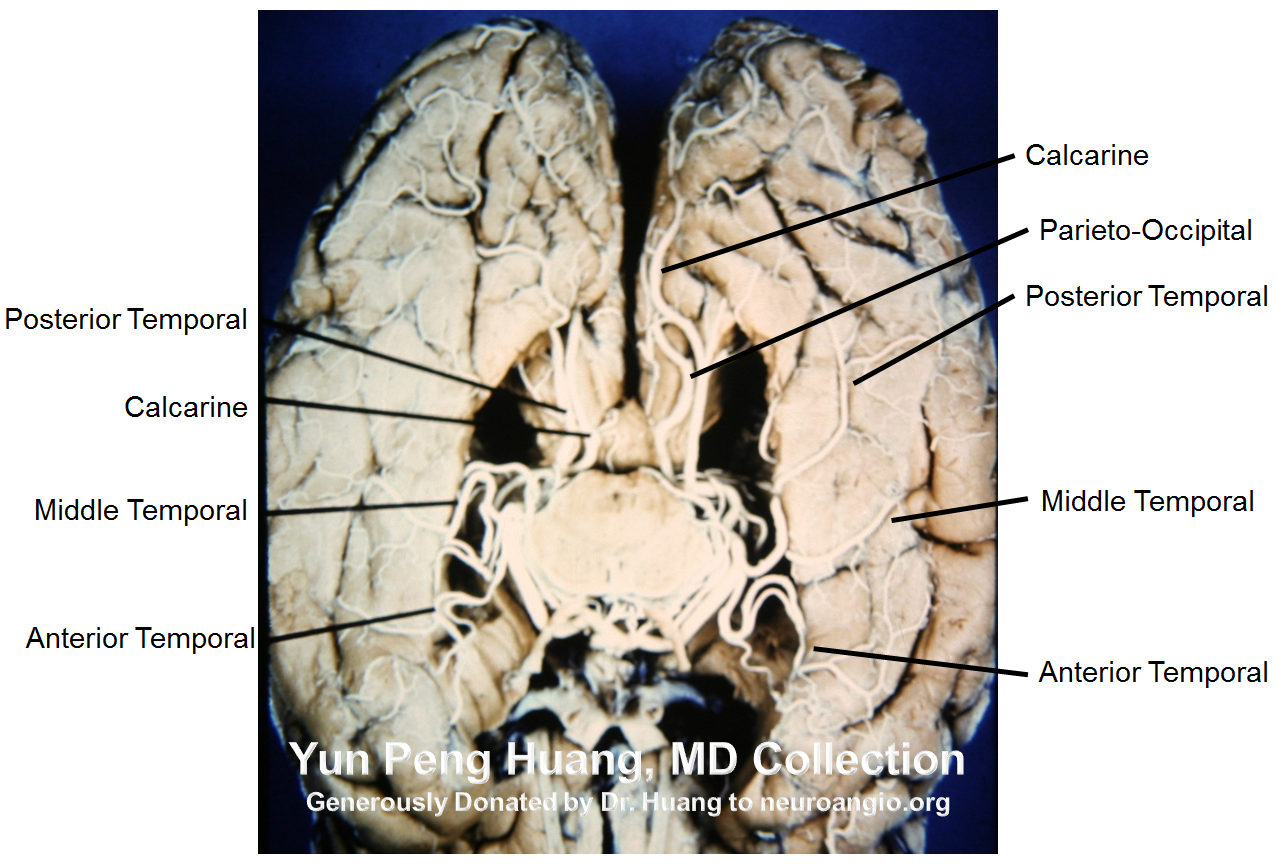
A detail of the photograph, with additional labels (see below)
Territory, variations, images
Nothing like an infarct to show the territory and variation. Rare case of bilateral carotid occlusions with DWI showing devastating infarcts, sparing both PCA territories. Note extent of thalamic supply. Case courtesy Drs. Omar Kass-Hout and Tibor Becske
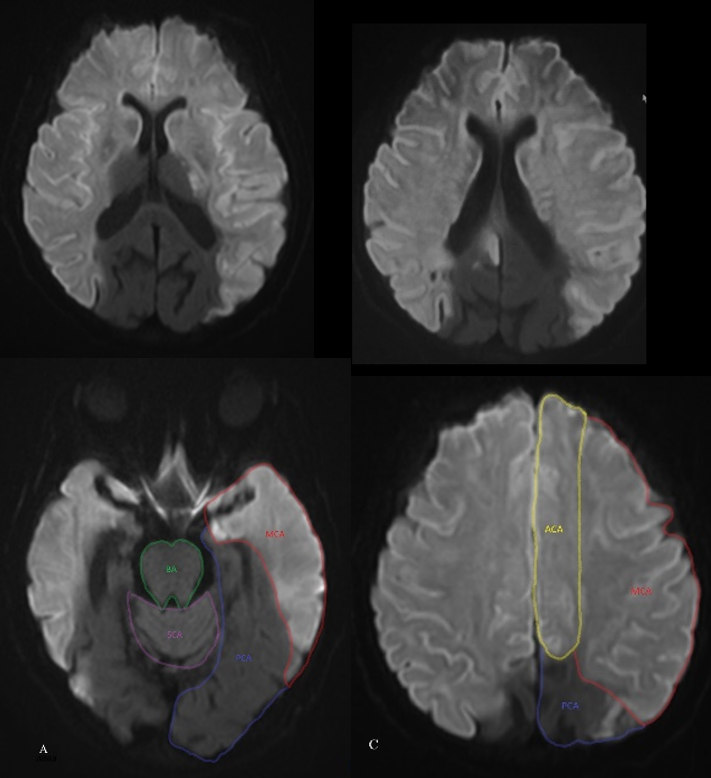
The more common reverse — top of basilar infarct in patient with fetal right PCOM, thus sparing right PCA. Extensive infarcts of SCA and left PCA territory. Note inferior temporal/hippocampal, and thalamic involvement.
B
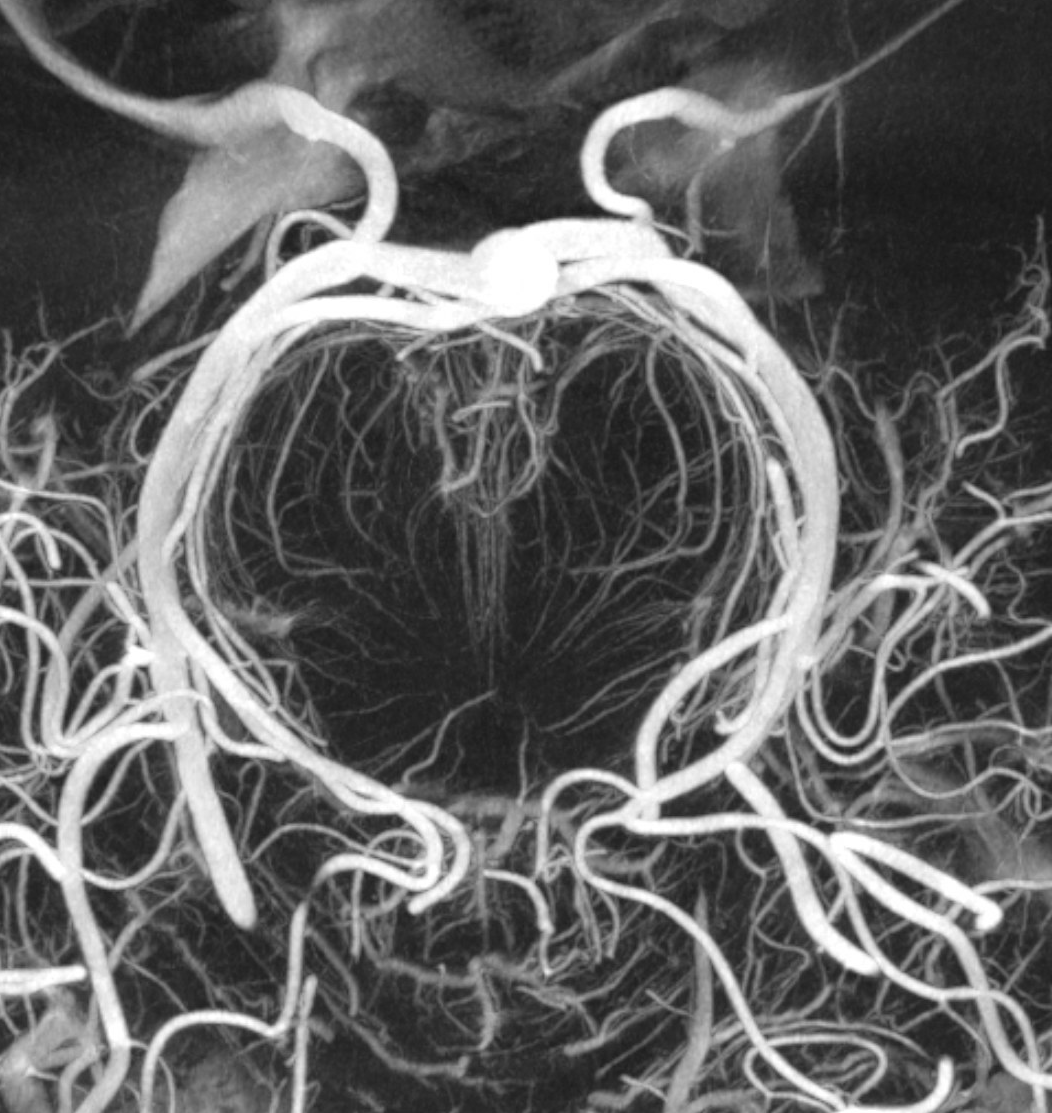
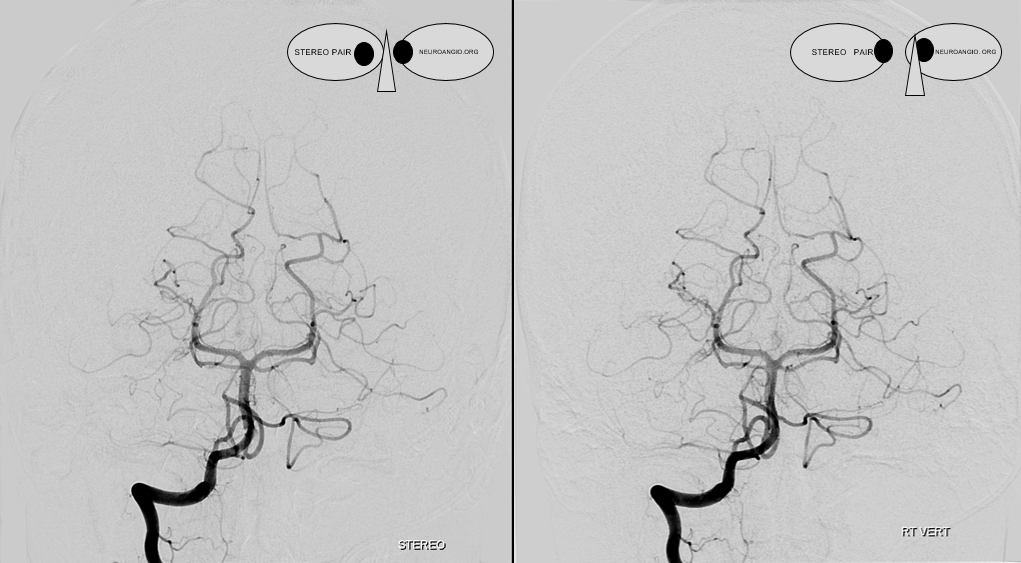
As mentioned above, the PCA is usually responsible for the supply of the Occipital, inferomesial Parietal, and inferior Temporal lobes, as well as the choroid plexus of the lateral (together with the anterior choroidal) and third ventricles. Importantly, by way of the collicular or circumcollicular arteries it also contributes to vascularization of cerebral peduncles and the collicular plate, its phylogenetically older territories. Angiographically, the PCA is best evaluated in steep frontal projections such as the Townes, which allow the various segments of the PCA trunk to be “opened up”. In the lateral, there is frequent superimposition, which can be resolved by yawing the lateral tube to separate the two. Stereoscopic imaging can help decide which side is which, if knowledge is otherwise insufficient. The above image from the YPH collection, with a corresponding drawing on the left, for reference:
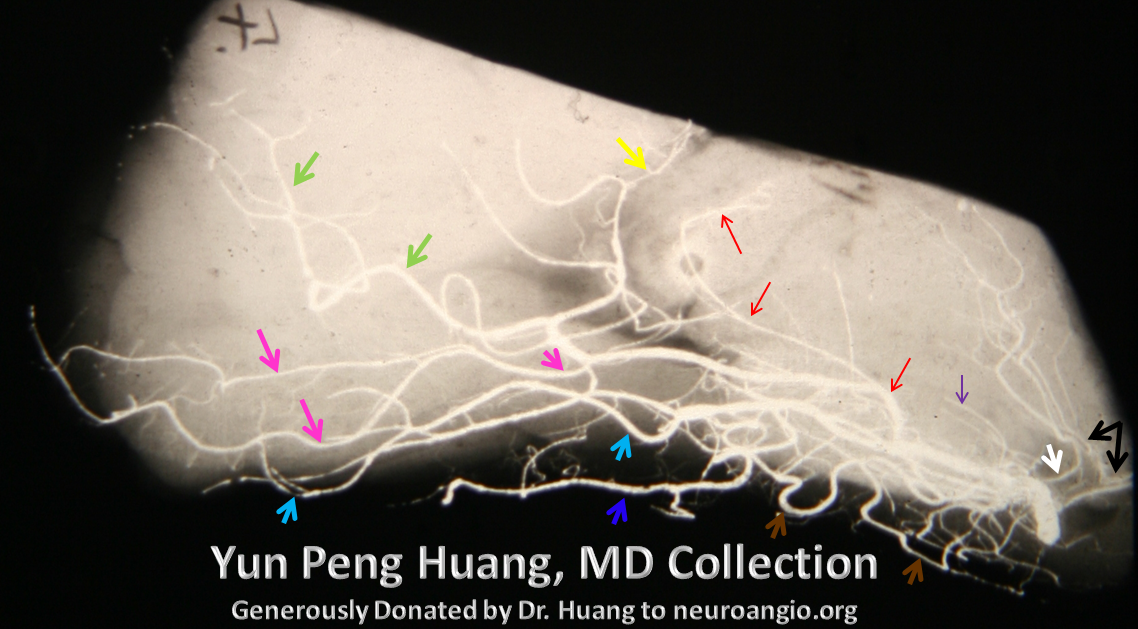
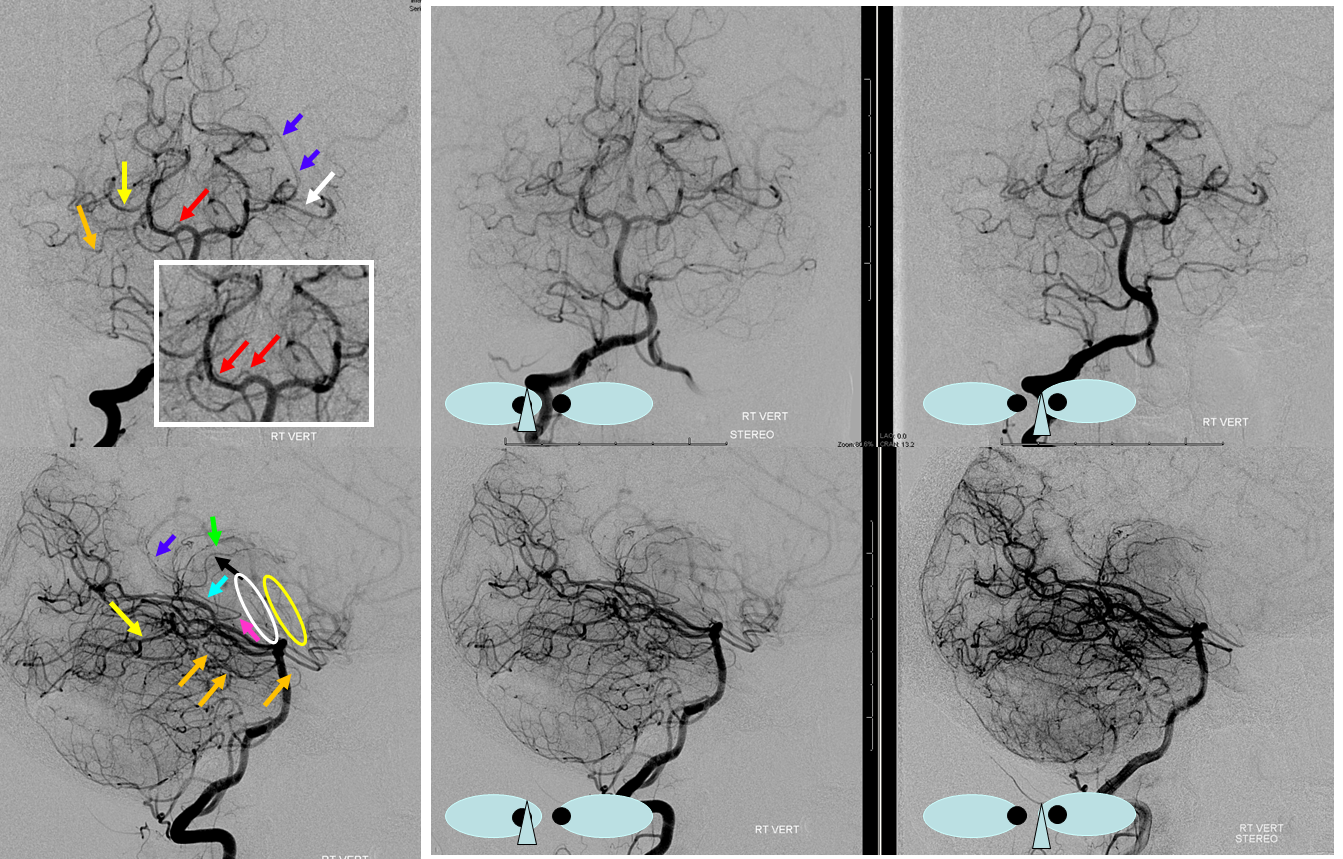
A lateral image of the x-ray specimen, with colored arrows this time. Inferior thalamoperforating arteries from the P1 segment (white) and PCOM (black). Geniculate = purple. Posterior lateral choroidal = red (note characteristic C-shaped curve). Posterior pericalossal (a.k.a. splenial branch) = yellow. Calcarine = pink. Parieto-occipital = green. Posterior Inferior Temporal = light blue. Middle Inferior Temporal = dark blue. Anterior Inferior Temporal = brown
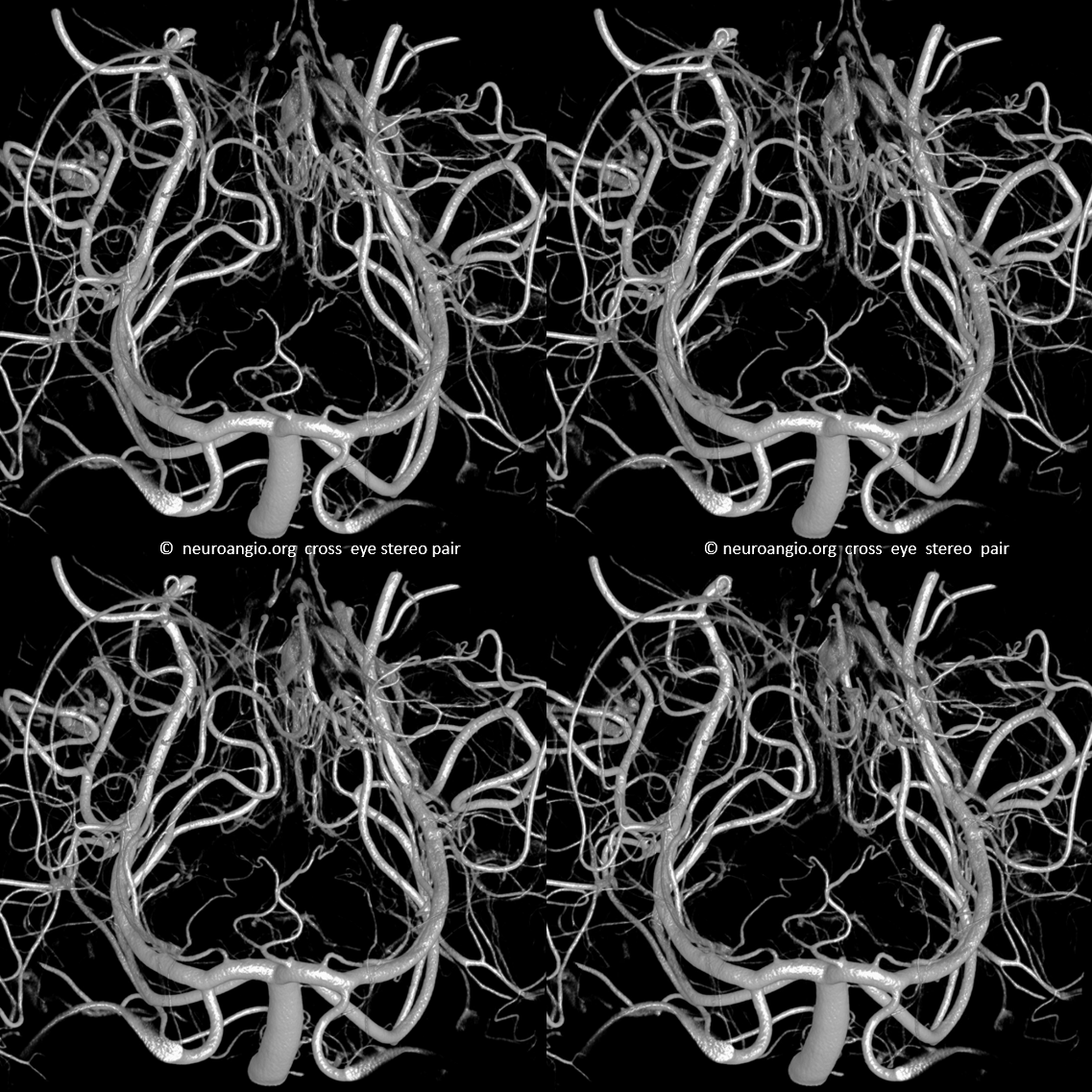
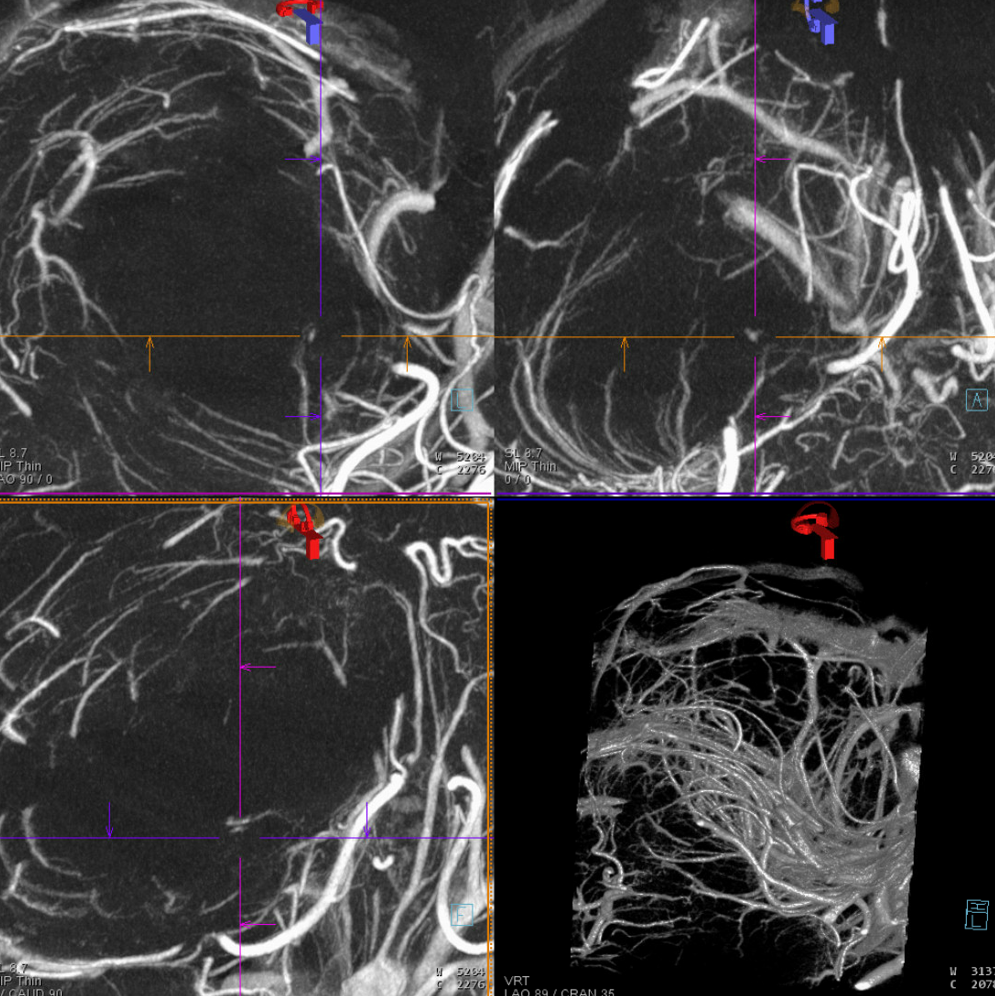
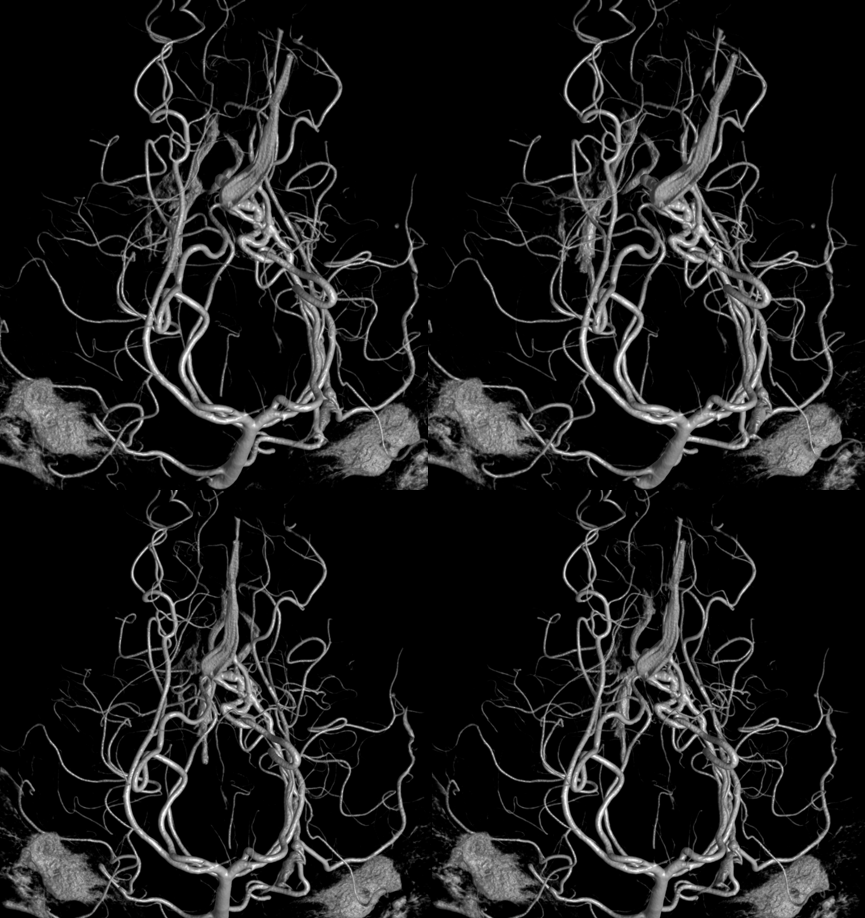
From the classic to the modern (by 2022). DYNA CT images of the PCA — artery of Percheron, circumcullicular arteries, posterior medial and lateral choroidal vessels all seen in incredible detail. By Dr. Eytan Raz. Stereo pairs of volume rendered images
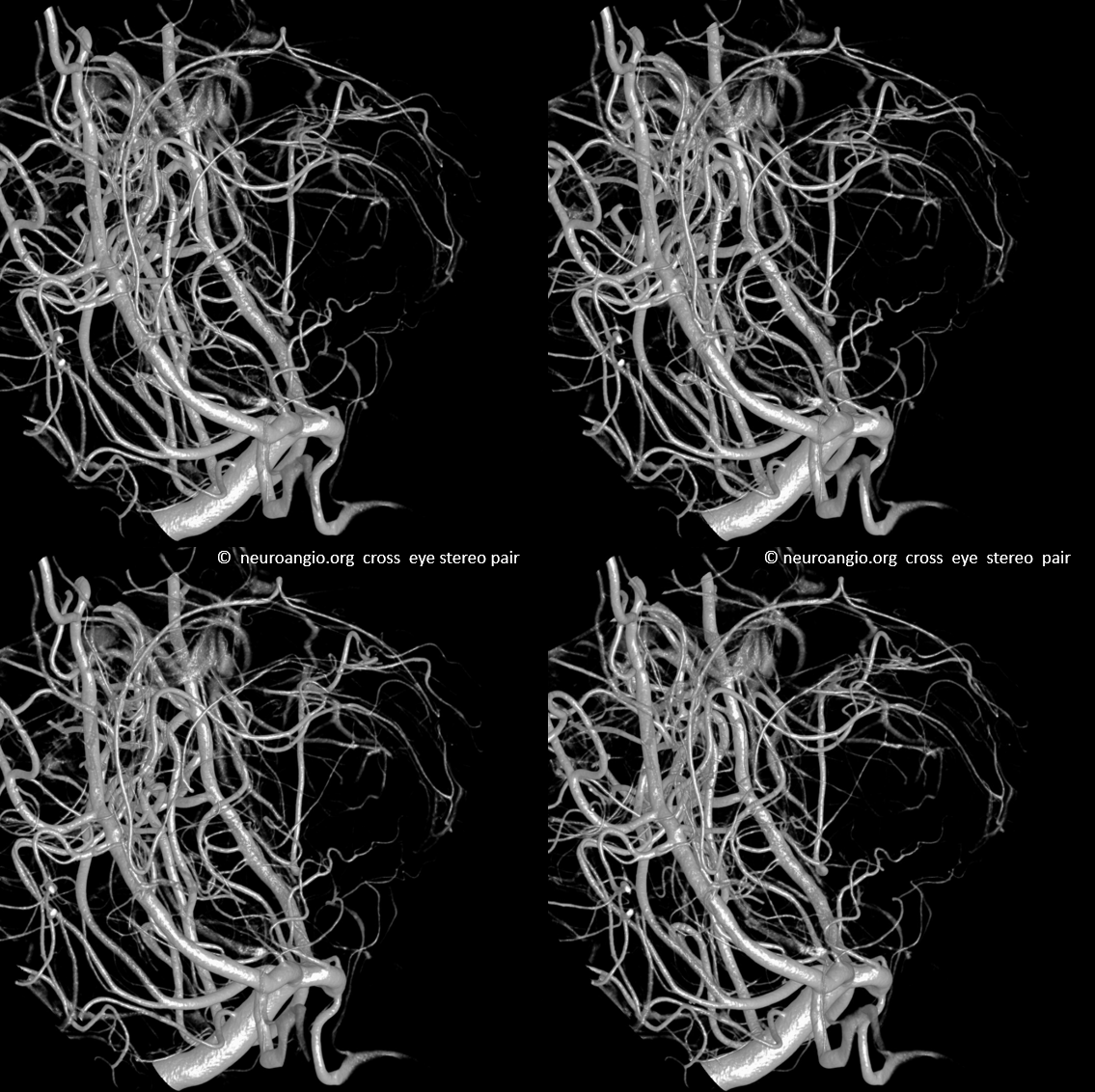
Anaglyph DYNAs
DYNA Movie — stop and scroll thru individual images
Spot sign of thalamic bleed in this patient
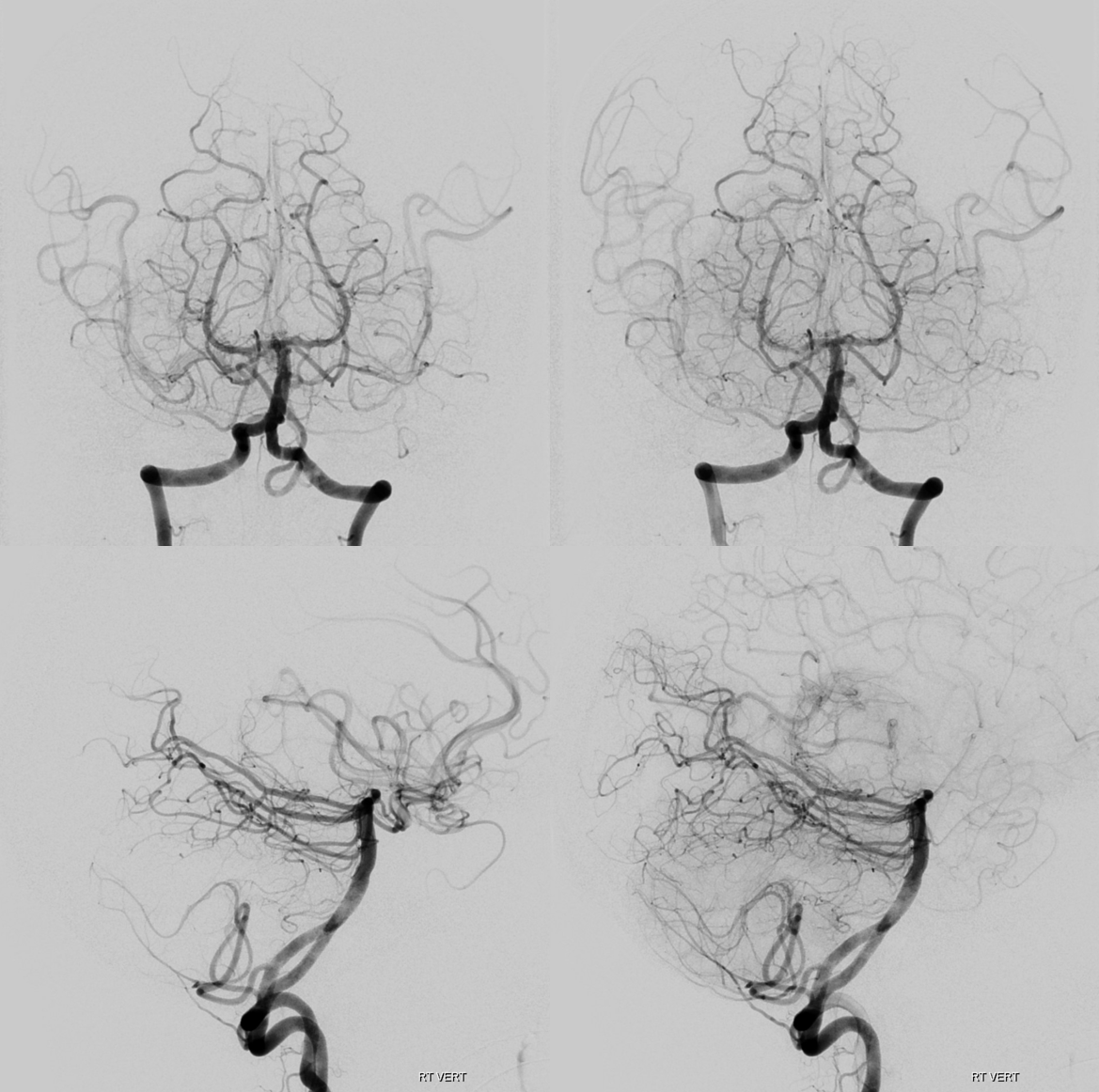
DSA — usual appearance, the difference in detail is incredible
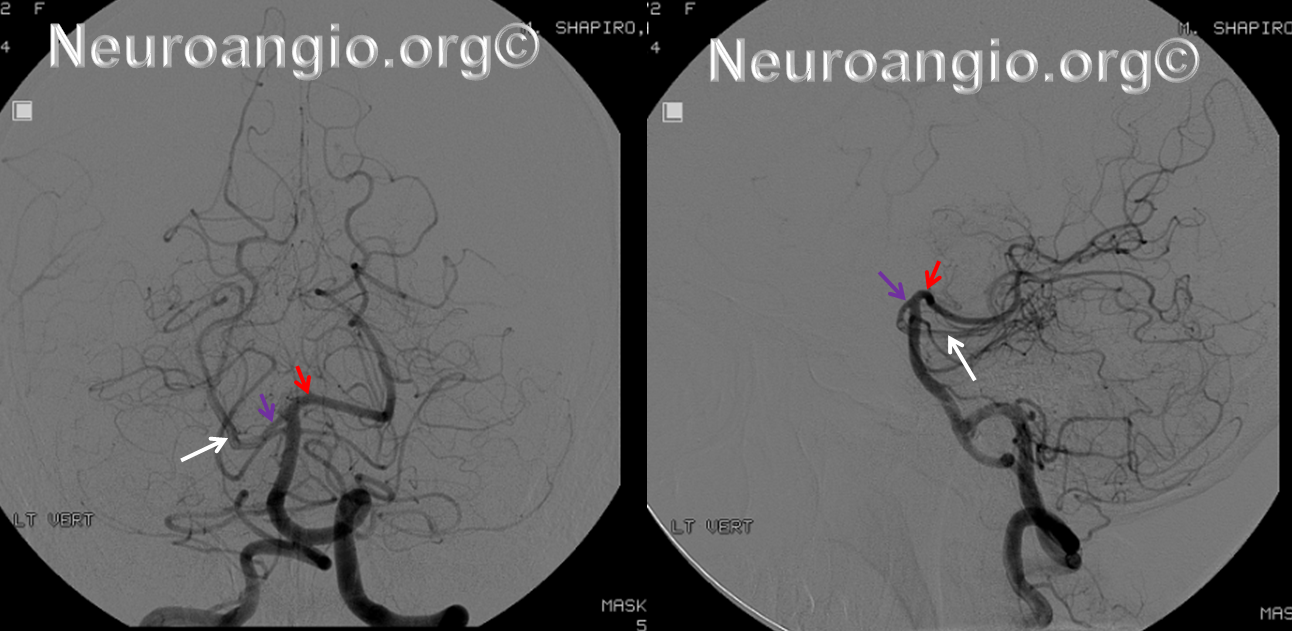
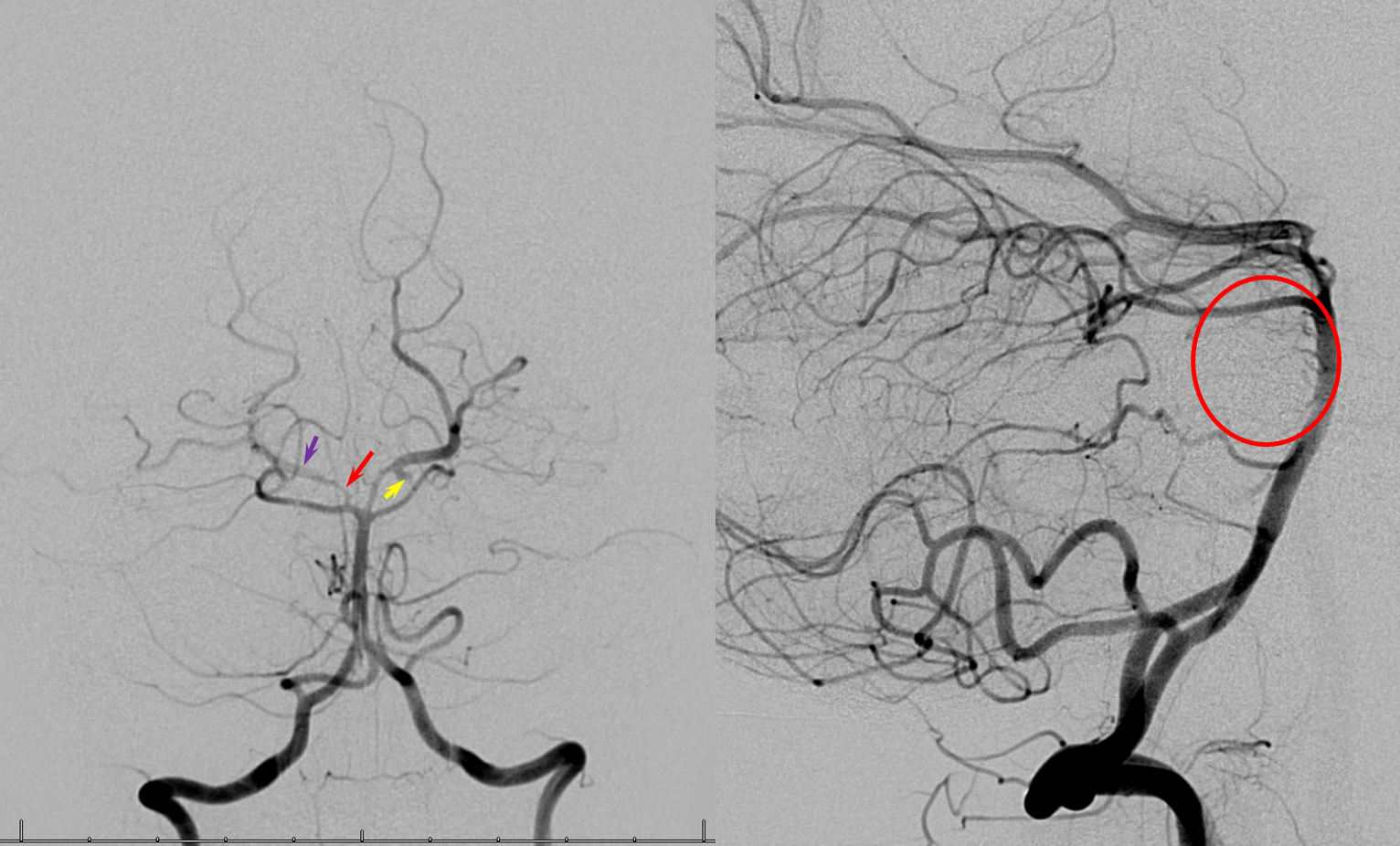
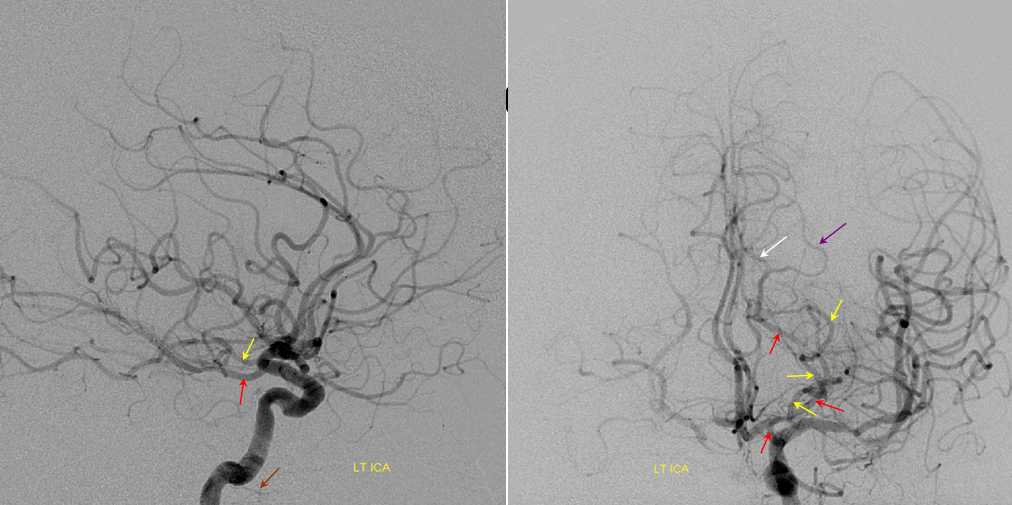
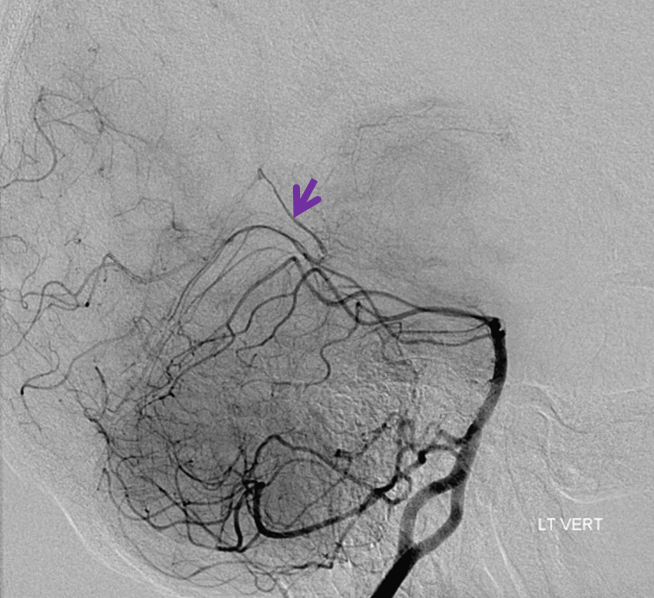
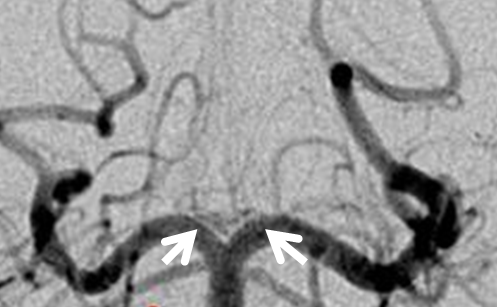
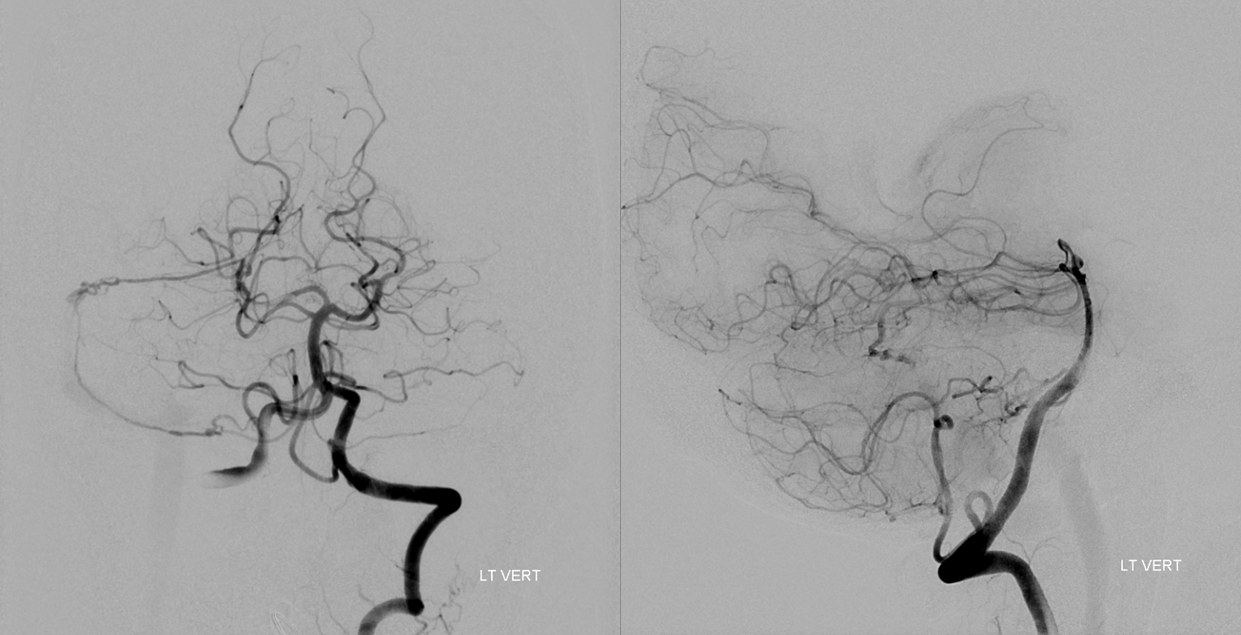
Below is a typical angiographic image of the PCA. On the right, the P1 (purple) segment is smaller than on the left (red), with streaming of unopacified blood (white) through the right PCOM visible distal to its otherwise invisible confluence with the P1 segment.
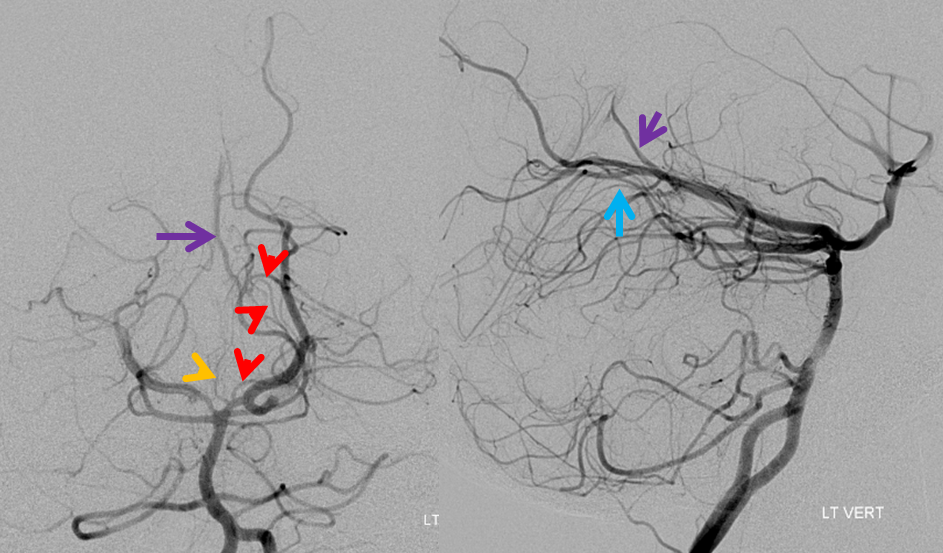
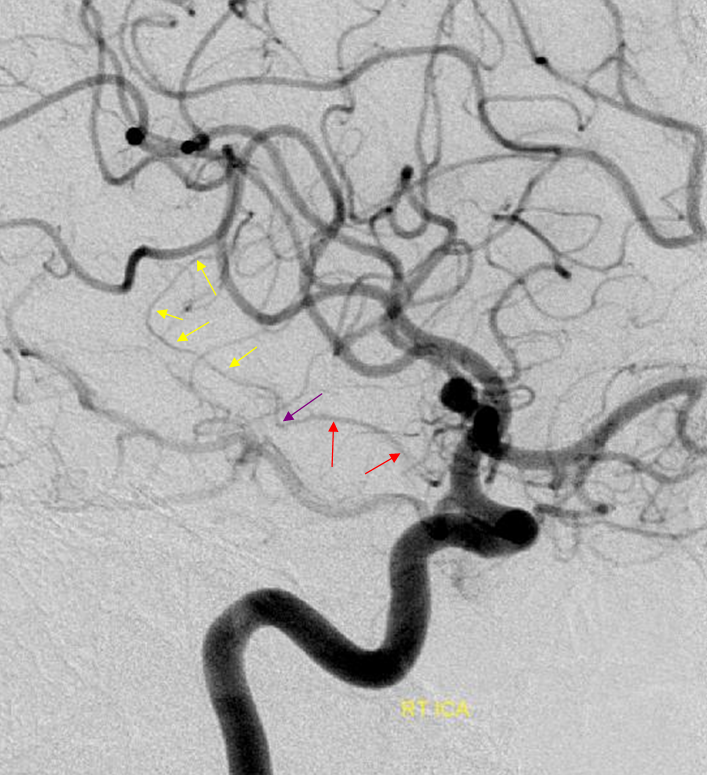
A typical “fetal PCOM”, ICA injection. Various branches of the PCA are marked, including PCOM (red) and posterior inferior temporal (yellow, green), middle temporal (black) and anterior temporal (white) branches arising from a common trunk, with the posterior temporal one being the largest. There is tremendous variation in how the inferior temporal branches are organized. The important part to understand is where the branch is relation to the brain.
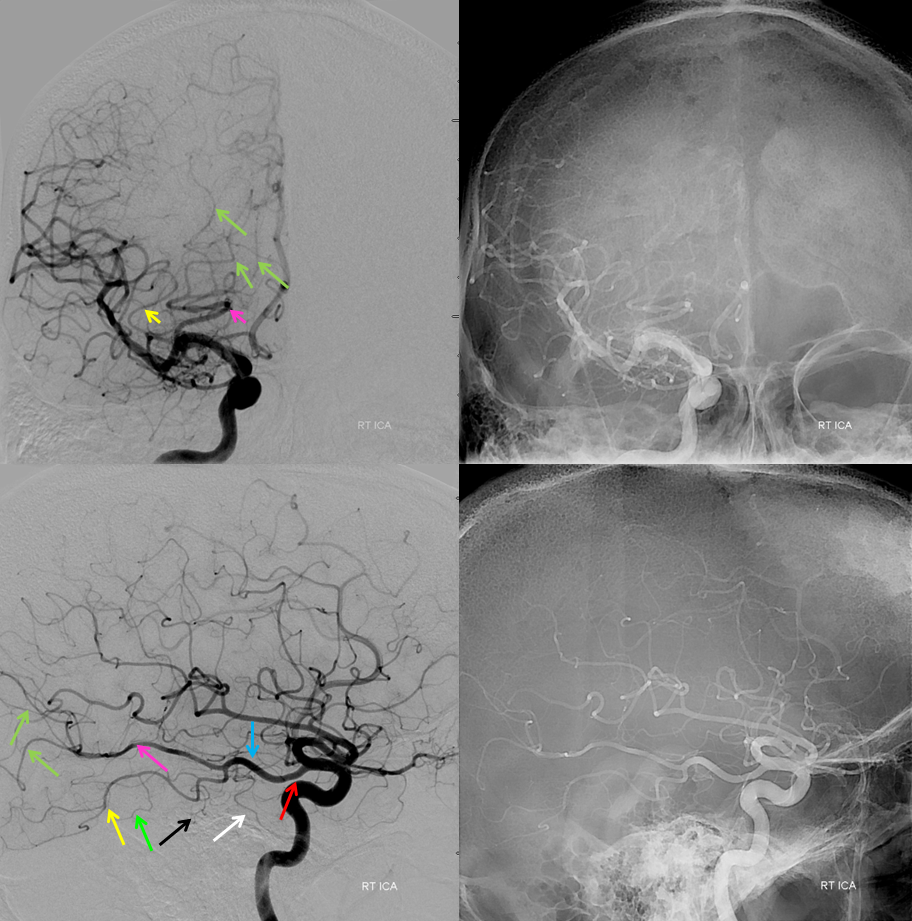
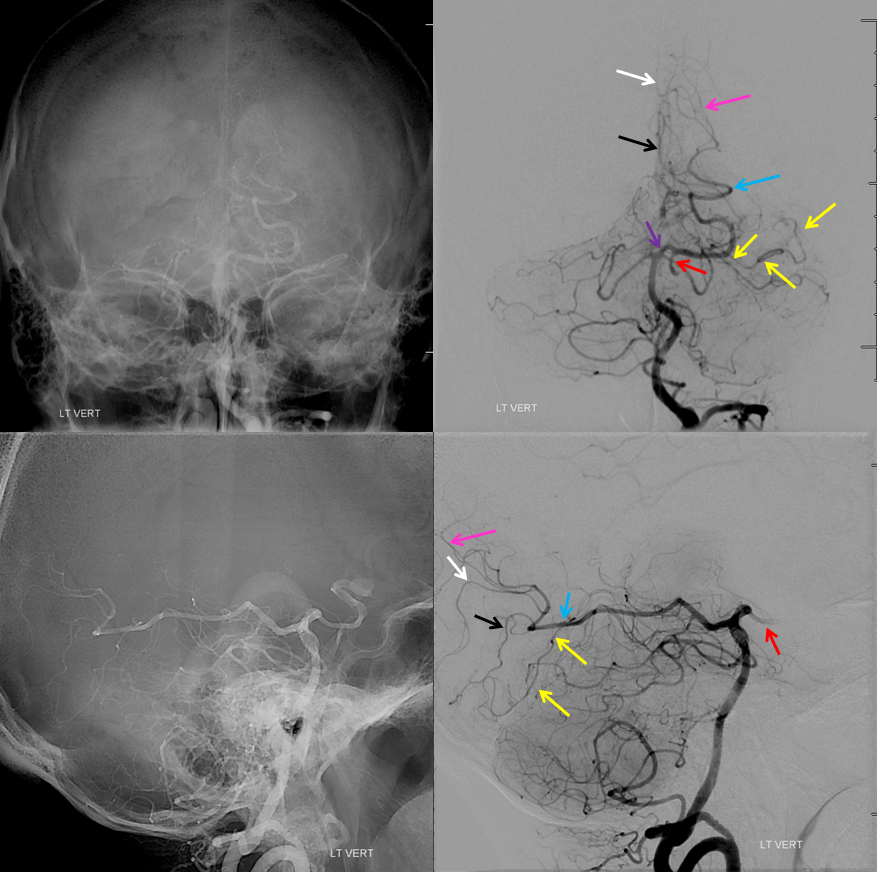
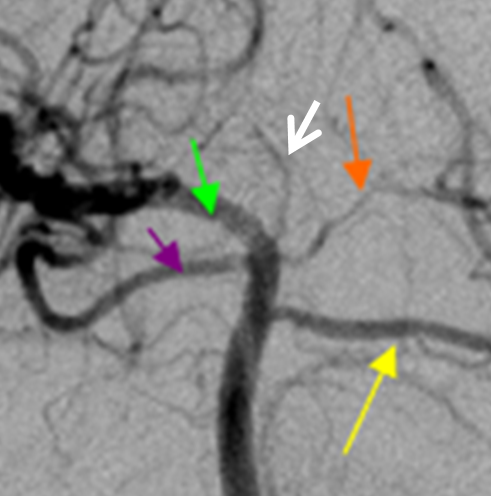
Left vertebral injection in the same patient. Notice a steeper “Townes” frontal projection, as compared with the standard AP above. The right P1 is “absent”, but only angiographically. The left PCOM (red) is transiently retrogradely opacified by the force of the injection. The portion which curves around the brainstem is the “P2”. Anything distal is P3, P4, and on. Because of the fetal disposition on the right, the lateral allows for left PCA view without superimposition. The all-important calcarine branch (black) is the paramedian branch just above the tent (where the calcarine cortex is). Importantly, it will be foreshortened in the Townes view because the tent will be sloping down. The parieto-occipital branch on the other hand (pink) is less foreshortened. It is typically large and should not be mistaken for the calcarine one.
Also notice several large posterior inferior temporal branches (yellow). Because of the shape of the tent, which slopes down as it stretches laterally, the temporal branches will ovelap the cerebellum in both frontal and lateral views. It is very important to understand that. The calcarine branch will never do that in the lateral view, because the medial occipital lobe is always above the cerebellum.
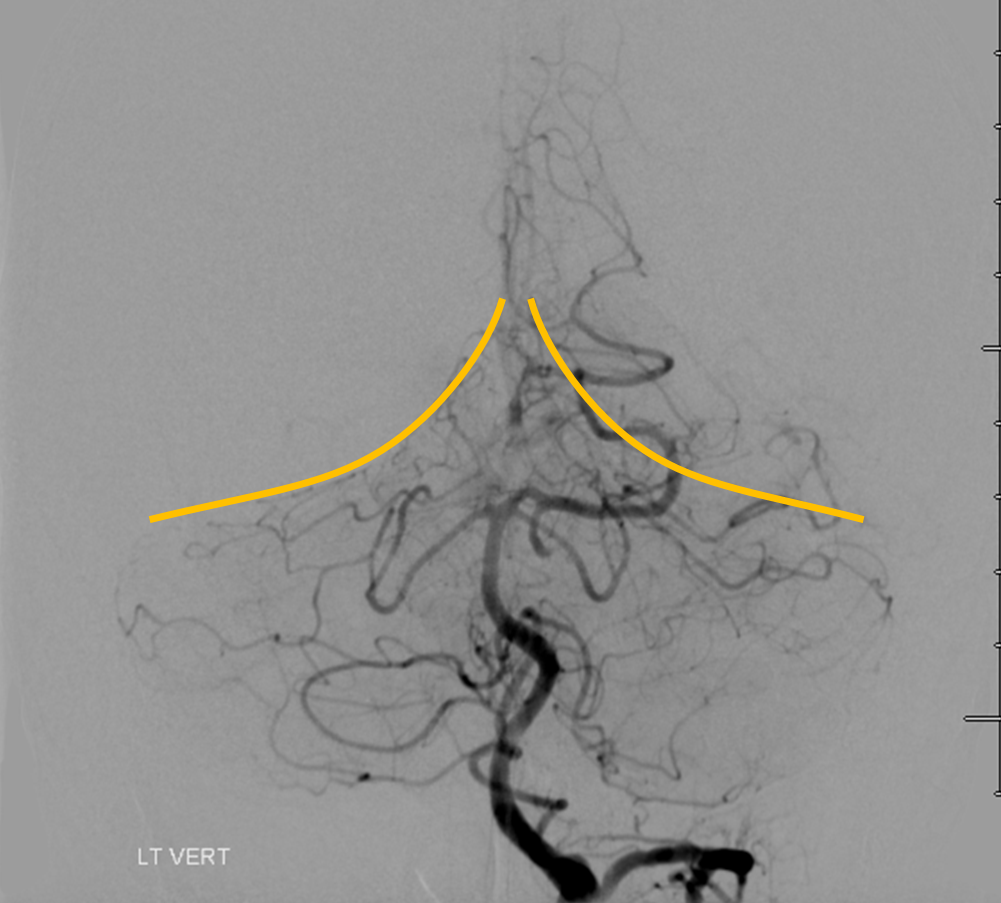
Here is the same patient with the tentorium cerebelli outlined, for clarity’s sake
Another fetal PCOM (by my definition), on the left. A small P1 segment is present (orange). Notice how well the left tent is outlined by the hemispheric branch of the left SCA plastered up against it (no arrows this time). A sizable basilar fenestration is shown by the brown arrow. Also note that the frontal left vertebral injection projection is not a Townes, but more like a Caldwell/Submental type. Thus, all PCA branches are superimposed on each other, with no good definition, except for one — the posterior lateral choroidal branch (white arrow) rises above the rest, to where the lateral ventricle would be.
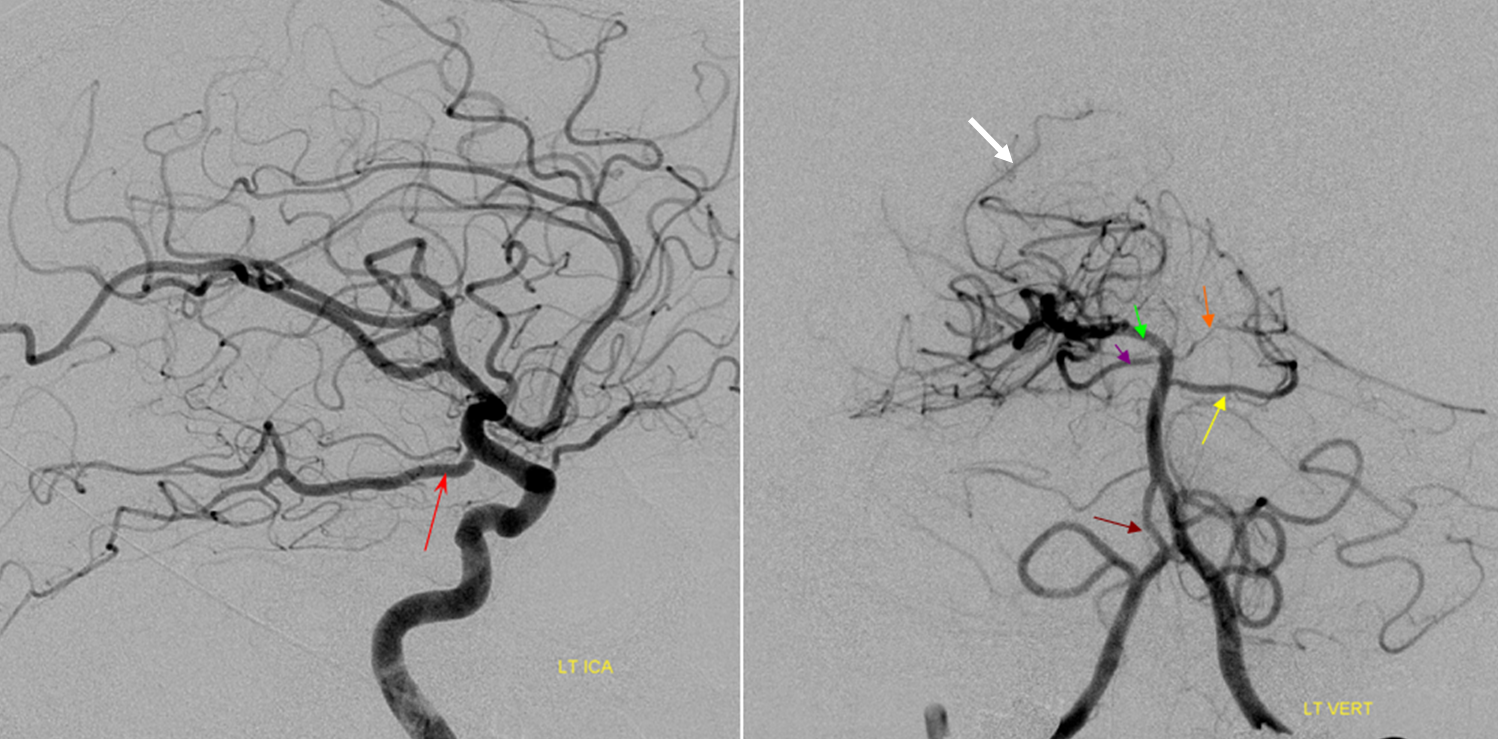
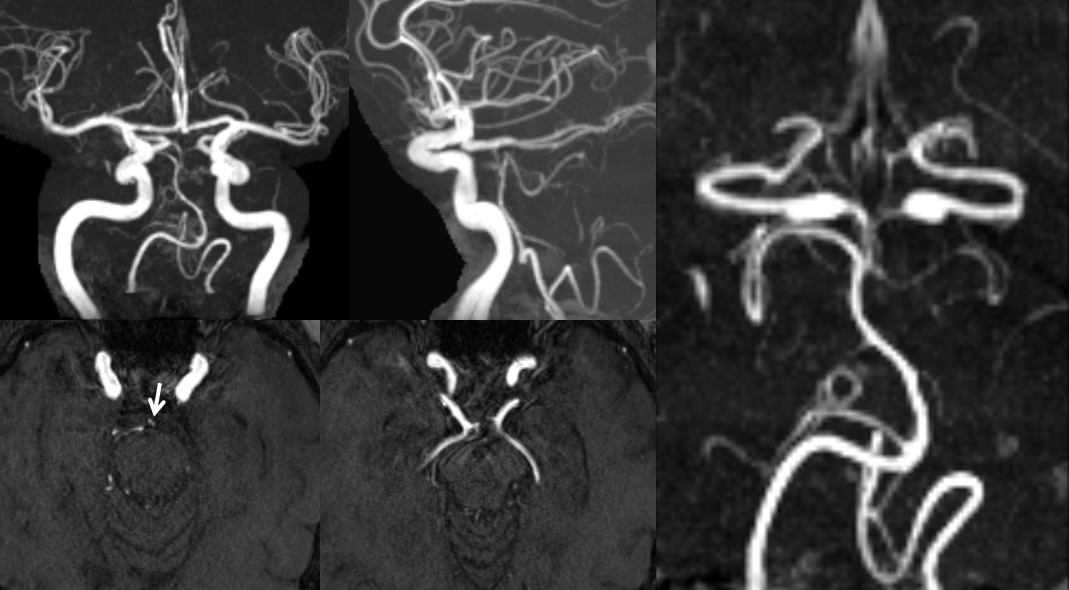
MRA of bilateral fetal PCOMs, with corresponding hypoplasia of the basilar artery. Notice the unfused long P1 segments above the superior cerebellar ostia (arrow). See Basilar Artery page for details
Posterior Communicating Artery Fenestration — not something you see everyday. Here is one, in association with a Trigeminal artery. So, what does that say about the embryology of the PCOM as a caudal ramus of the ICA?
Cortical Territory
Of course, there is balance in everything — the supply is in balance with MCA and ACA. Importantly, the undersurface of the temporal lobe is PCA territory, not MCA (except for the pole region that is usually fed by anterior temporal branch of the MCA) This includes hippocampus — a minor detail when WADAs are performed, but they do work somehow anyway — probably knock off the Papez circuit elsewhere. Below is a hippocampal AVM with exclusive anterior inferior temporal branch supply. MCA does nothing.
“Superior Cerebellar Artery origin from the PCA”
The apparent origin of the PCA from the P1 segment is, in fact, directly related to the embryology of the basilar artery, which is formed by fusion of paired longitudinal neural arteries. The extent of fusion determines the length of the basilar, and some of its variations. The position of the SCAs with respect to the P1 segment is related to the degree of upper basilar fusion, with the “unfused” configuration corresponding to SCA origin from the PCA. Imagine the basilar artery as a zipper:
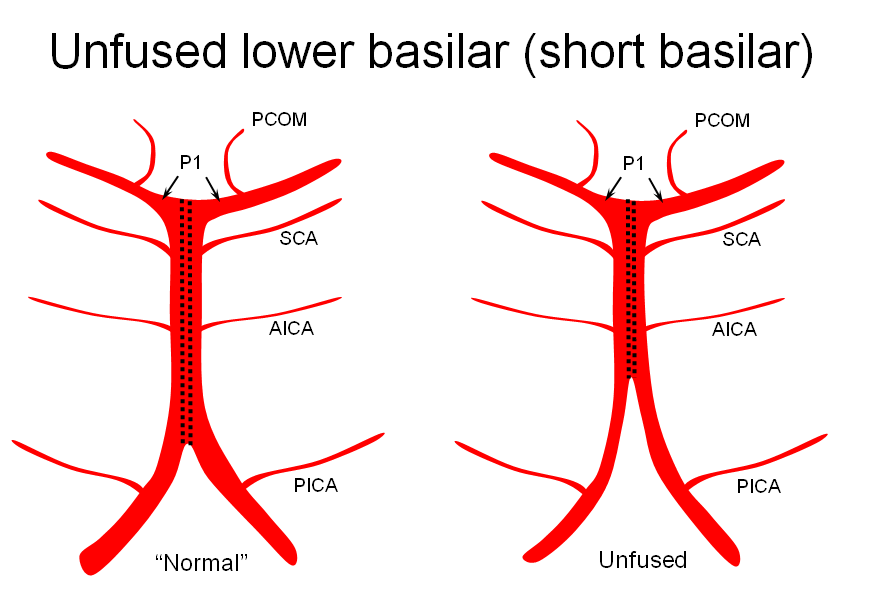
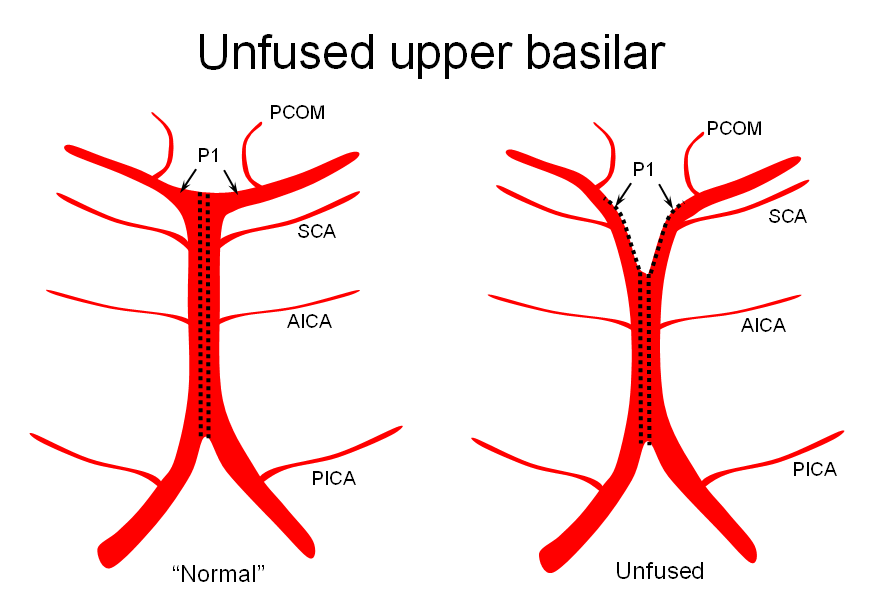
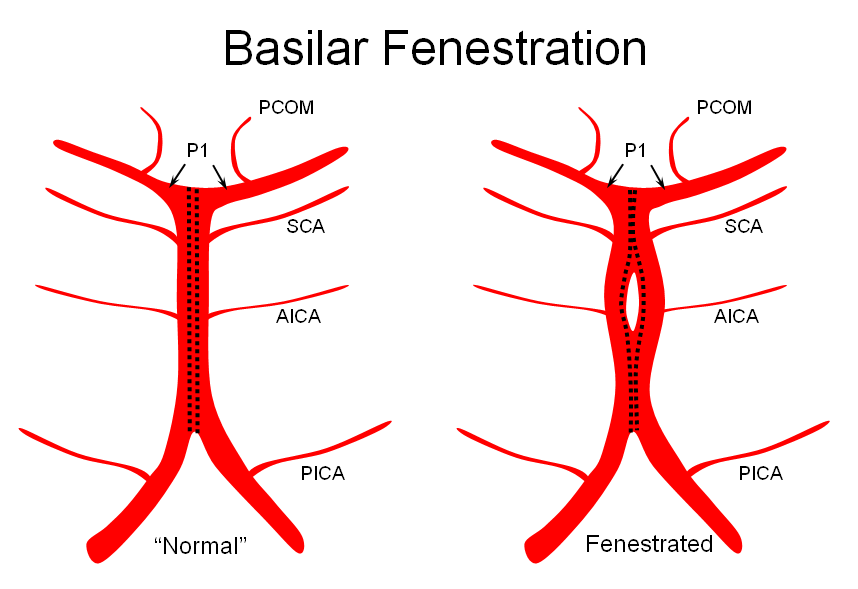
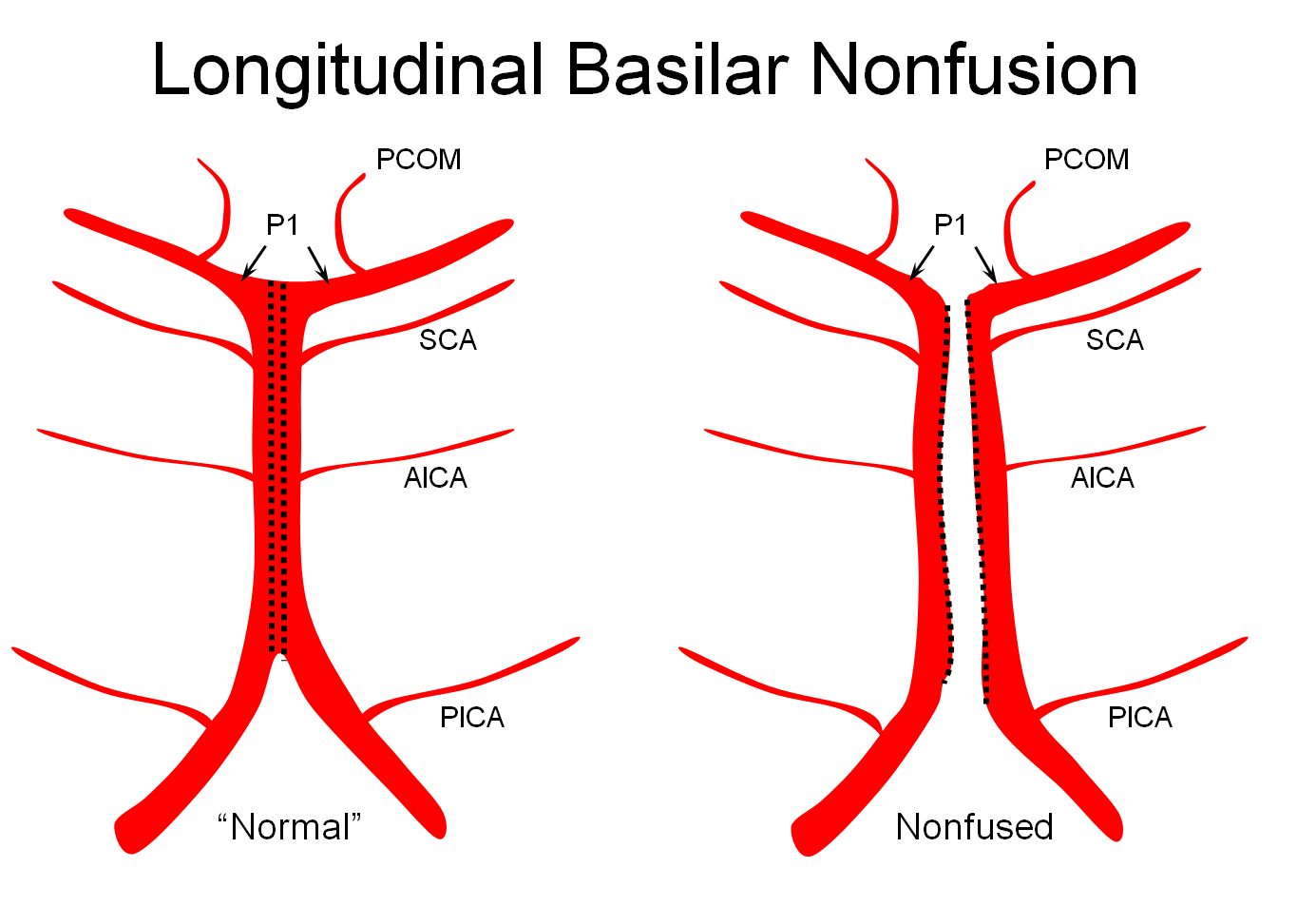
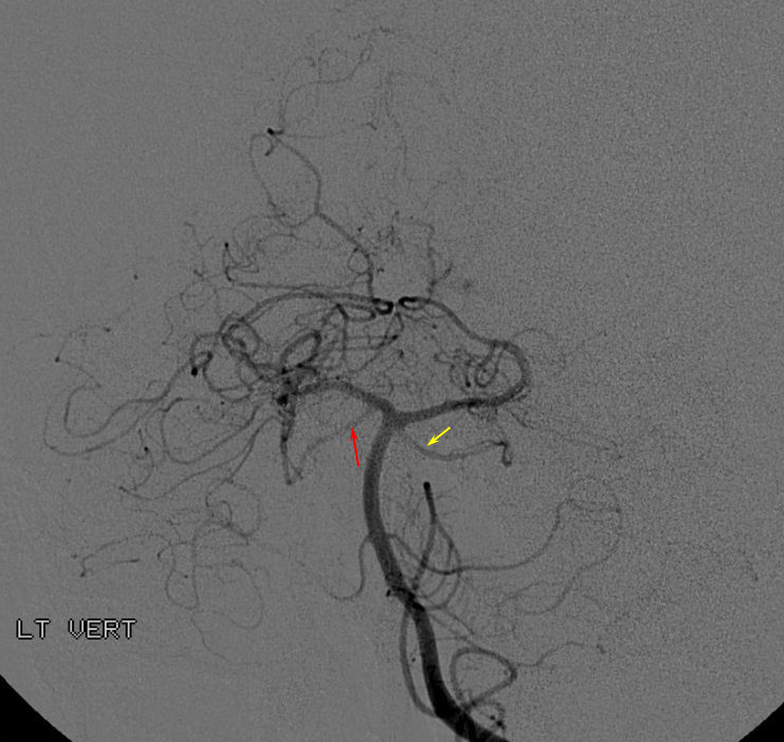
Origin of superior cerebellar artery at “top” of the basilar on the left (yellow), and from P1 segment on the right (red arrow)– seen often, can be conceptualized as a “short” basilar which did not undergo enough coalescence at the top to incorporate the superior cerebellar artery.
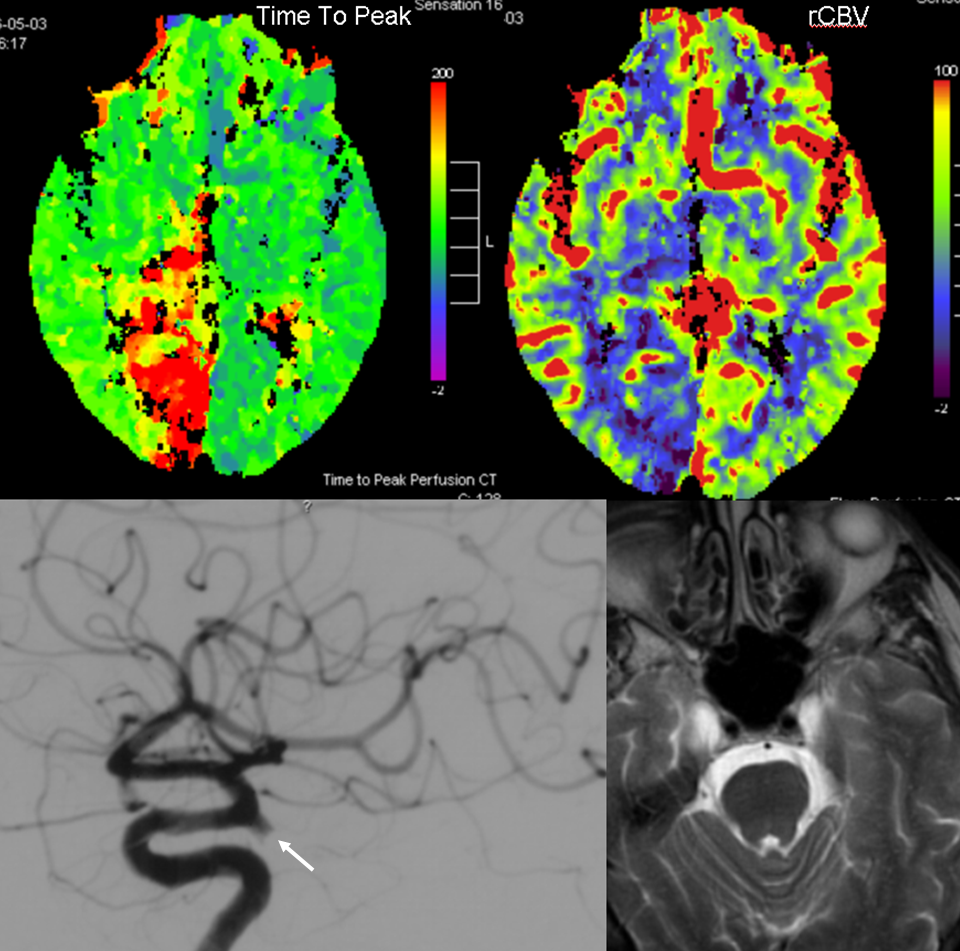
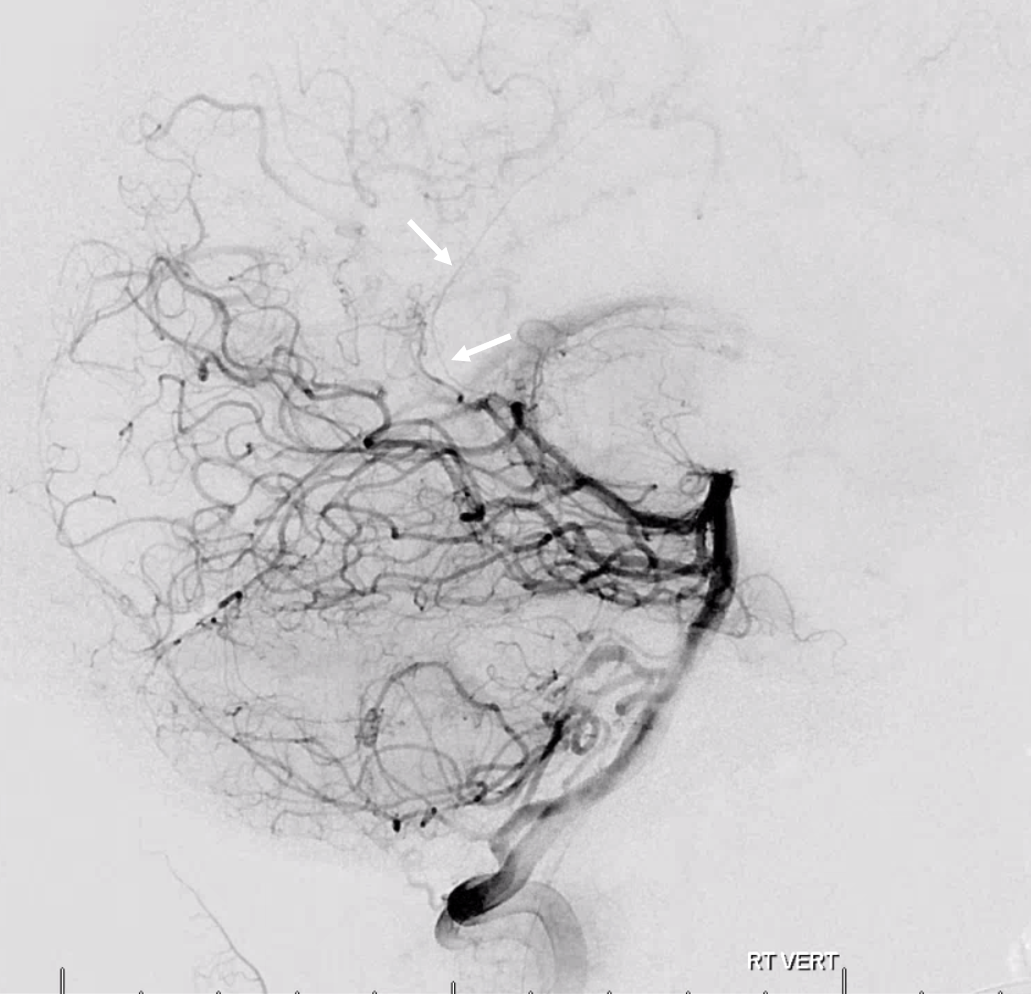
Below is an example of an embolus from the carotid artery into a fetal PCOM (white arrow). Notice markedly reduced CBV values on this CT perfusion map, indicative of a “completed” infarction with no penumbra. This, unfortunately, is too often true with occipital infarcts. The medial occipital area is not well-supported via leptomeningeal collaterals, being at the distal end of both ACA and MCA territories, and cortical visual field deficits too often show minimal to no recovery. Also appreciate the relatively “straight” shot from the supraclinoid ICA into the PCOM, suggesting that this was the hemodynamically preferable route for the embolus.
Calcarine Branch
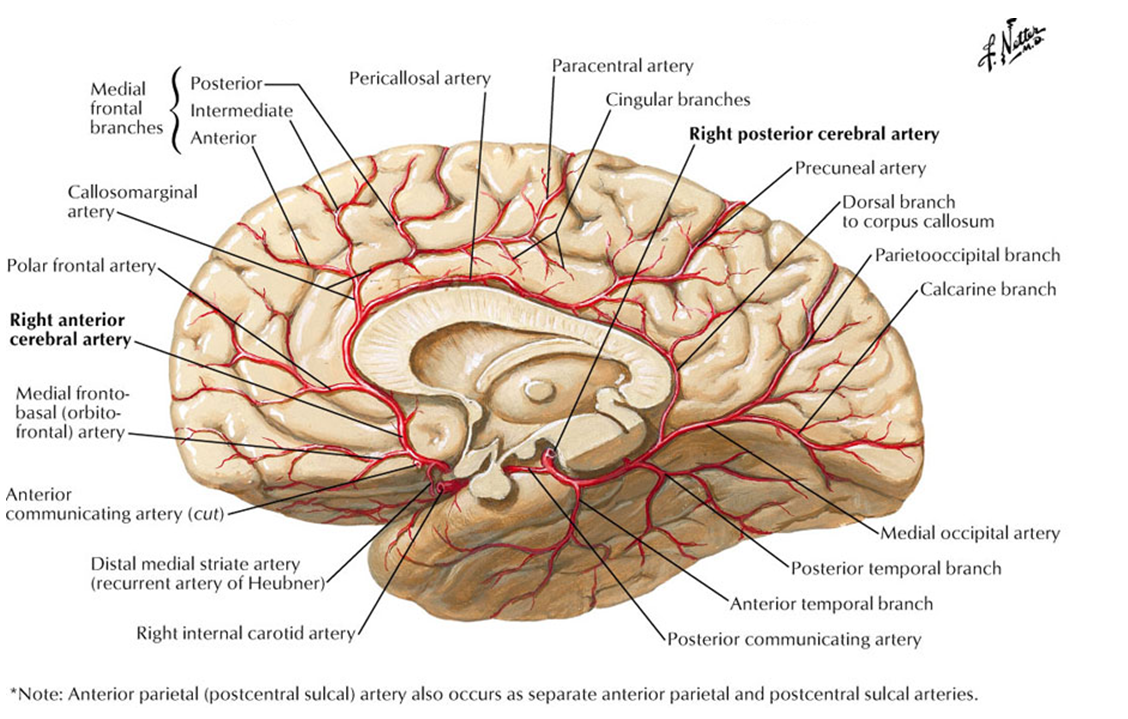
Frank Netter drawing, emphasizing inferior location of the Calcarine branch in relation to the Parietooccipital branch which is situated in the sulcus of the same name.
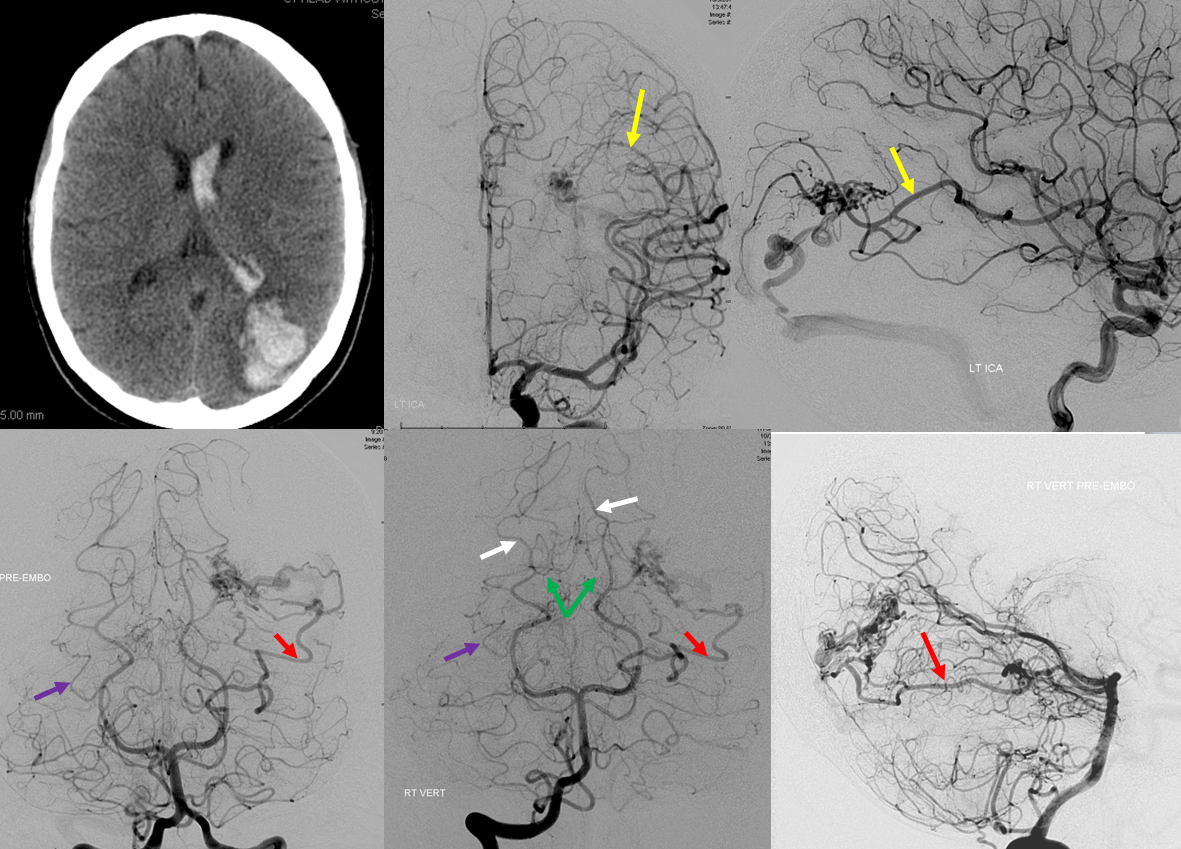
The temporo-occipital territory sits at the further edge of two potential sources of supply — anteriorly from the temporo-occipital branches of the MCA, and inferiorly from the posterior inferior temporal artery. Here is an illustration of this phenomenon, in a patient with the territory of interest demarcated by the parenchymal hemorrhage component. Both ICA and vertebral injections are required to fully delineate the AVM, supplied by the parieto-occipital branch of the MCA (yellow) and posterior inferior temporal branches of the PCA (red). Notice how far back the branch extends on the lateral view — again not to be confused with the calcarine branches (green), which are superimposed on the nidus in the lateral projection. The parieto-occipital branches (white) are medial and do not contribute to the AVM. A normal posterior inferior temporal branch is marked with a purple arrow
Leptomeningeal Collaterals in anterior circulation occlusion
The PCA can be very effective in leptomeningeal collateralization of the MCA and even ACA territories. The inferior temporal branches (green) will attempt to reconstitute the upper, perisylvian portions of the temporal lobe, while the parieto-occipital branch fills in variable territories of the superior parietal lobule, precuneus, and possibly the posterior frontal convexity, depending on whether or not the hemodynamic constraint affects the MCA, ACA, or both. In this way, the inferior temporal branches can help salvage the Wernicke area. In most cases of acute occlusion, however, PCA cortical branches are too far posterior and inferior to effectively resupply the frontal lobe, which depends on the ACA in cases of insufficient MCA perfusion.
In this ICA embolus case, the posterior inferior temporal branch (red) and middle inferior temporal branch (purple) leptomeningeal vessels help reconstitute a sizable portion of the temporal lobe (light blue oval, parenchymal phase), retrogradely opacifying several inferior division temporal (green) and inferior parietal (yellow) branches of the MCA. The parieto-occipital artery attempts to revascularize the cuneus, reconstituting a superior mesial parietal branch (white) of the ACA (purple arrow). The extent of collateral support in the temporal lobe territory is fairly robust. The posterior pericallosal artery (black arrow) is normally a very poor collateral to the distal pericallosal (light blue) territory of the ACA. In this case, a small leptomeningeal network (pink) is trying its best. Notice normal-appearing posterior (gray), middle (brown) and anterior (orange) inferior temporal branches on the right.
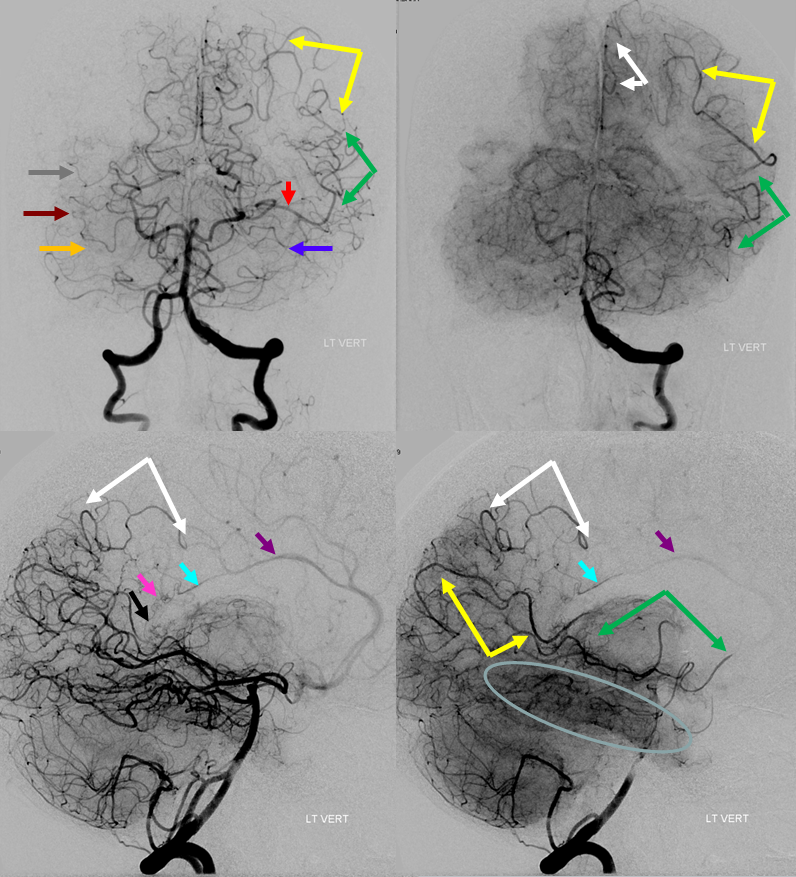
Here is another example of rather effective leptomeningeal collateral response through the parieto-occipital territory supporting the superior parietal lobule (red) and great inferior temporal support of the MCA inferior division (green). Nearly the entire temporal lobe is adequately perfused.
Posterior Pericalossal-Pericalossal Anastomosis
As mentioned above, in the acute setting the connection between the posterior pericalossal branch of the PCA and the distal pericallosal branch of the ACA is rather inadequate for meaningful reperfusion of either vessel by the other. However, any slowly progressive constraint is another matter. The typical scenario is Moya-Moya, where very proximal bilateral MCA and ACA constraint calls the PCA into action. However, it need not be Moya-Moya — any slowly evolving process will do.
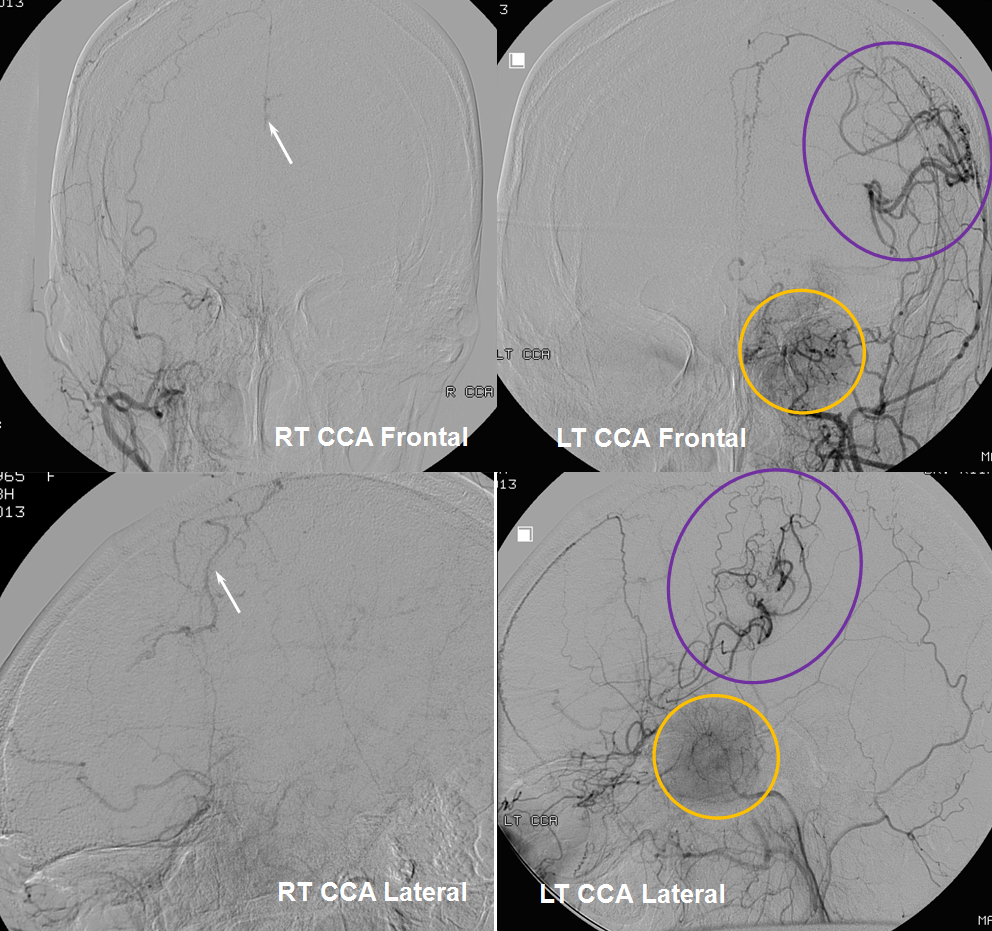
In the following patient, a giant sphenoid wing meningioma resulted in occlusion of both supraclinoid ICAs, similar to a Moya-Moya pattern. Therefore, the primary method of reconstitution is via leptomeningeal PCA-MCA (purple arrows) and PCA-ACA (light blue arrows) collaterals, the posterior to anterior pericallosal (yellow) anastomosis, and left more than right middle meningeal artery auto-synangiosis with the MCA territory on the left (motor strip, purple oval) and right MMA to left ACA territory as well (white arrows). Notice meningioma tumor blush (orange oval)
Anterior Choroidal Artery Capture of Posterior Inferior Temporal Territory
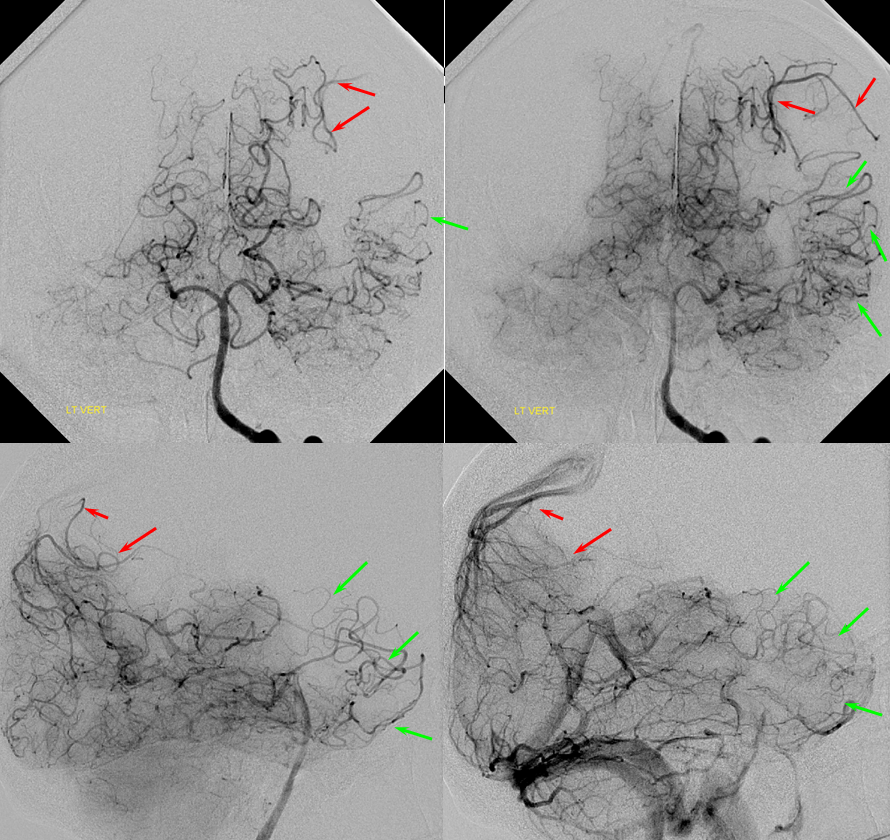
Supply of the Cerebral Peduncle and Quadrigeminal Plate — Collicular (Quadrigeminal) Artery and Variants
The P2 segment of the PCA swings around the cerebral peduncle, underneath the thalamus, towards the quadrigeminal plate, an further dorsal towards the occipital area. Branches of the PCA supply the thalamus (inferior medial and lateral thalamus – geniculate area), the peduncle, and the collicular plate. There is wide variation in the description of this supply. Sometimes it is depicted as perforators arising directly from the P2 segment, which makes sense geographically. For example, see diagram from none less than Netter below:
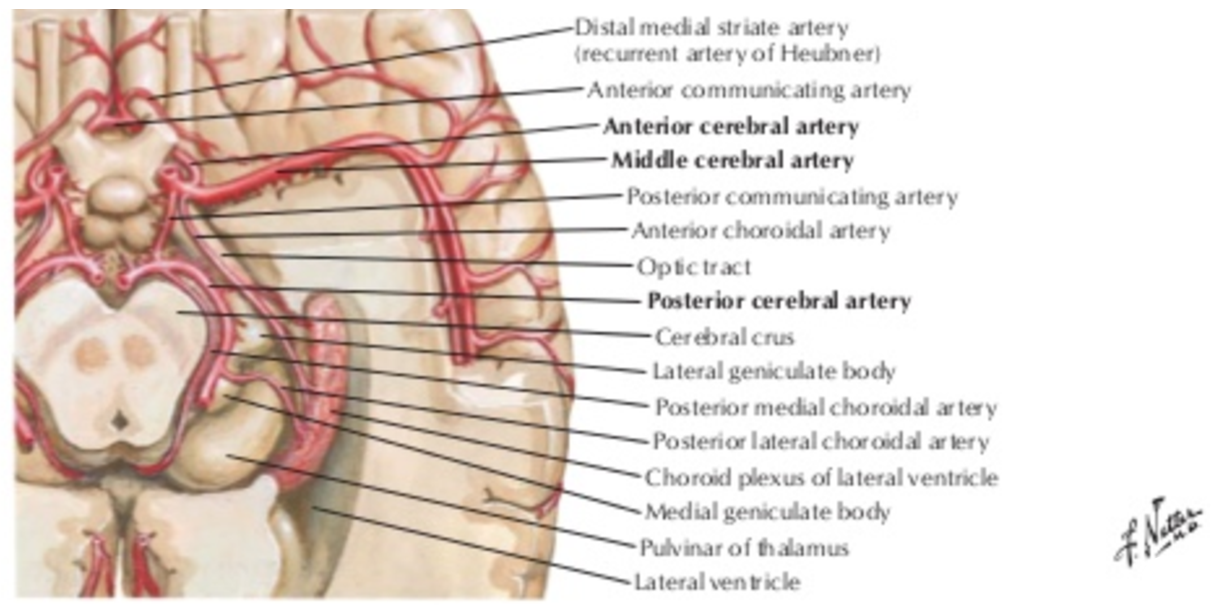
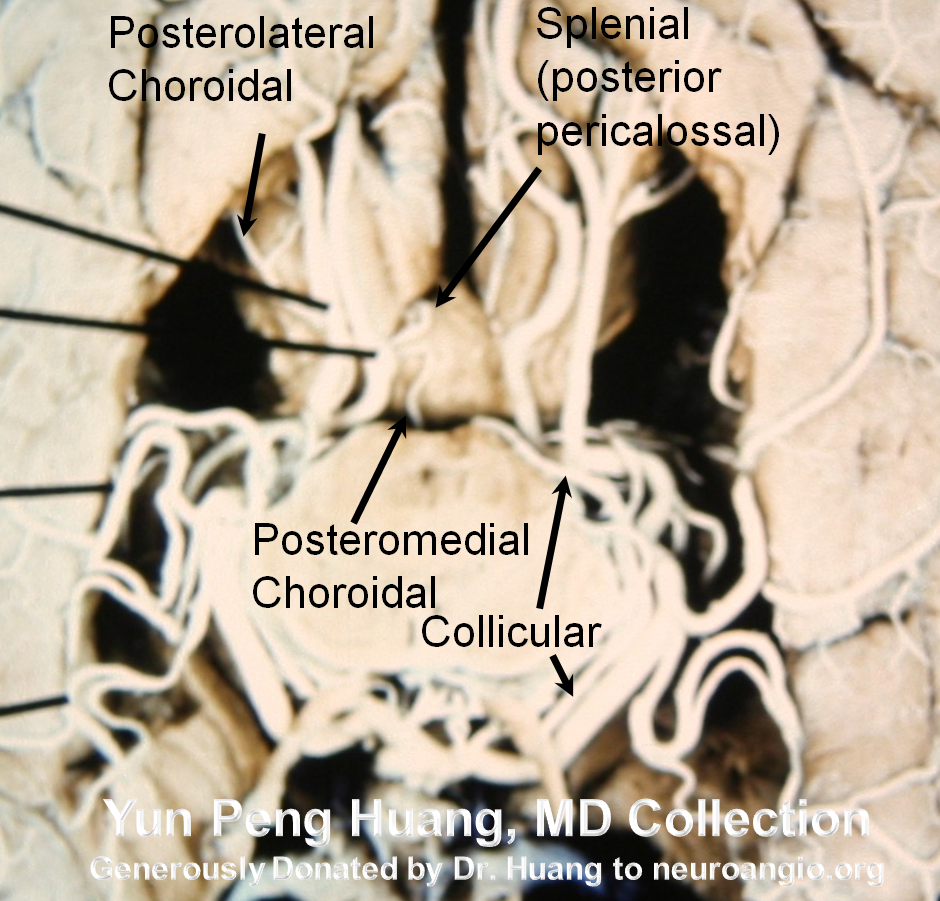
Others describe a stand-alone branch arising from P1 or proximal P2 segment, — the Collicular (or Circumcollicular) artery — and courses just medial to the main P2 segment, around the cerebral peduncle, towards the collicular plate (hence the name). The artery (red) is beautifully depicted in this specimen x-ray from the Yun Peng Huang collection. Perforators to the peduncle (pink) are also visualized:
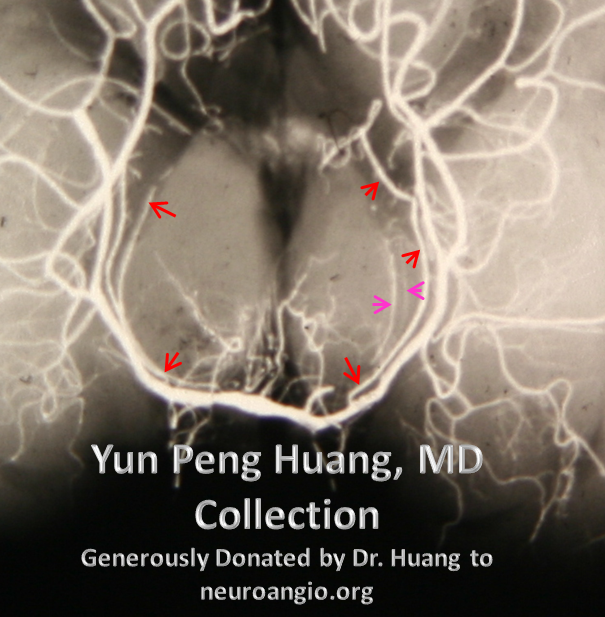
Photo specimen, as above:

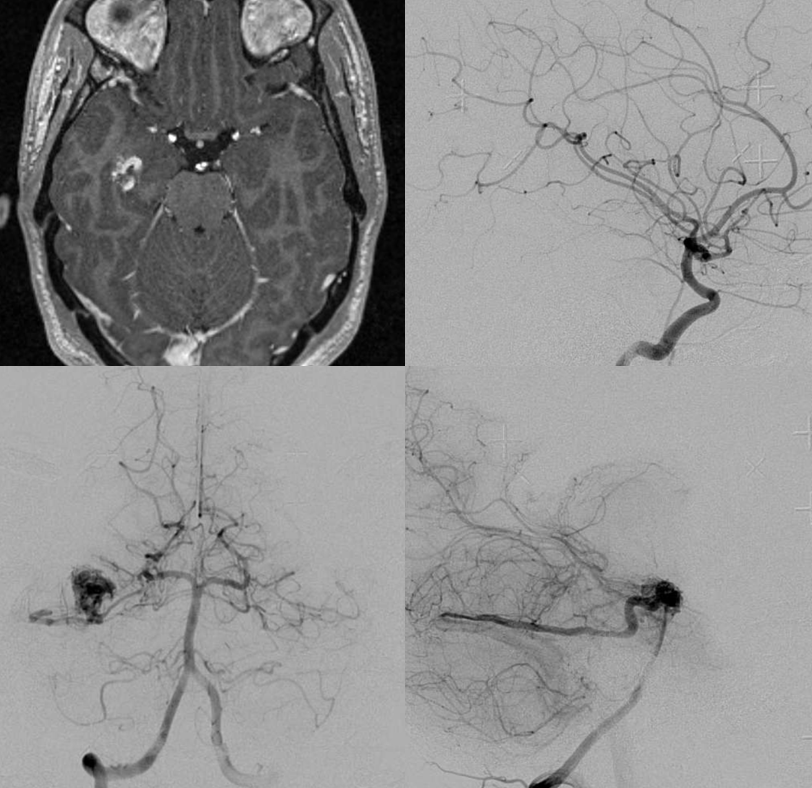
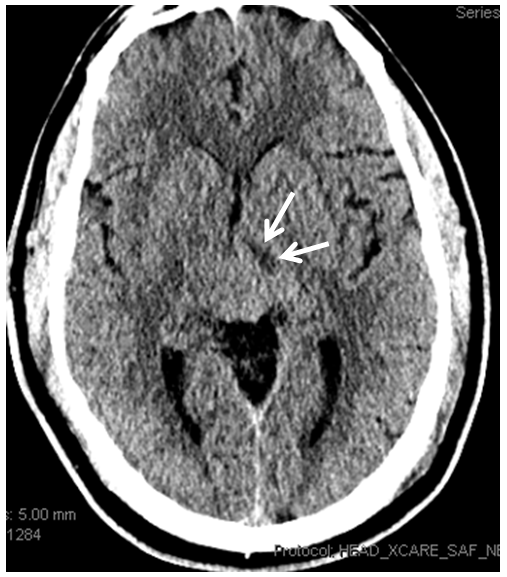
The picture is one of large P2 and slender collicular vessel just medial to it, often too small and too superimposed on the PCA to be individually resolved on any modality. Its importance comes from the territory it supplies — the cerebral peduncle and quadrigeminal plate. Thus, damage to the artery can lead to one instance of PCA-related hemiparesis, as seen in the image below with a developing infarct in the cerebral peduncle:
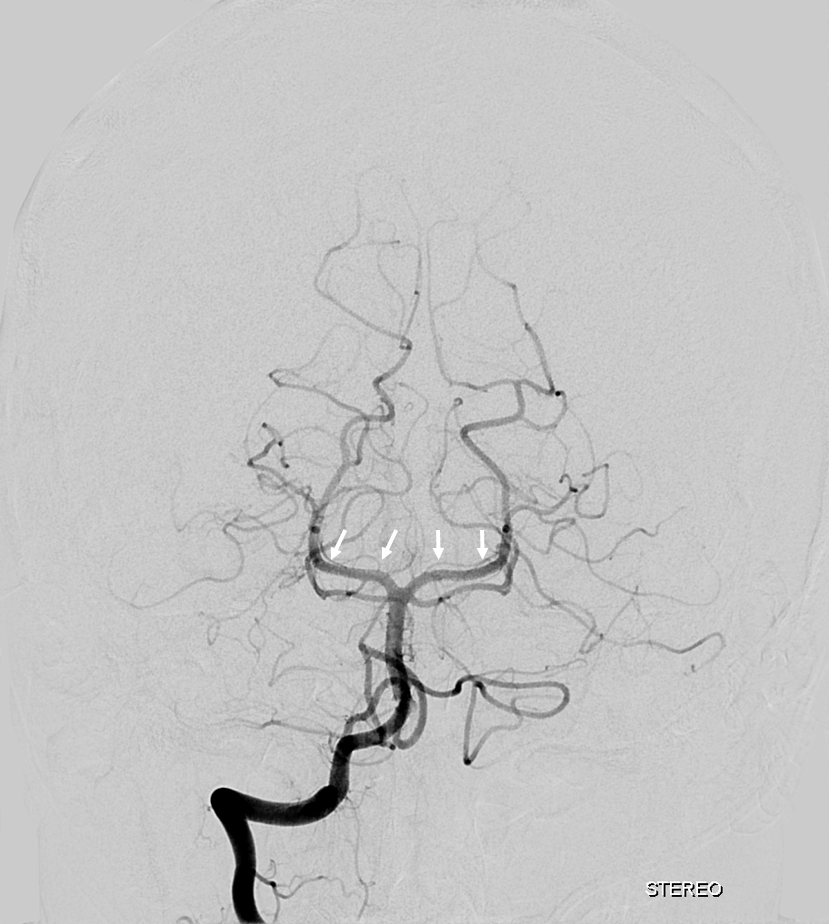
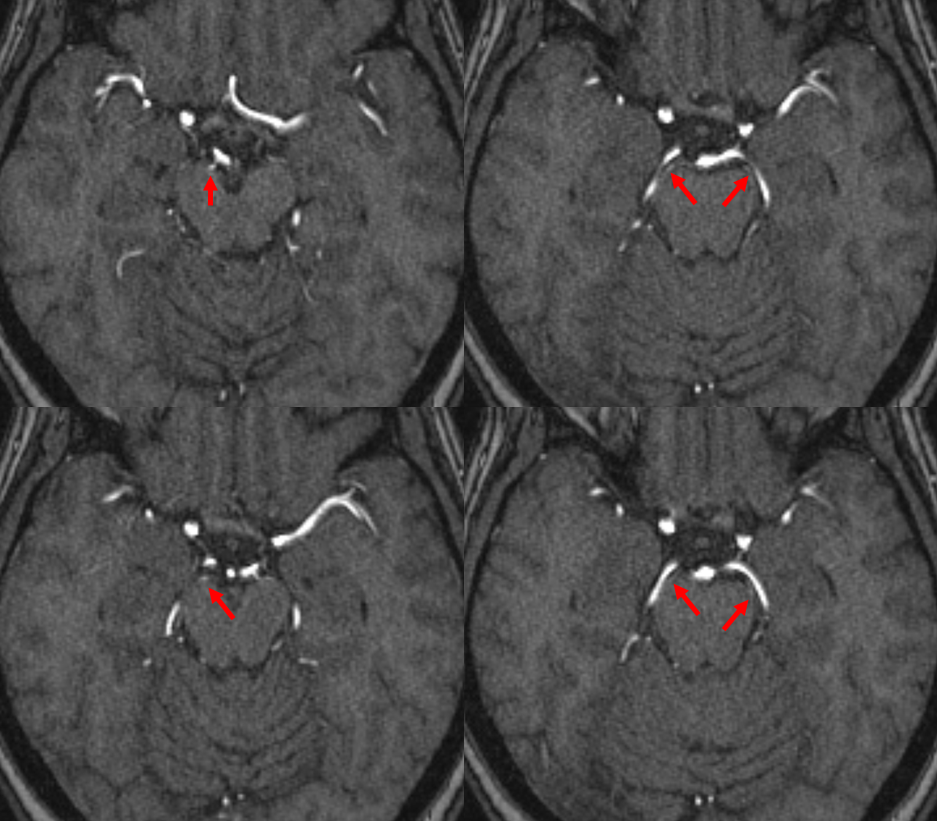
At least on some occasions the Collicular artery can be resolved, both with MRA and angiography. Here you can see these bilaterally (white arrows)
Cross Eye Stereo Pair
Anaglyph
Here is another example of one (red arrows), located just medial to the P2 segment.
Here is an MRA of a different patient, with the same artery seen bilaterally. Could this be the posteromedial choroidal artery instead? Certainly. That’s what Netter would say.
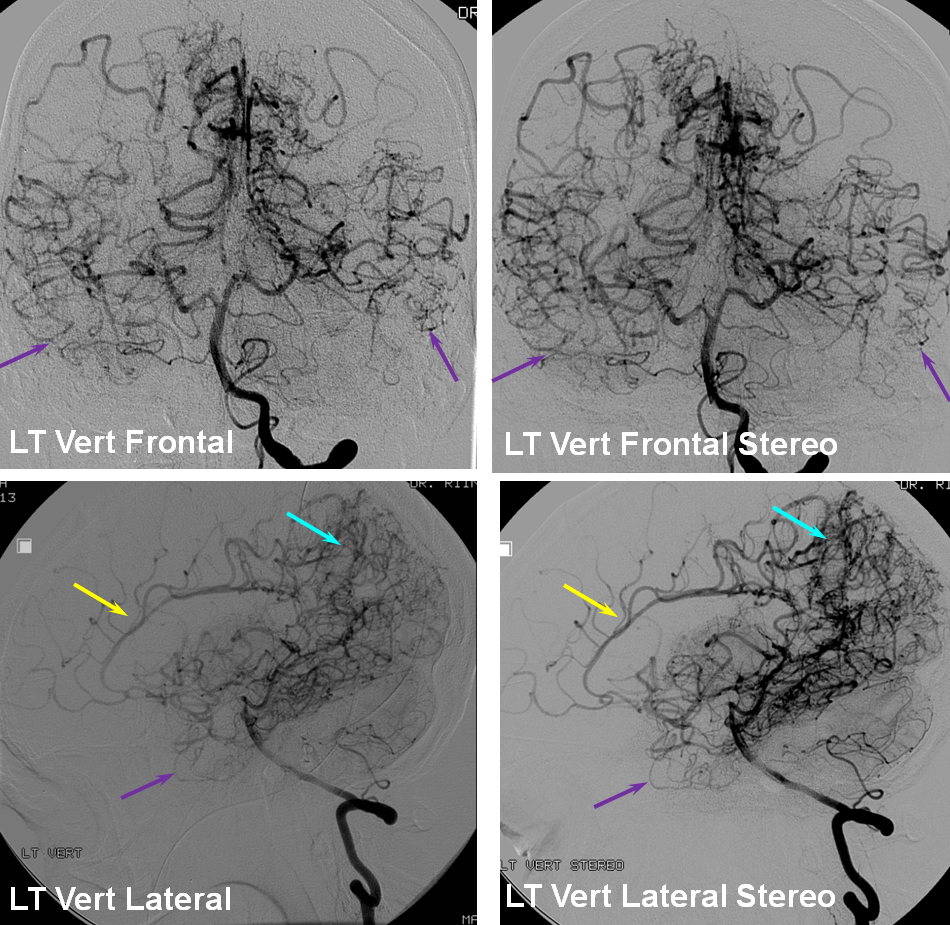
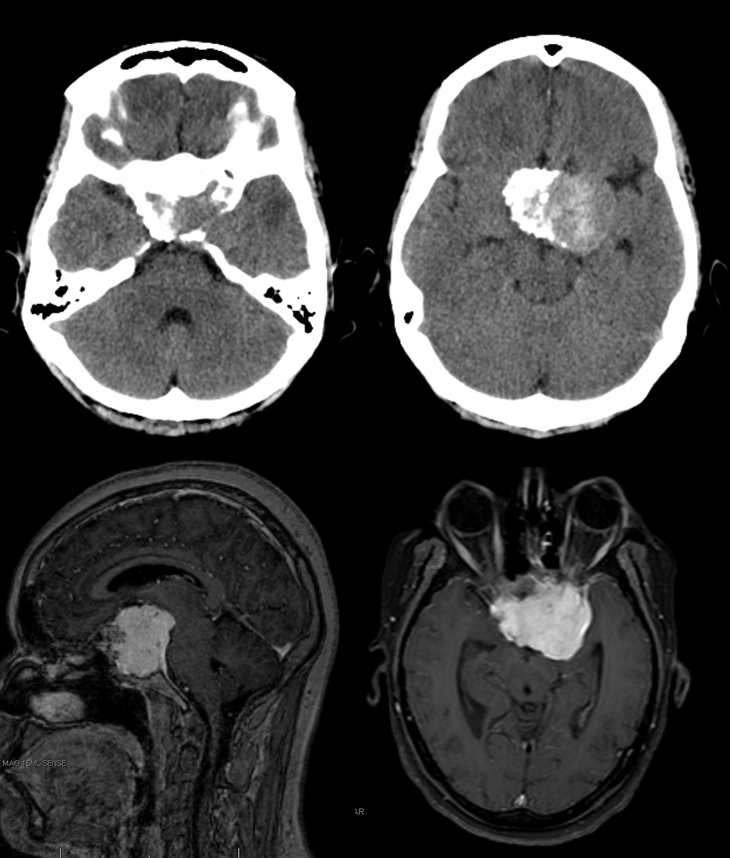
Here is an example of a focal hemorrhage in the quadrigeminal plate:
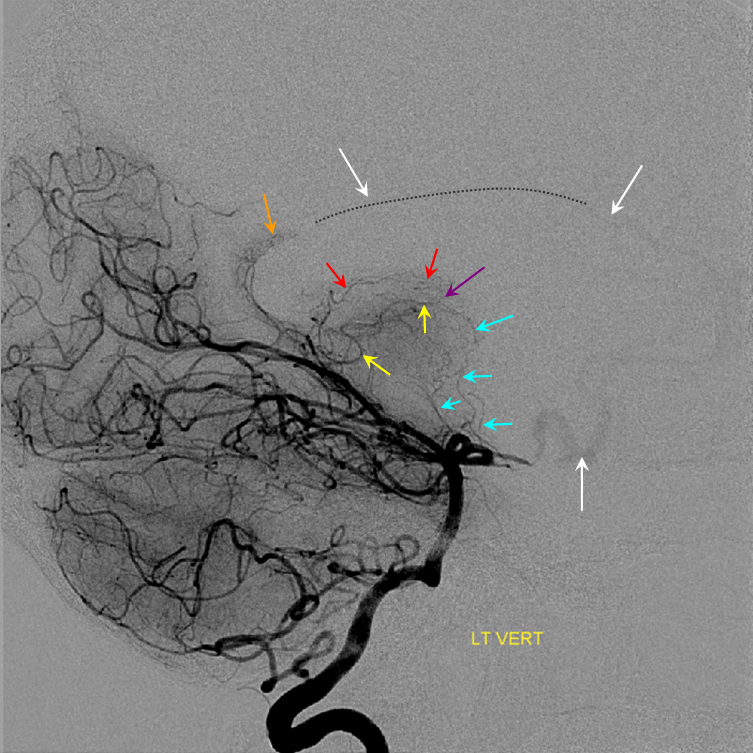
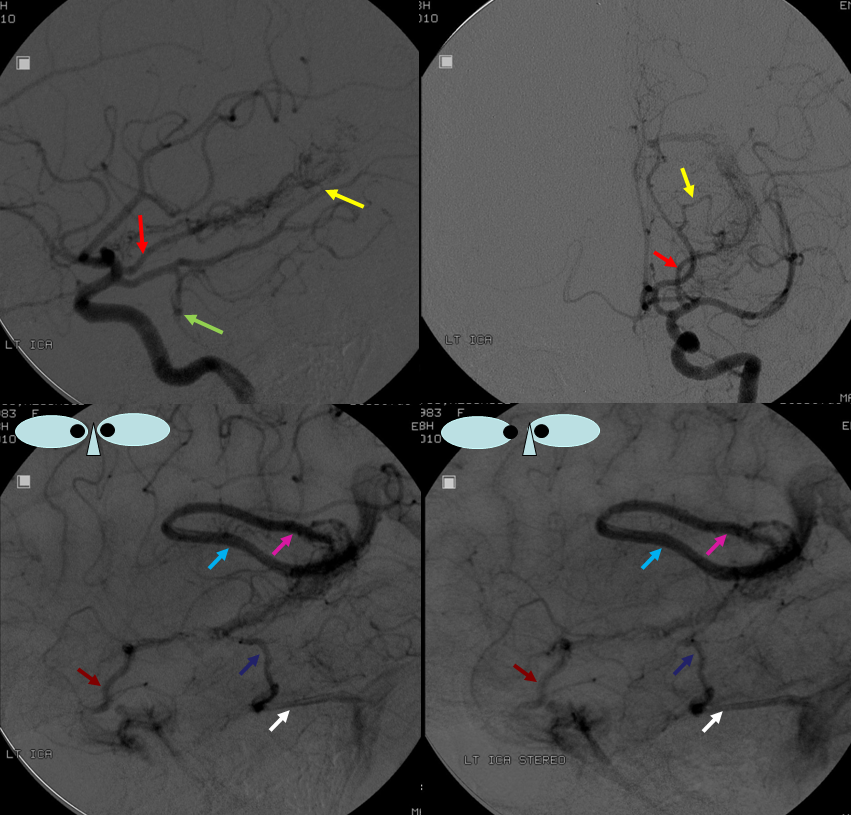
Angiography, performed because we didn’t want to settle for a cavernoma, shows a shunt in the the region of the quadrigeminal plate, with premature opacification of the Precentral Vein (purple)
Delayed angiography in frontal and lateral planes better displays the shunt following resolution of the hematoma, now faintly shows some superior vermian veins (light blue), and the Collicular Artery (red). Also note medial thalamic perforators originating as a single trunk, known as the artery of Percheron (orange)
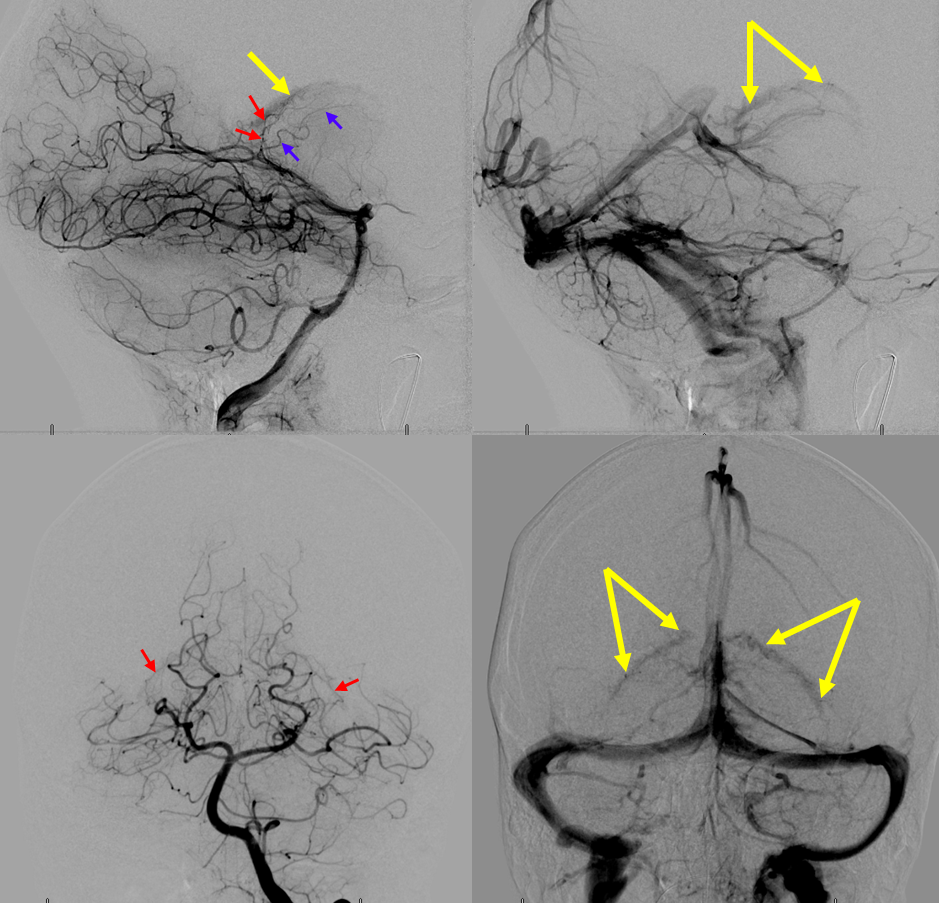
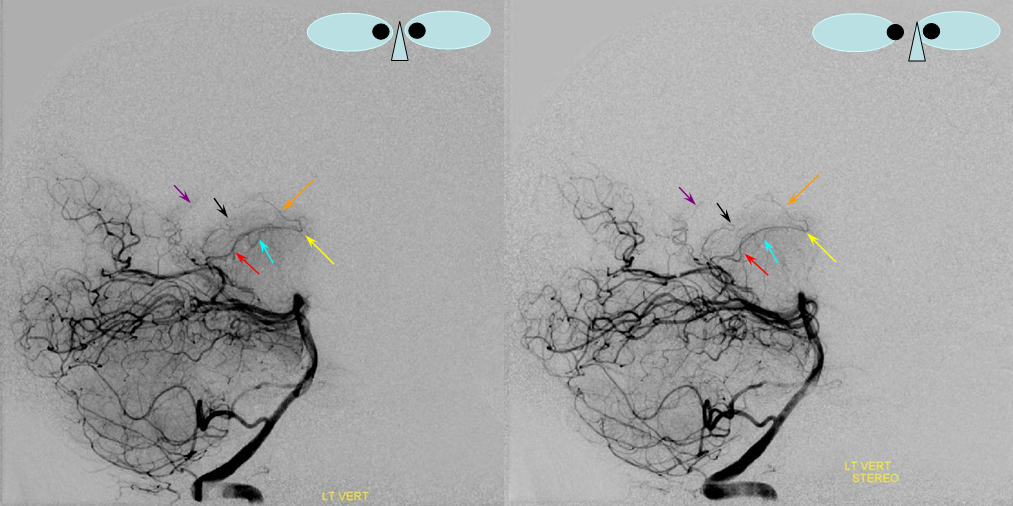
Superselective angiography with the microcatheter at the ostium of the Collicular artery (red) demonstrates perfectly its course outlining the cerebral peduncle, with a small AVM (white) at the collicular plate, draining via the Precentral Vein (purple) into the straight sinus (dark blue), and retrogradely congesting paired superior cerebellar veins (light blue). Notice several slender perforators to the peduncle (pink). The silly Percheron is orange. The top pair of frontal images is stereoscopic
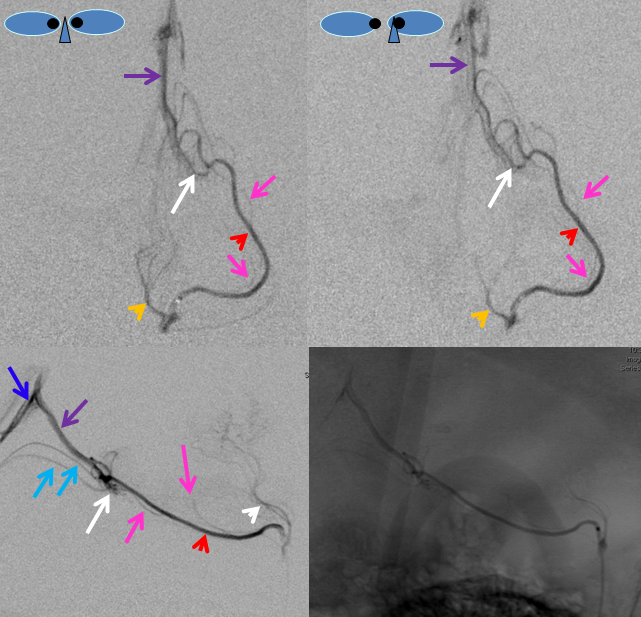
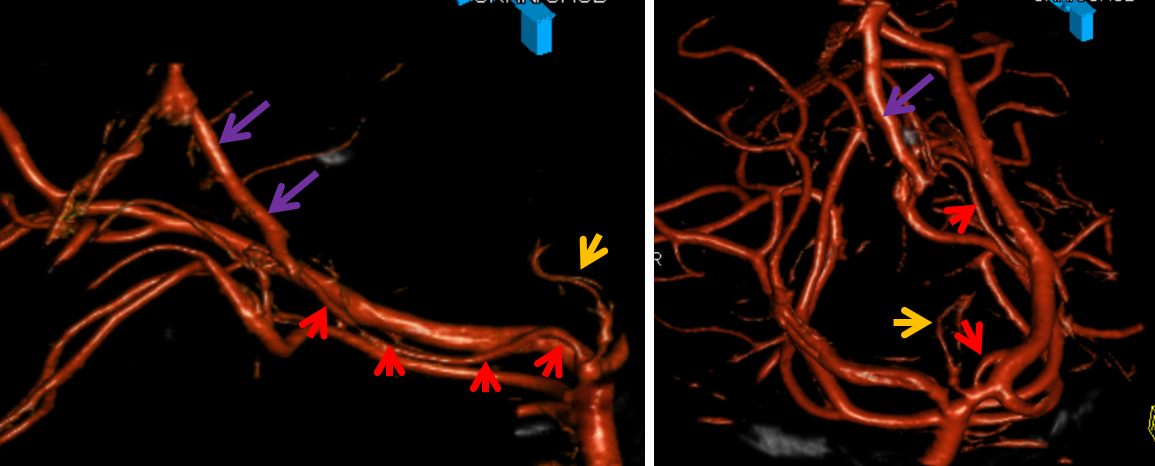
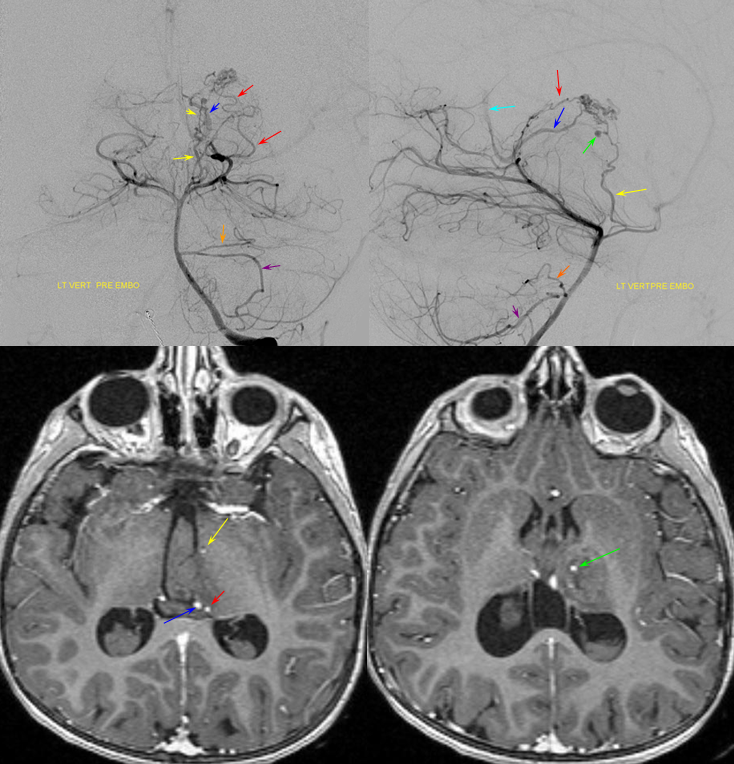
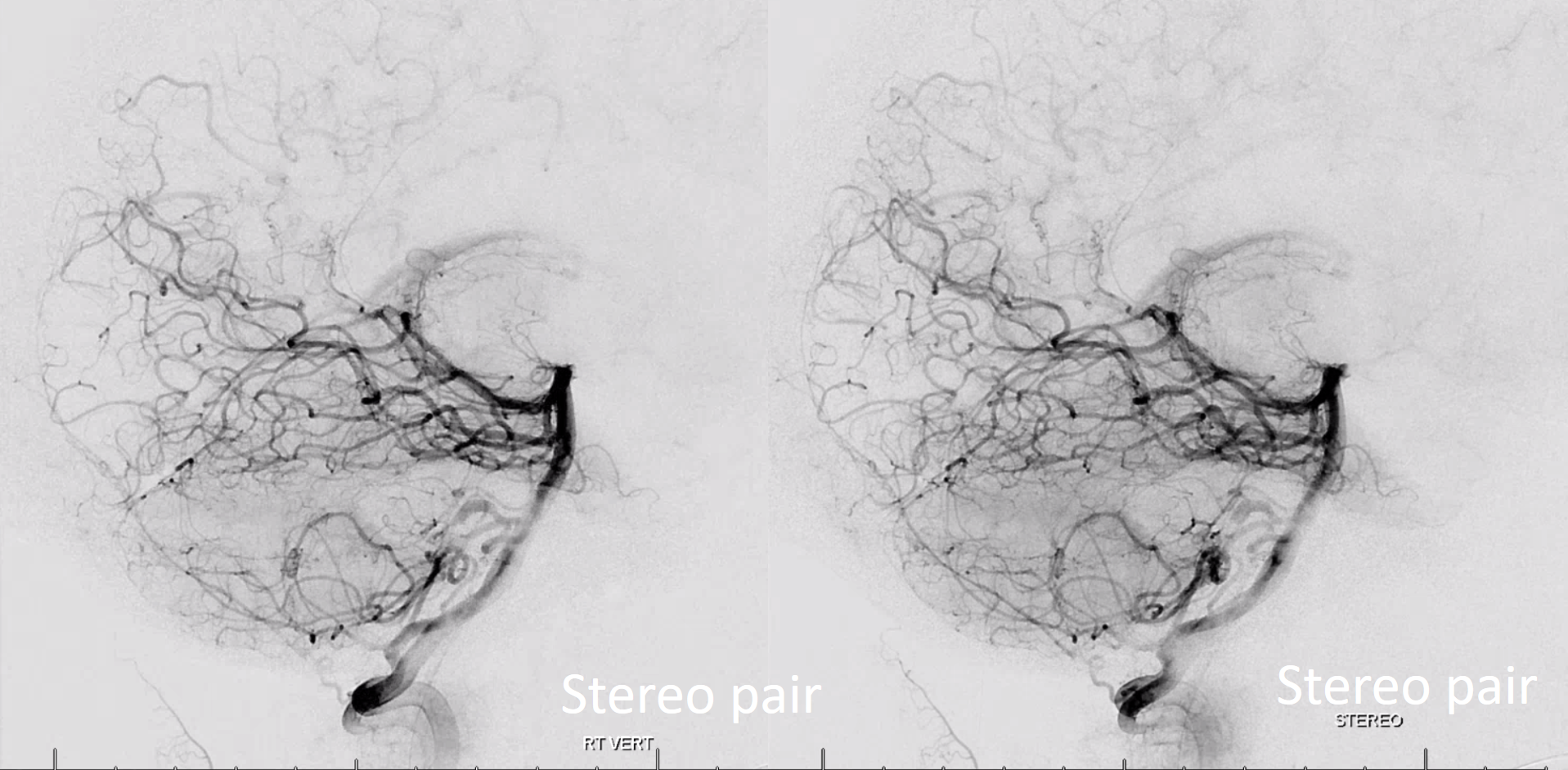
3D-DSA angiographic views demonstrating the relationship of the Collicular artery (red) to the much larger P2 segment of the PCA. Even in its AVM-related enlarged state, the Collicular artery remains slender and difficult to differentiate from the P2 segment on nonselective vertebral angiography. No wonder it is so often missing from descriptions. I hope that this case, and the image of the stroke, are convincing enough.
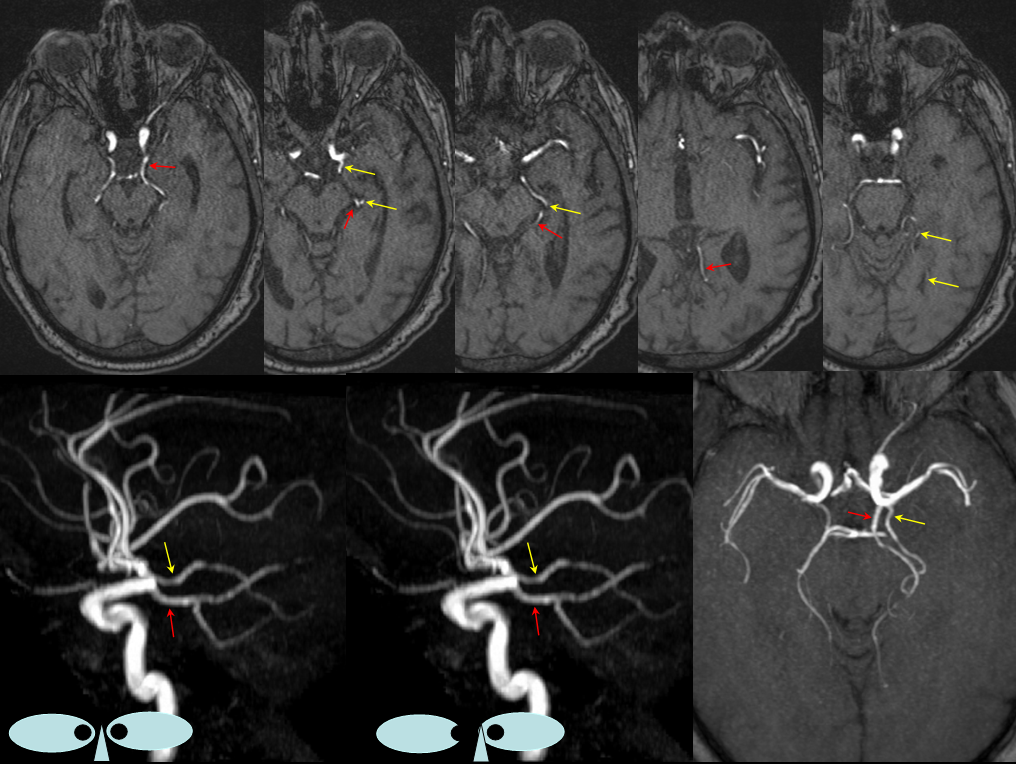
I think that it is best to think of the Collicular artery as a perforator. For example, one sometimes finds a common trunk for a number of lenticulostriate perforators from the MCA, though more often they originate separately. It may be that the Collicular artery is an example of one such common trunk, while at other times perforators to the peduncle could arise from the P2 segment (geniculate branches shown in Netter for example are direct P2 perforators). Along the same lines, medial thalamic perforators sometimes originate as a single trunk, with a totally out-of-proportion fame as the “artery of Percheron”, or come off separately with the exact same effect and zero fanfare (white arrows)
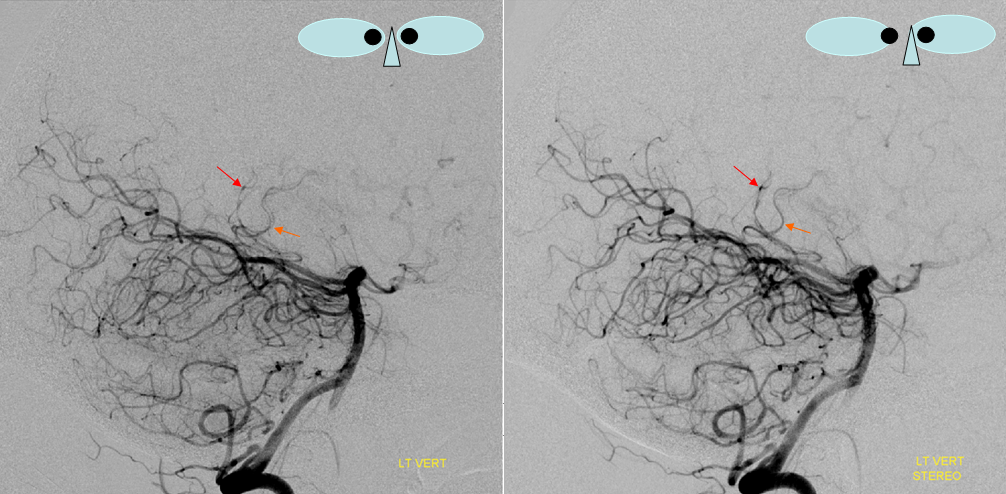
Below is another example of the Percheron (white), a detail from one of the images shown above. The hypoplastic left P1 is orange.
Posterior Lateral and Medial Choroidal Arteries
These important arteries — see dedicated Posterior Choroidal Arteries page here — supply the choroid plexus of the third and lateral (together with the anterior choroidal) ventricles. A few simple and helpful observations:
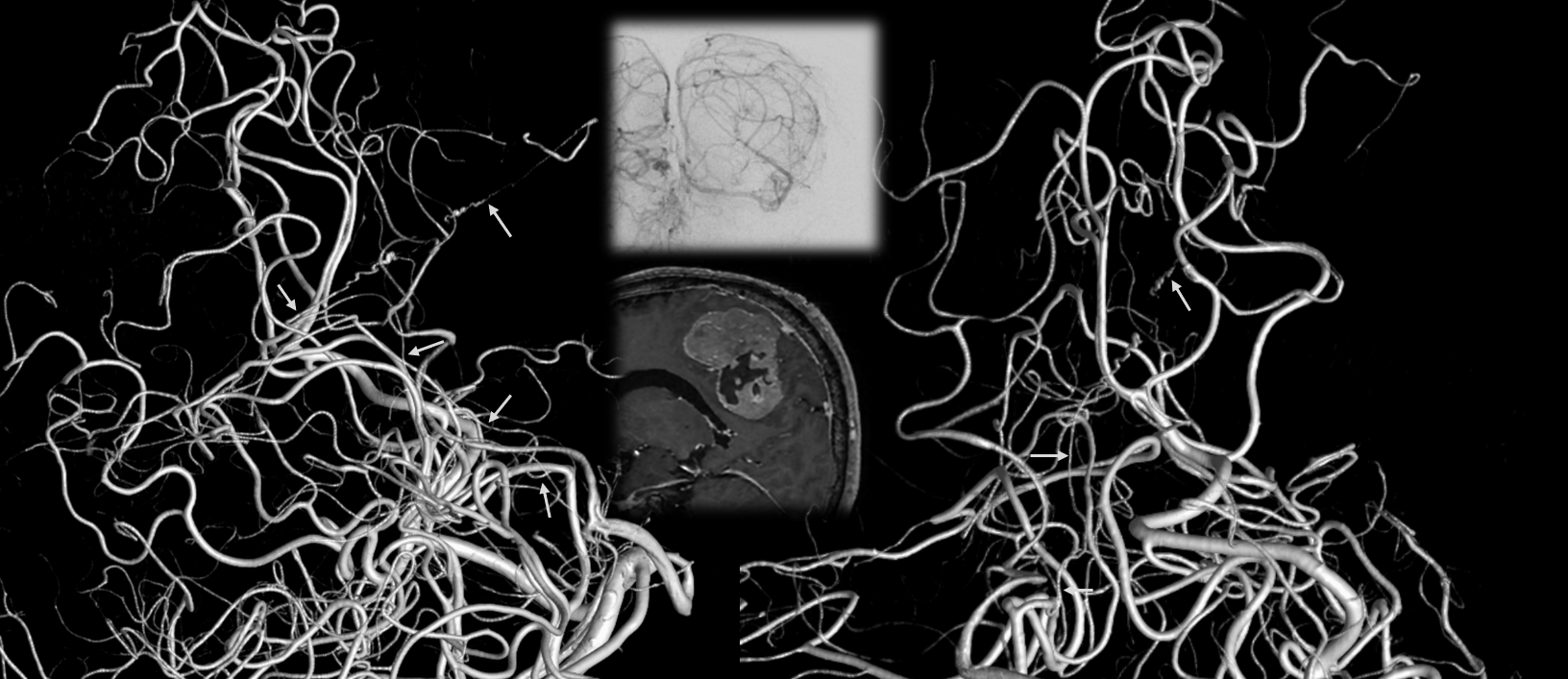
– the atrium of the lateral ventricle is located above the third ventricle. Therefore, the lateral choroidal artery (red) will be situated above the medial choroidal (purple) on lateral views. Notice, in this case, the relatively intense and persistent staining of the lateral ventricular choroid plexus (yellow), well into the late venous phase. In isolation, it is of no clinical significance.
– the atrium of the lateral ventricle is located on roughly the same level as the occipital lobe, and much more anterior. Therefore, a submental or Caldwell view will elevate the lateral choroidal artery (white, image below) above the hemispheric branches of the PCA for a less obstructed view — one of the key uses of a Caldwell in the posterior fossa

Stereo Lateral below, again showing the lateral choroidal above the medial one; Red=posterior lateral choroidal; Orange=posterior Medial choroidal (bilateral are seen on the left image) The lateral choroidal has an inverted C-shape. The medial choroidal usually has a more S-shaped feature
The Davidoff-Schechter
One of those “famous” eponyms. A dural artery arising from the PCA — in our experience from the Posterior Medial Choroidal specifically — near the falcotentorial junction. Why do PCA and SCA seem willing and able to form a pial-dural anastomoses in this area? Our theory is that the falcotentorial junction is a kind of dural “watershed” — an area away from the main dural branches such as middle, posterior, and anterior meningeal arteries. The area is thus supplied either by terminal branches of these vessels, or by a piodural origin branch — the Davidoff-Schechter.
From this, one might also see how Davidoff-Schechter is really not a single artery — it is an expression of a tendency for formation of pial-dural anastomoses in this area. Sometimes you can see a little artery heading into the falx cerebri from PCA or less commonly the SCA (SCA origin dural branch is called Wollschleger and Wollschleger) — a dural branch. This vessel or more appropriately vessels are most frequently seen in dural fistulas of the falcotentorial junction. There can be hundreds of little Davidoffs and Schechters. Again — in this sense the artery is not a single thing — its an idea that a pial-dural anastomosis here is possible, in all forms.
Here is a simple classical Davidoff-Schechter
Stereo pair
Anaglyph
Another example below
Technique is of course important. its a small artery. also, its dural — which means that its vascular bed resistance will be higher than brain as a rule. which means that it will be seen in a later phase than brain arteries. thats why recurrent meningeal (Ophthalmic artery origin of the MMA) is best seen in late arterial or parenchymal brain phases of the ICA injection. Here its the same — see below: arterial phase (left) shows nothing. Parenchymal brain phase shows both the Davidoff-Schechter (PCA origin, solid arrow) and Wollschleger-Wollschleger arteries (SCA origin, projecting inferiorly, dashed arrows)

without labels below
As you might expect, HR-CBCT is much better at visualizing this artery than 2D-DSA. Here is an example — bottom row are axial MIPs tracing the artery to posterior medial choroidal.
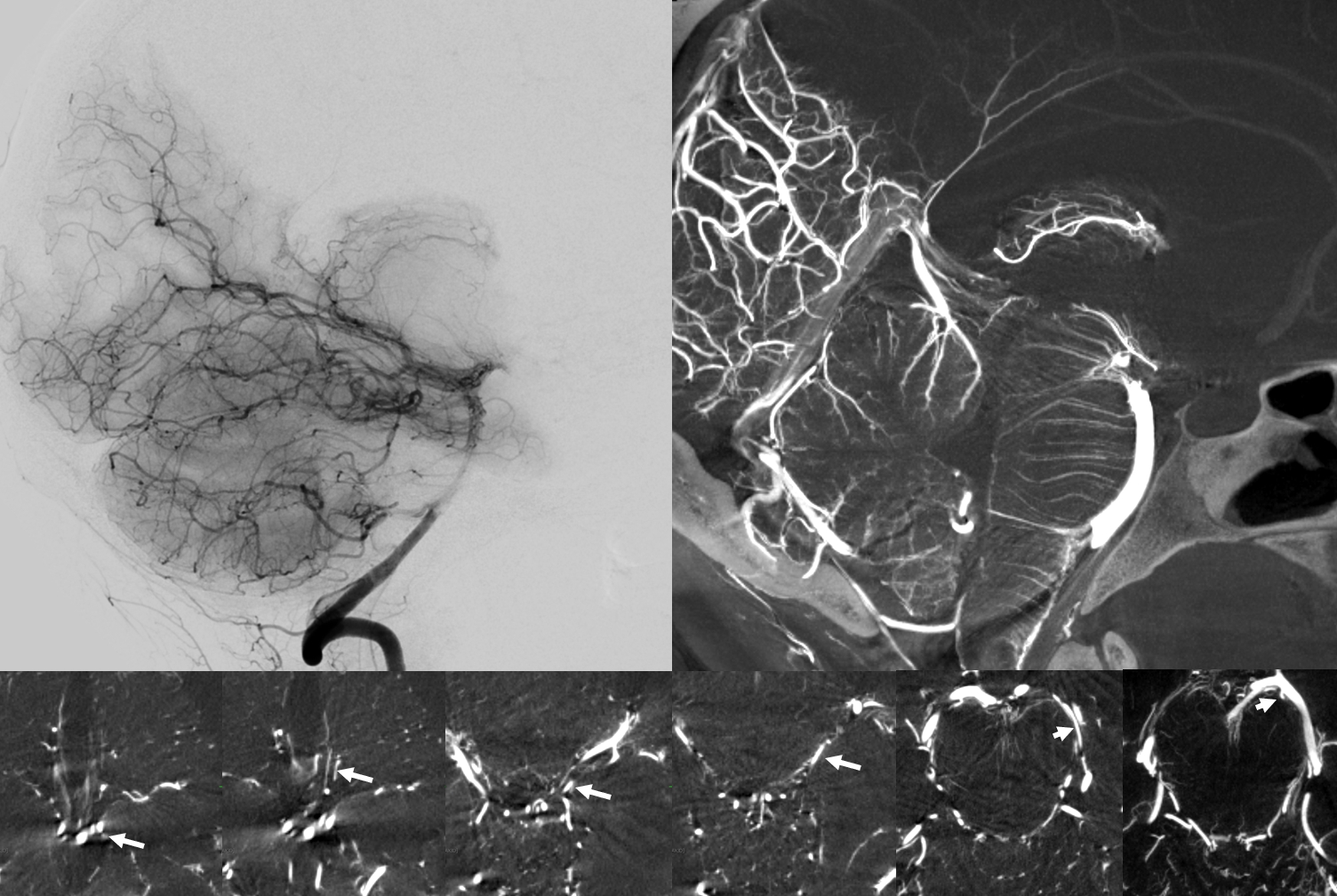
Another Example — also from posterior medial choroidal — and of course also much better seen on HR-CBCT
Stereo pair below — solid arrows — Davidoff-Schechter. Dashed arrows — posterior medial choroidal. The loop area of Davidoff-Schechter is where it picks up the dura
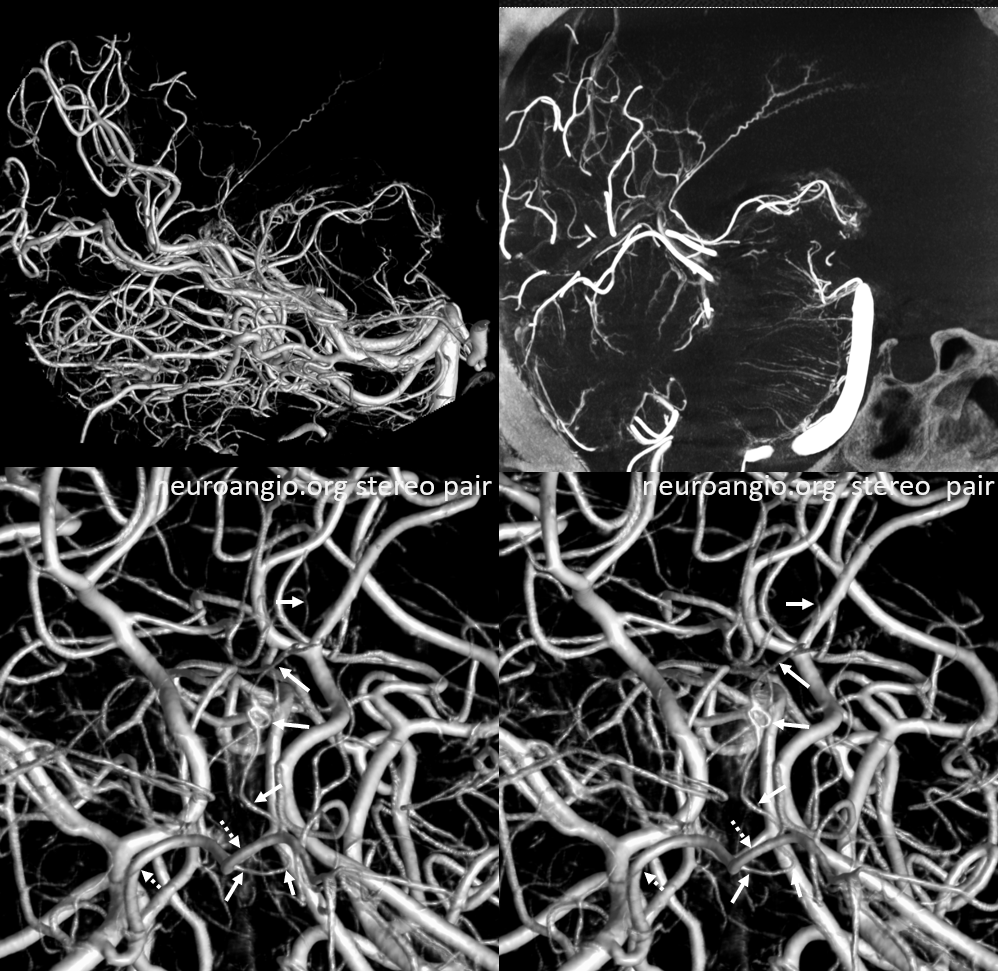
Davidoff-Schechter in Disease — Dural Fistulas
As we said, there is no one Davidoff-Schechter. This artery simply reflects the propensity of pial-dural anastomoses to form in this area. They can be seen with dural fistulas, AVMs, etc — and there might be hundreds of Davidoff-Schechters in the area….
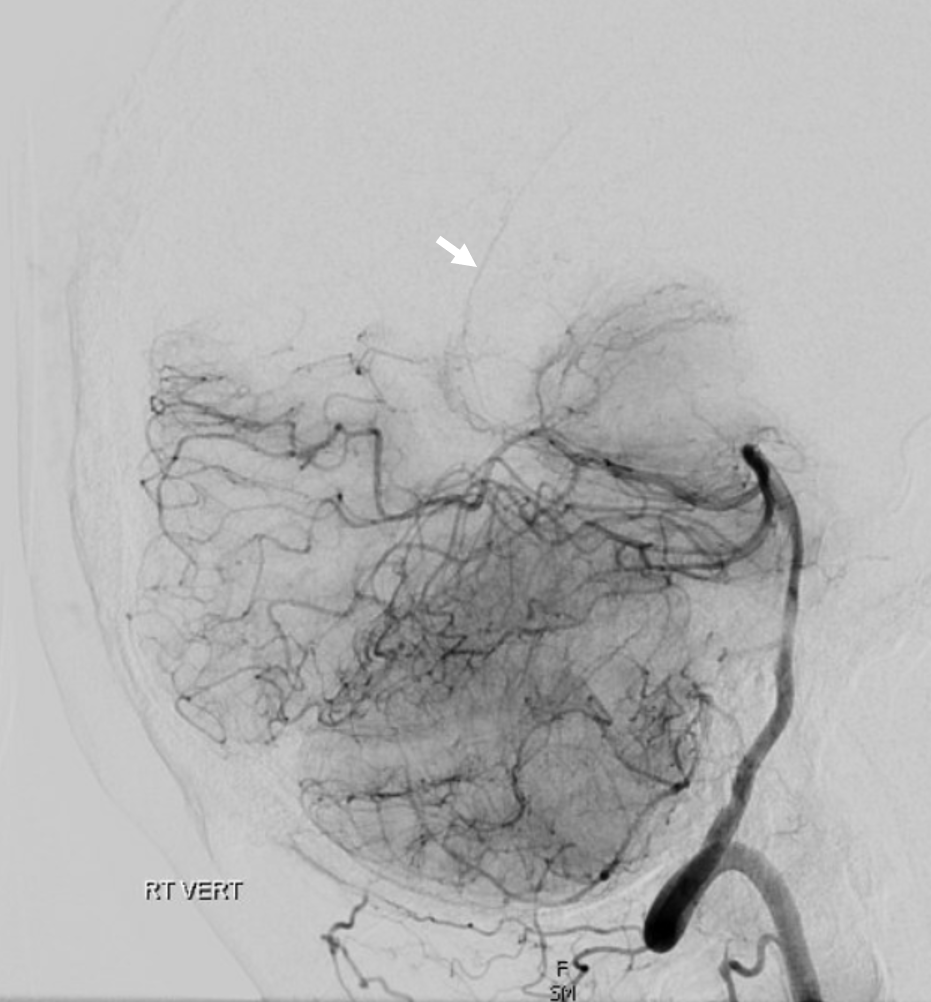
Below is a patient with a large petrous ridge (NOT sigmoid sinus) dural fistula
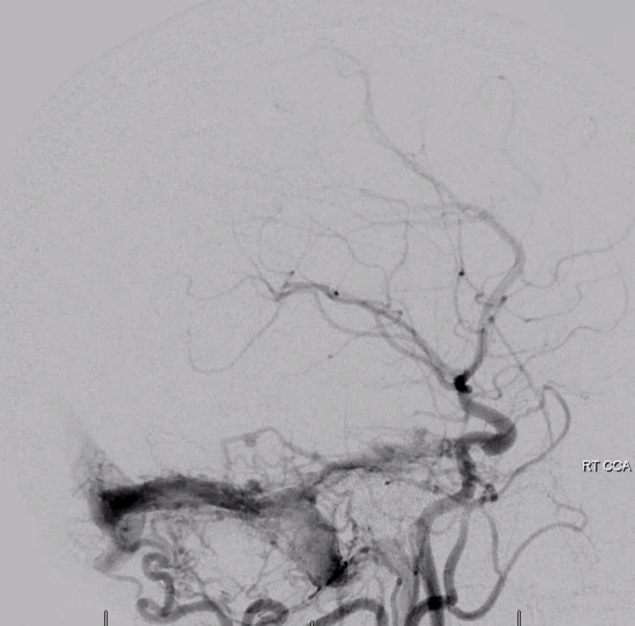
Check out this occipital injection pharyngo-occipital variant jugular division (white arrows)
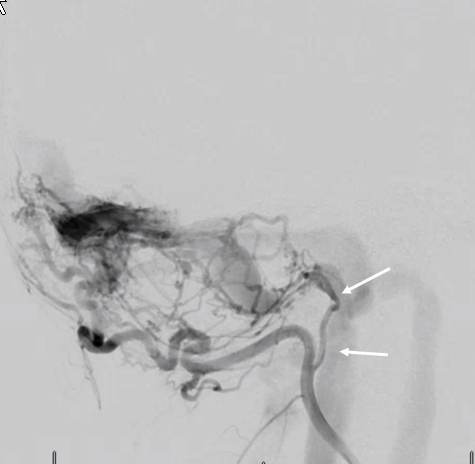
Here is the Davidoff-Schechter dural branch supplying the same fistula (white arrows). Look at beautiful notion of choroidal ring (black arrows) as well
Anaglyph stereo
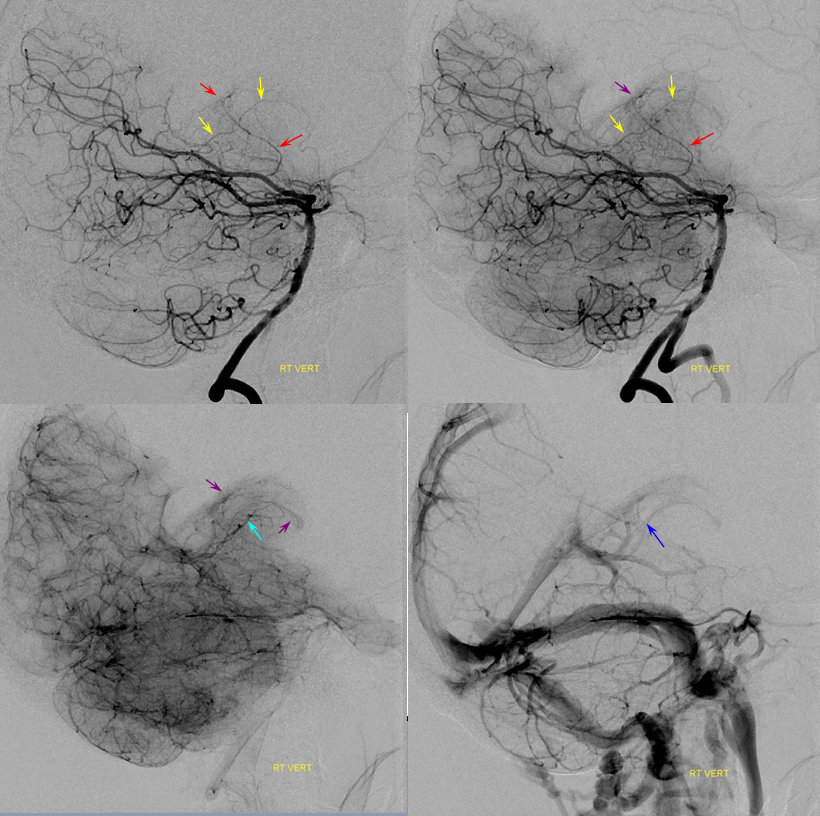
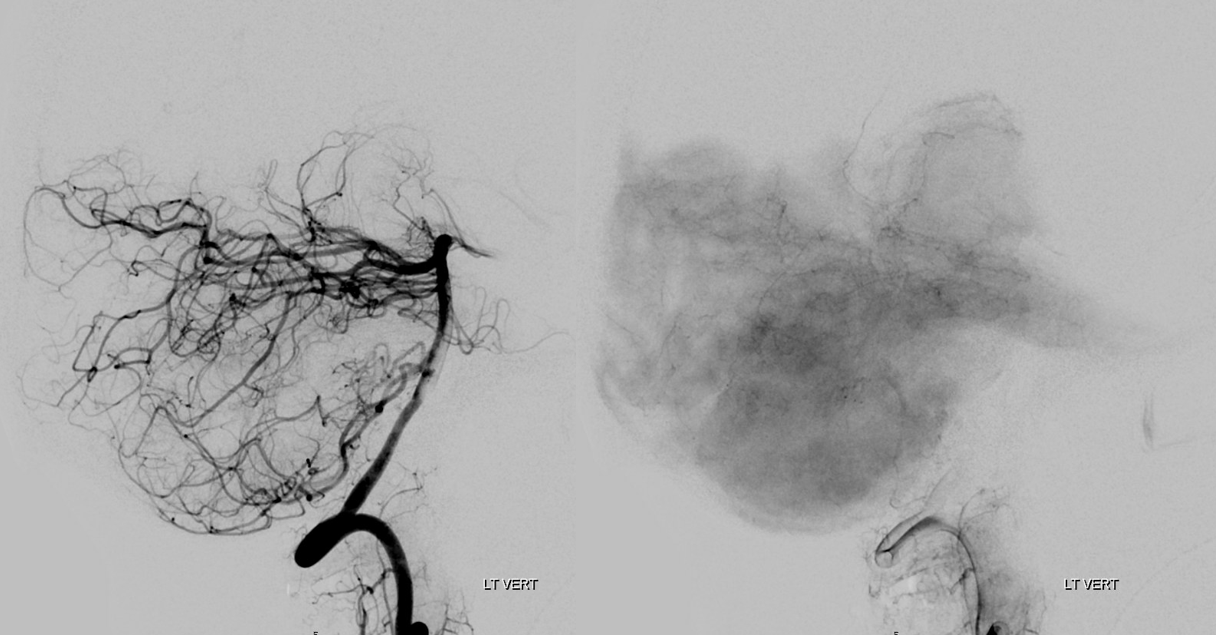
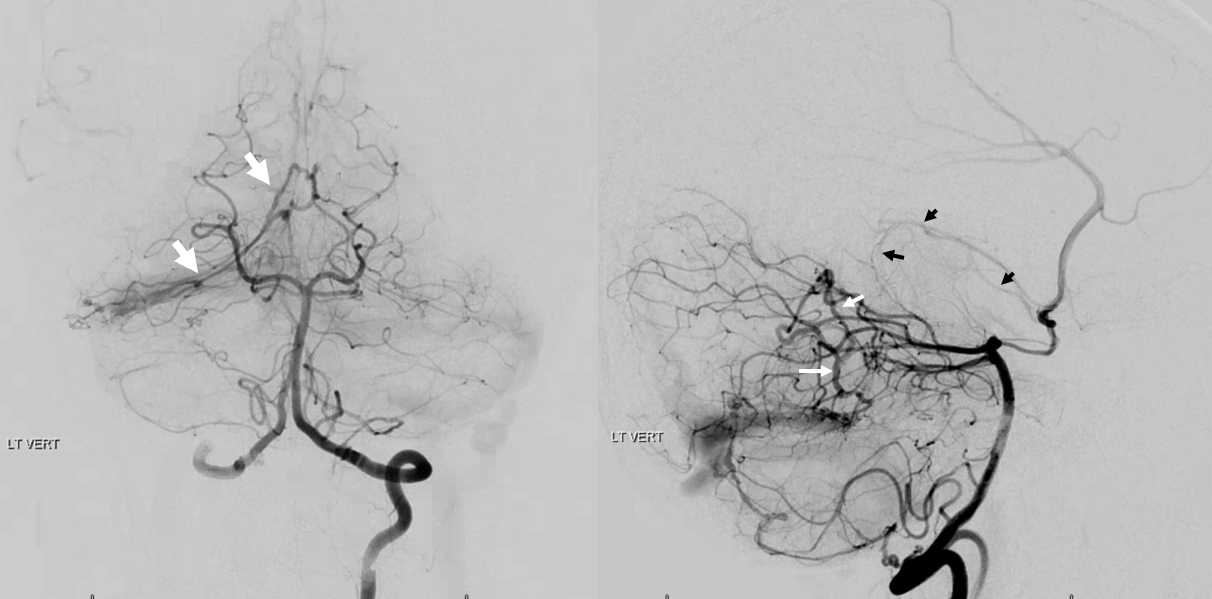
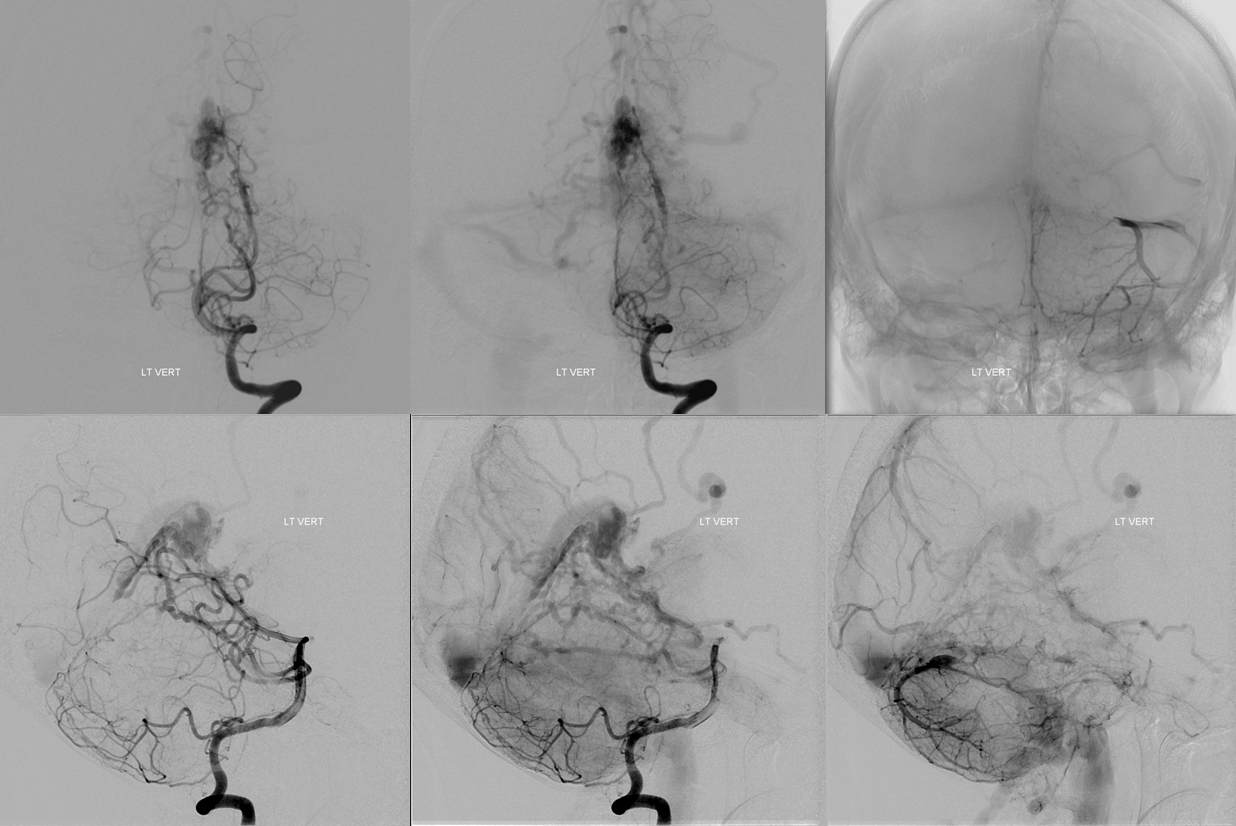
Here is a falcotentorial junction dural fistula — with tons of Davidoff-Schechter like arteries there. Below is a vert injection — all those vessels going to the shunt are Davidoff-Schechters… Full case is here
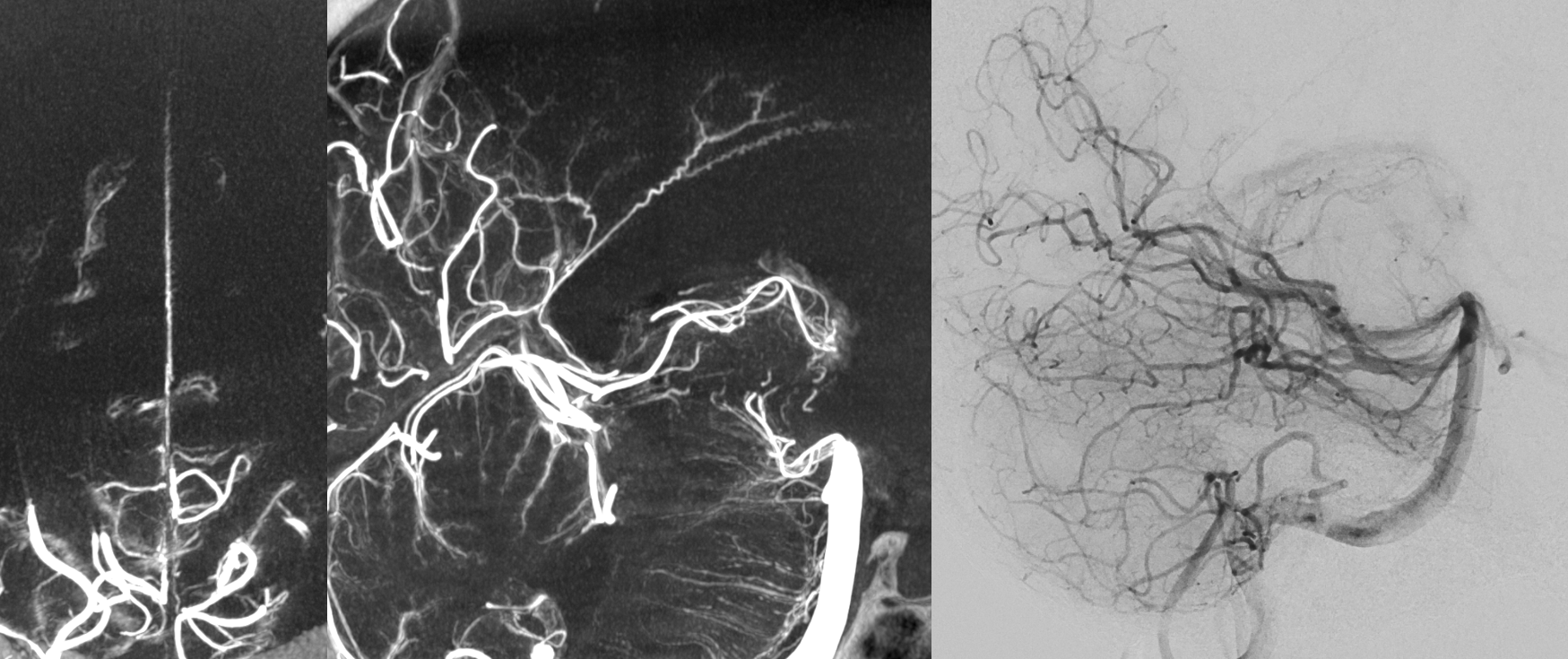
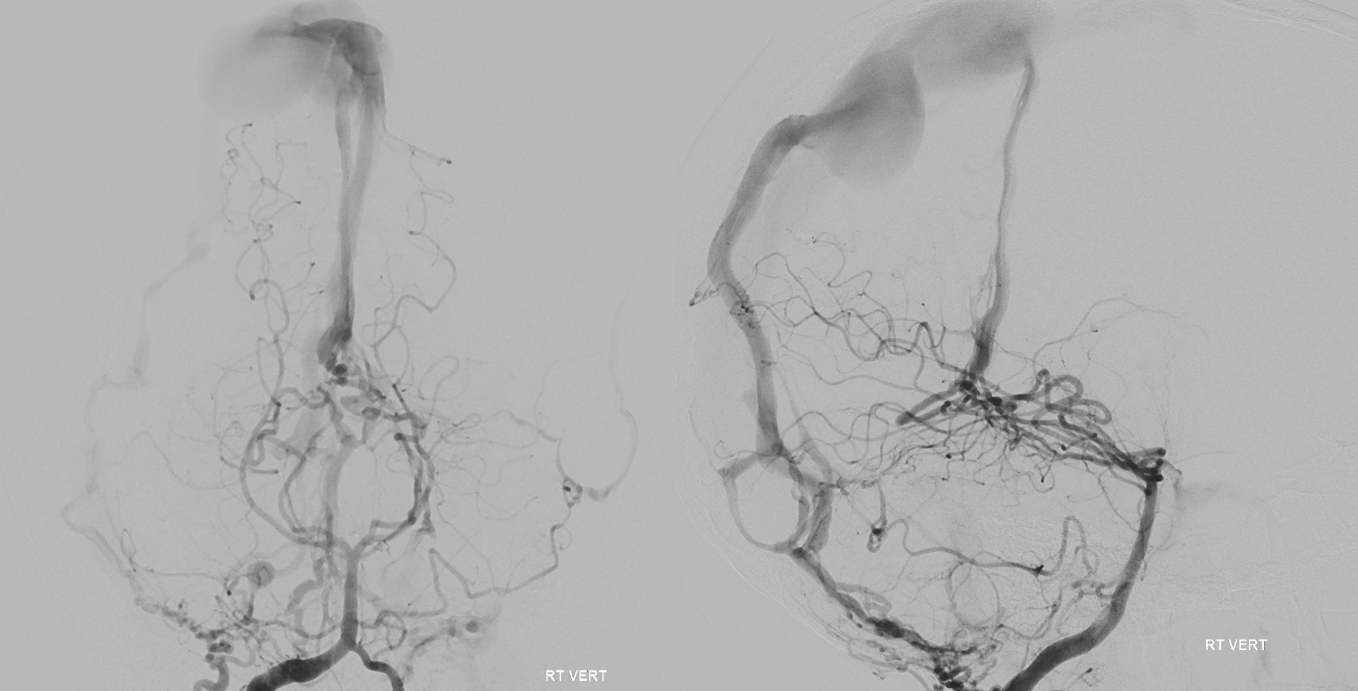
Yet another case — the fistula is in the SSS. One of many fistulas. Some are supplied by giant MMA branches. This one favors falcine arterial channels which are supplied by piodural Davidoff-Schechter branches of both PCAs. These are easily seen to be posterior medial choroidal arteries. Indeed, the “Davidoff-Schechter” refers to piodural supply. From which PCA branch do they arise? Usually from the posterior medial choroidals. Stereo views
Note another huge fistula in the more proximal SSS, supplied by enormously hypertrophied branches of the posterior meningeal arterial network.
Another posterior fossa SCA origin pial-dural anastomosis here — in a case of sigmoid sinus dural fistula treated by our “magic bullet” approach — full case here
Davidoff-Schechter in Meningioma
Direct origin off right PCA

Spectrum of Posterior Medial, Posterior Lateral, and Anterior Choroidal Arteries
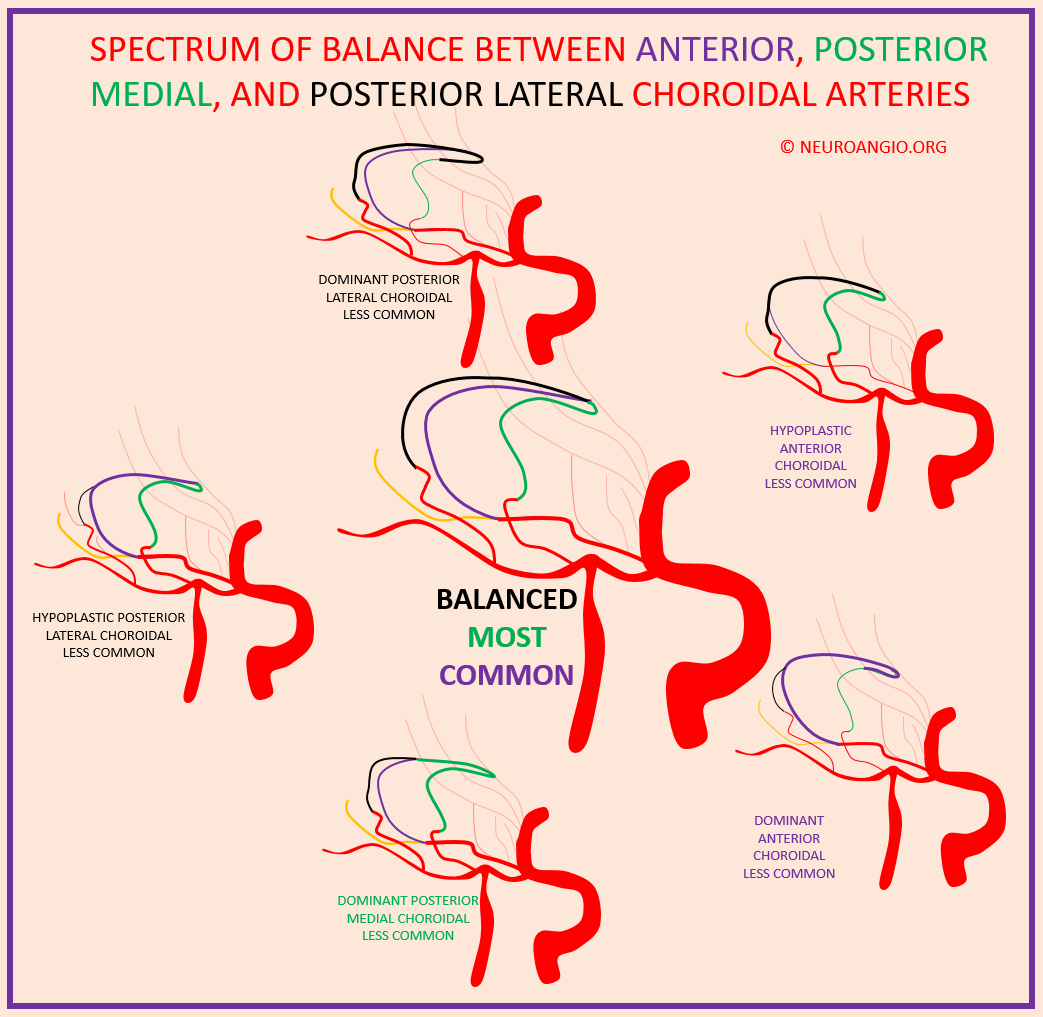
This notion, very powerful, is shown on the Anterior Choroidal Artery page
That’s it for now. Will be updating as good cases come along. Hope the level of detail here makes up for tardiness in creation of this page.