Ventral Ophthalmic Artery
This variant is extremely rare in the human — a persistent embryonic connection between the area of the anterior communicating artery and the intraorbital ophthalmic — (A) in the diagram below:
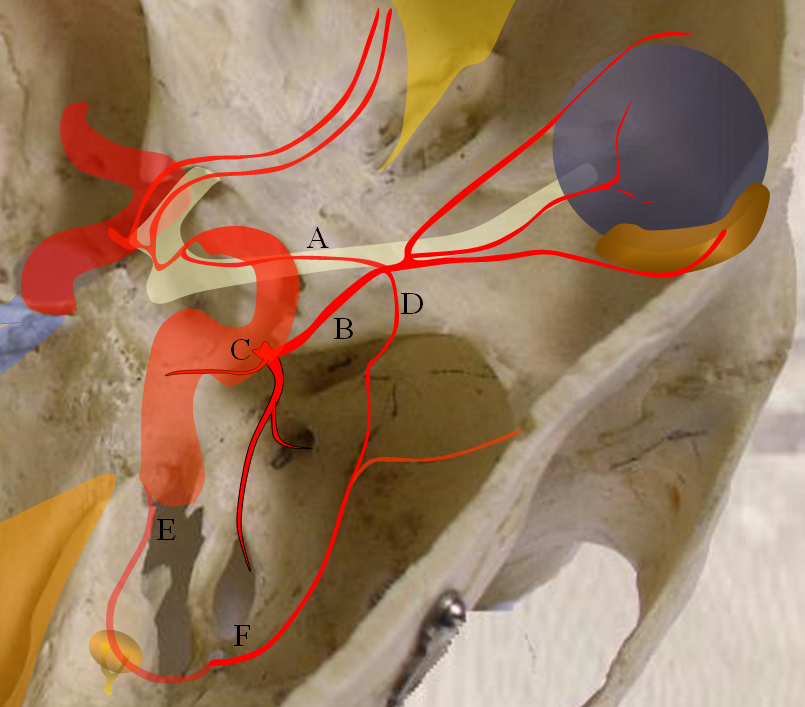
A — ventral ophthalmic artery, B — dorsal ophthalmic artery, C — Inferolateral Trunk, D — meningo-ophthalmic artery, E — stapedial artery, F — forerunner of the middle meninigeal artery originating from the stapedial system at this point
Embryology: For a full discussion of this endless topic, you can consult Lasjaunias, Berenstein, and Ter Brugge. Surgical Neuroangiography, 2nd edition, 1st volume, A limited but essential understanding of the topic is provided here by way of general important knowledge. A brief discussion with some illustrations is provided here. All of these observations were advanced first and foremost by anatomical work of D. Padget in his work The development of the cranial arteries in the human embryo. Contrib Embryol 1948; 32:207 –262 and discussed in detail by Lasjaunias P, Berenstein A, Ter Brugge KG in Clinical vascular anatomy and variations. Berlin, Germany: Springer,2001 : 414–424
The evolution of orbital supply seems quite strange in that the adult ophthalmic does not appear to come from a known precursor vessel in its expected location. In the early embryonic life, the orbit seems to be supplied by a balance of three arteries, all of which subsequently take a step back in favor of a somewhat mysteriously appearing adult ophthalmic. These three early arteries, illustrated in Figure 1 are:
1) Dorsal ophthalmic artery — originating from the adult location of the ILT and entering the orbit via the superior orbital fissure. The role of this artery gradually diminishes. In the adult, the anteromedial branch of the ILT and the recurrent tentorial branch of the ophthalmic artery (see figure below) correspond to vestiges of the embryonic dorsal ophthalmic. Very rarely, this artery persists into adult life to maintain dominant supply of the orbit, as illustrated in a case below.
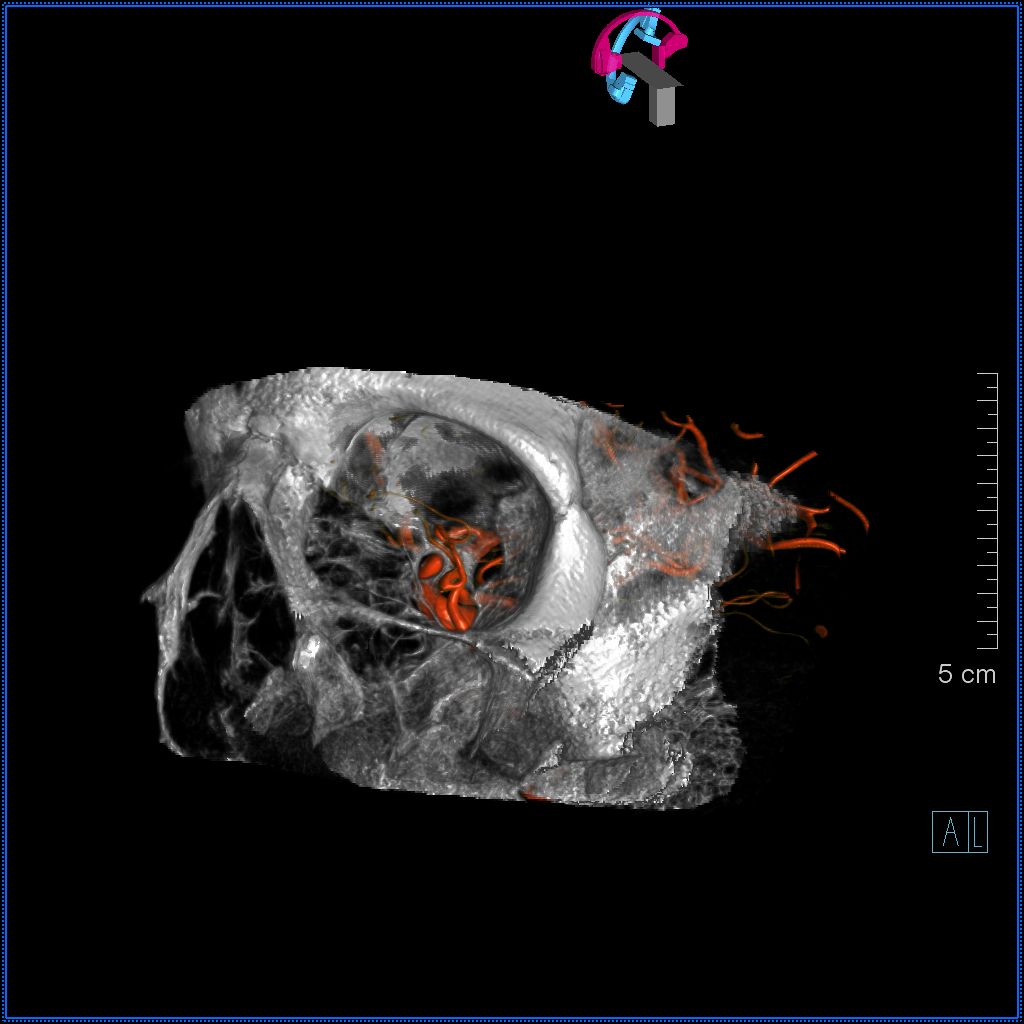
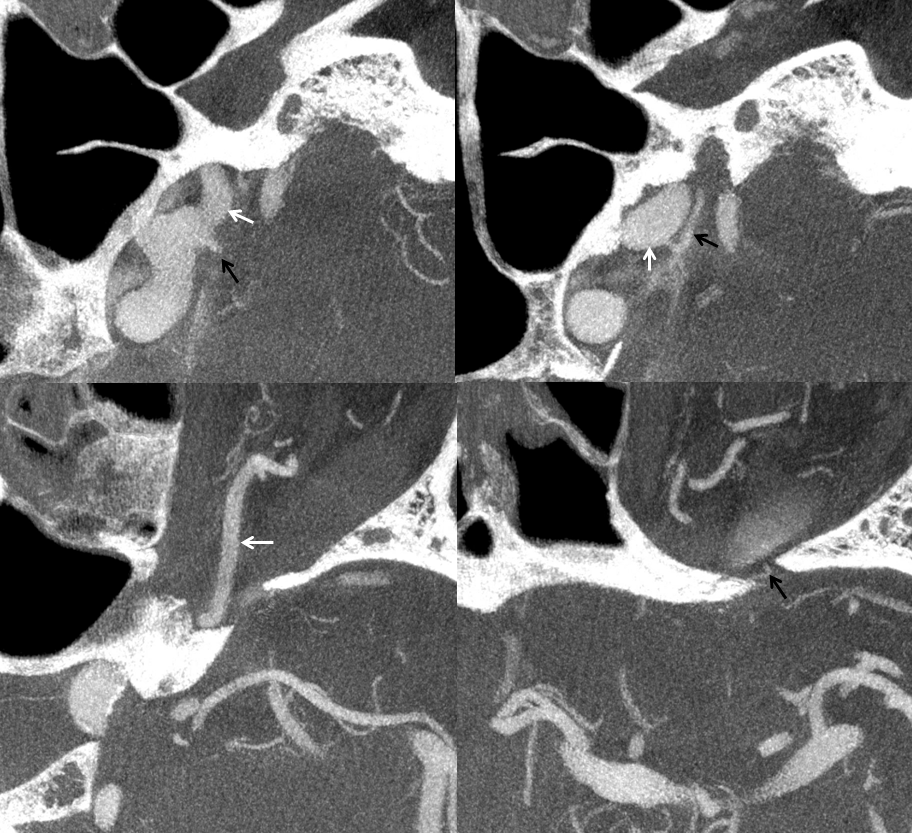
2) Ventral ophthalmic artery — this seemingly strange vessel orginates from the supracliniod carotid artery or the anterior cerebral artery (what would later become the A1 segment) and enters the orbit via the optic canal to anastomose with the dorsal ophthalmic artery. Some mammals, such as horse, maintain this disposition as the dominant solution to orbital supply. On occasion, this arrangement can persist in the human also (more common than persistence of dorsal ophthalmic). Literature reports of an unusual ophthalmic artery origin with the vessel coursing above the optic nerve in fact are describing this disposition (see example below).
3) Middle meningeal artery — techinically speaking this vessel is not yet called the MMA — as in embryonic life it originates from the stapedial artery. The MMA undergoes a complex evolution of its own with eventual transfer of MMA territory from internal carotid to external carotid systems. At the embryonic point in question, however, the “MMA” supplies the orbit through its meningo-ophthalmic branch. This, too, can on occasion persist into adult life, much more commonly than either dorsal or ventral ophthalmic persistence (see cases below). From a clinical standpoint this is the MOST IMPORTANT variant. Embolization of the middle meningeal artery from a point proximal to the takeoff of the meningo-ophthalmic artery will have the same effect as embolization of the “usual” ophthalmic artery. See MMA page for more info
All of the above arrangements can be either complete or partial. For example, a “usual” origin ophthalmic artery may supply the globe (and central retinal artery) while the MMA vascularises the lacrimal gland (in which case it is called the meningo-lacrimal variant). So, variability is the rule. From a clinical perspective, the most important vessel is the one producing the choroid blush, although any variable supply to the orbit should give one great pause prior to any kind of embolization. Intraorbital anastomoses between separate systems will exist and embolization of the central retinal artery will be a major disaster.
Getting back to embryology, it is not clear to me how the above-describes triumvirate looses out to a vessel that seemingly comes from nowhere. Some very respectable authorities (Pierce Morris, for example) believe that the adult ophthalmic artery evloves via gradual transfer of the origin of the ventral ophthalmic artery caudally along the ICA until it reaches its usual point origin at what is eventually called the ophthalmic segment. The mechanism of this is unclear and I do not think that all authorities on the subject share this view.
The above figure explains persistent ventral ophthalmic, which in a schematic looks like this:
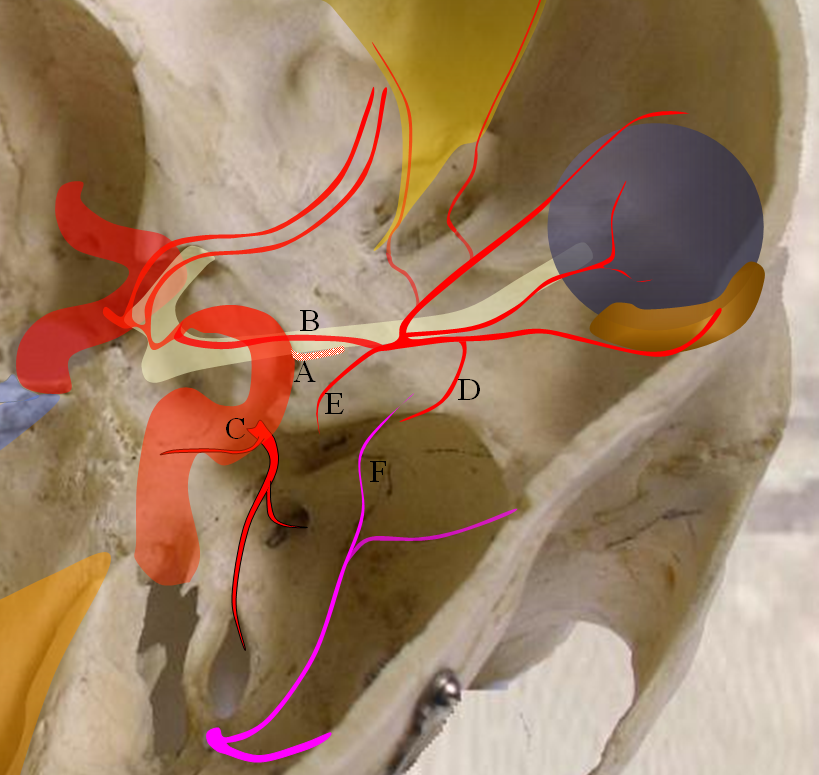
Figure 3 — persistence of ventral ophthalmic artery. A — site of “classic” ophthalmic artery origin; B — ventral ophthalmic artery; C — future ILT; D — recurrent meningeal branch of the ophthalmic artery; E — recurrent tentorial branch of the ophthalmic artery; F — MMA branches
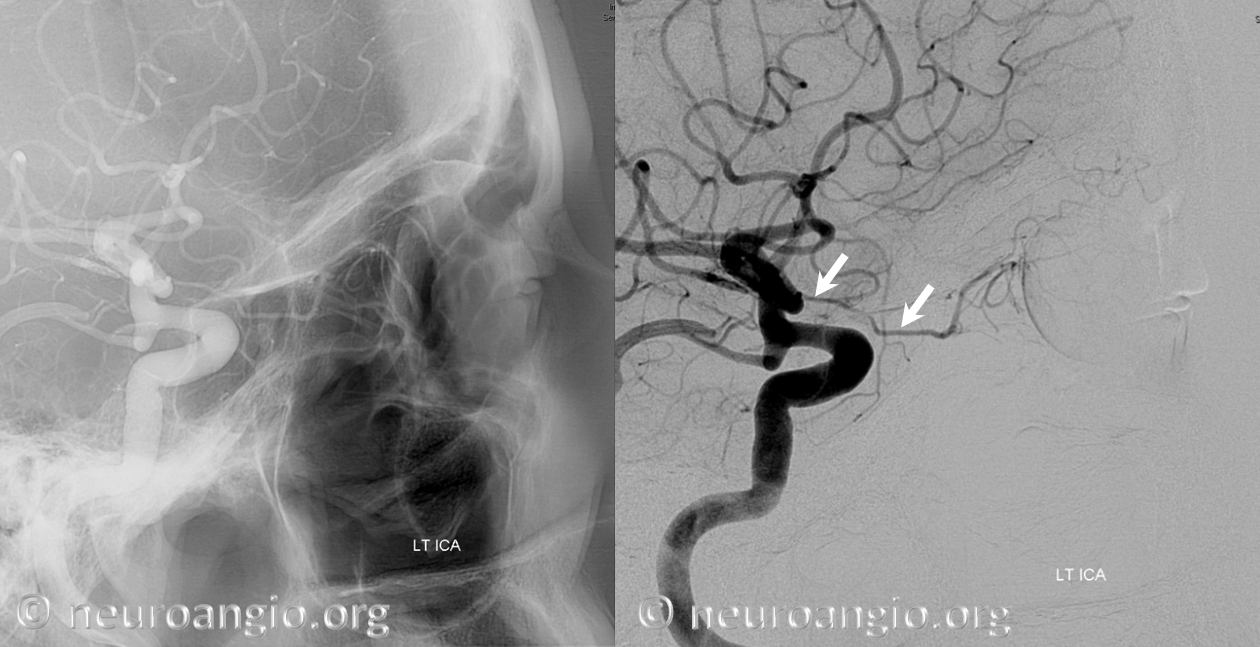
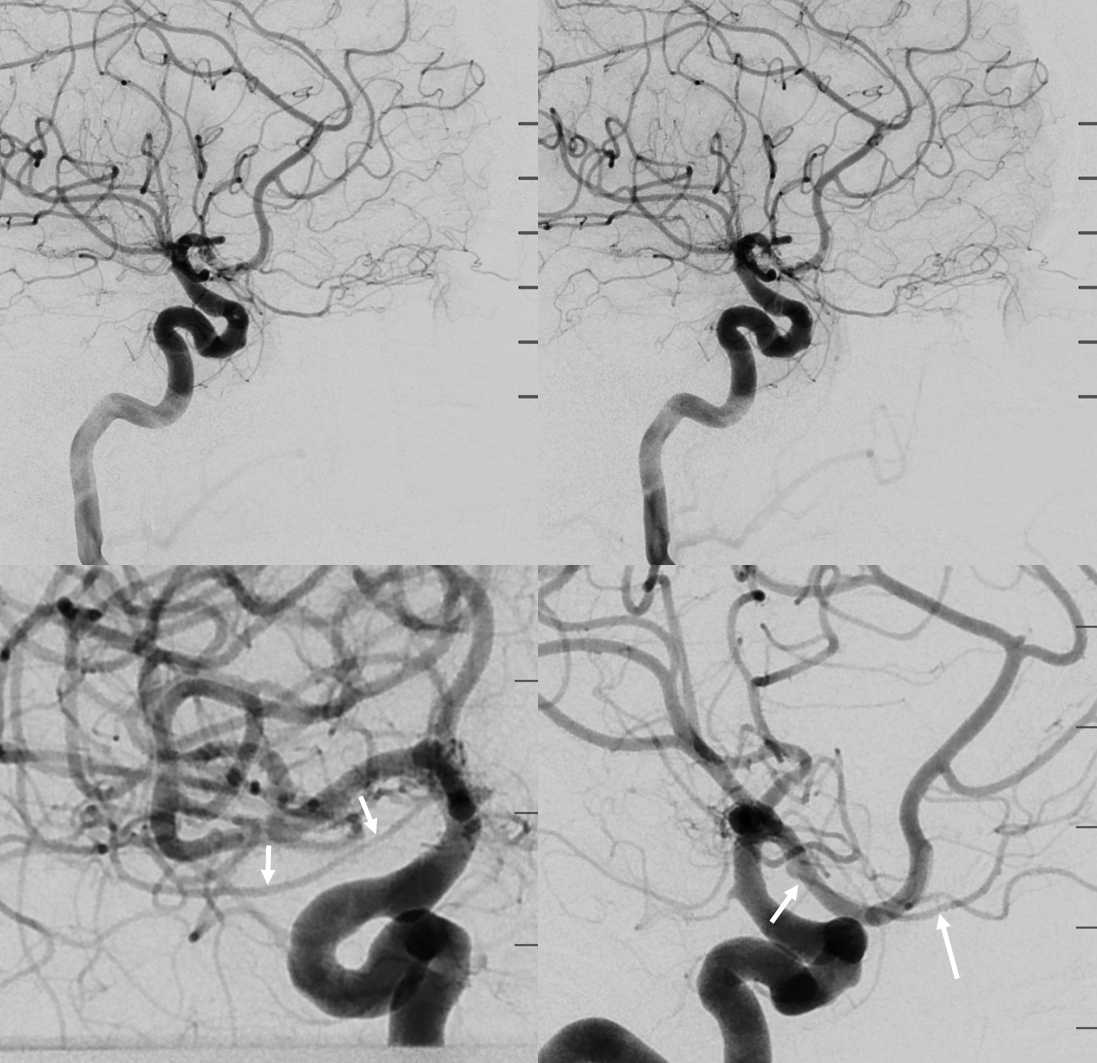
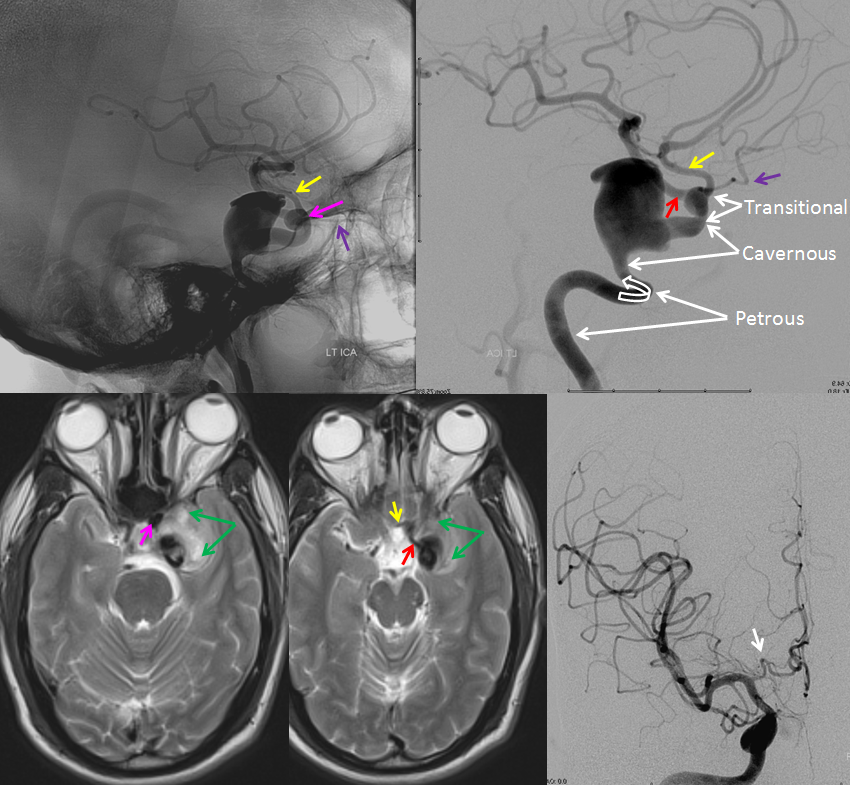
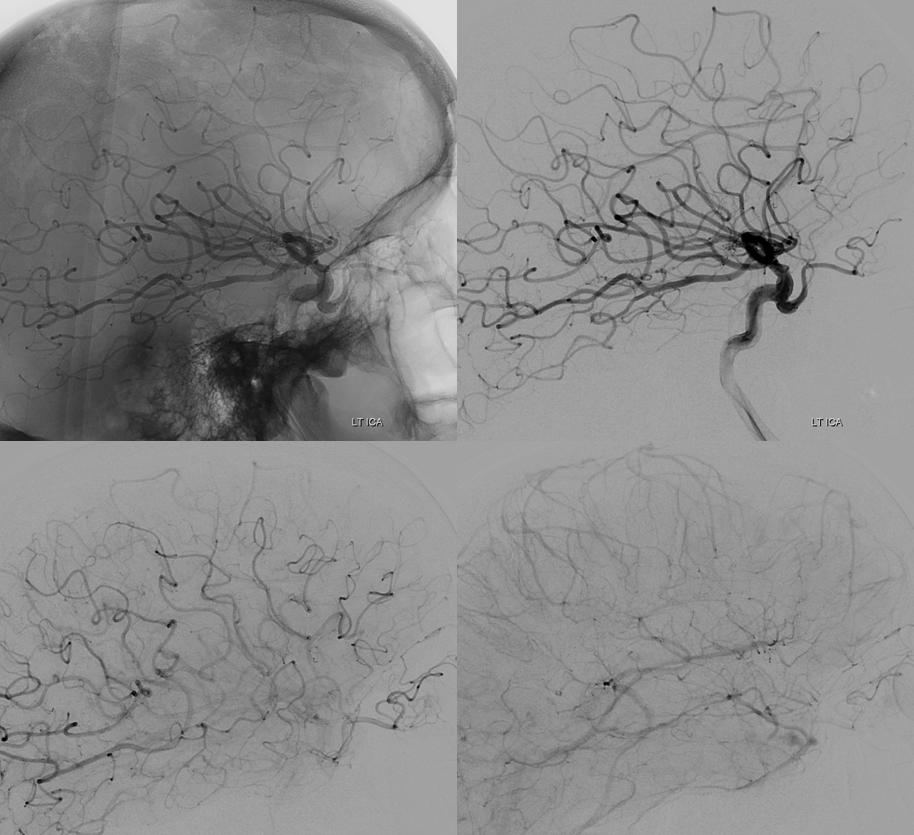
Below is an illustration of an incidentally found ventral ophthalmic artery (white arrows). Note opacification of the globe choroid (not labeled)
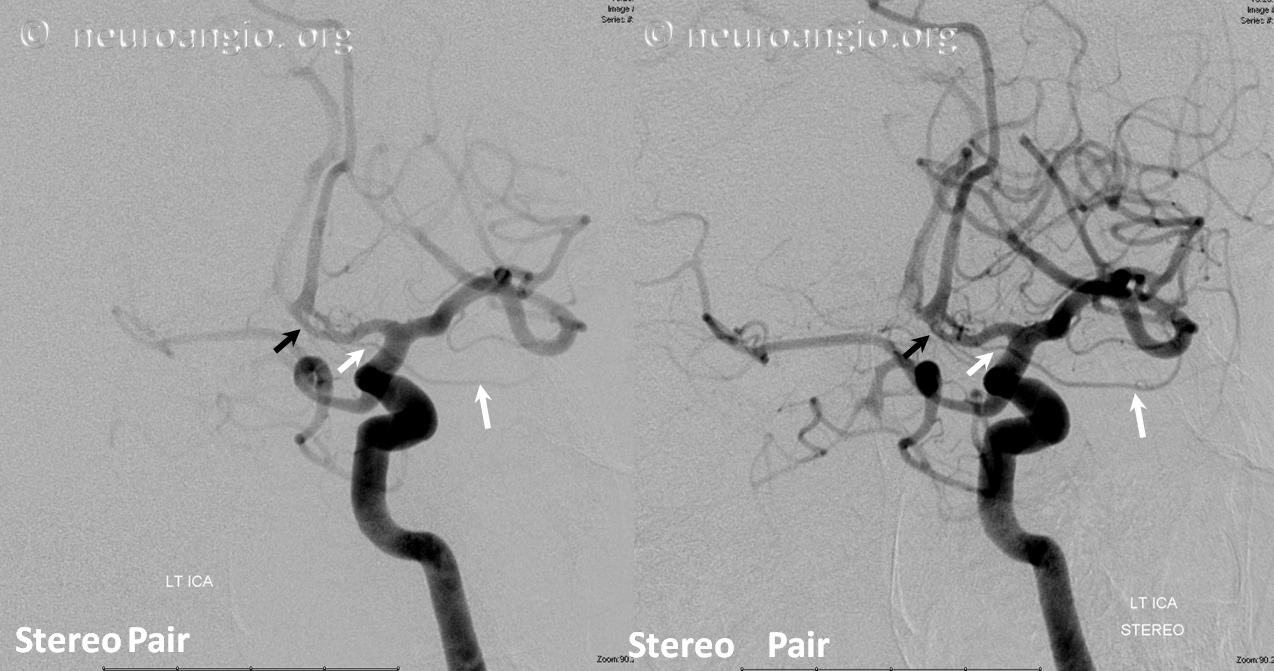
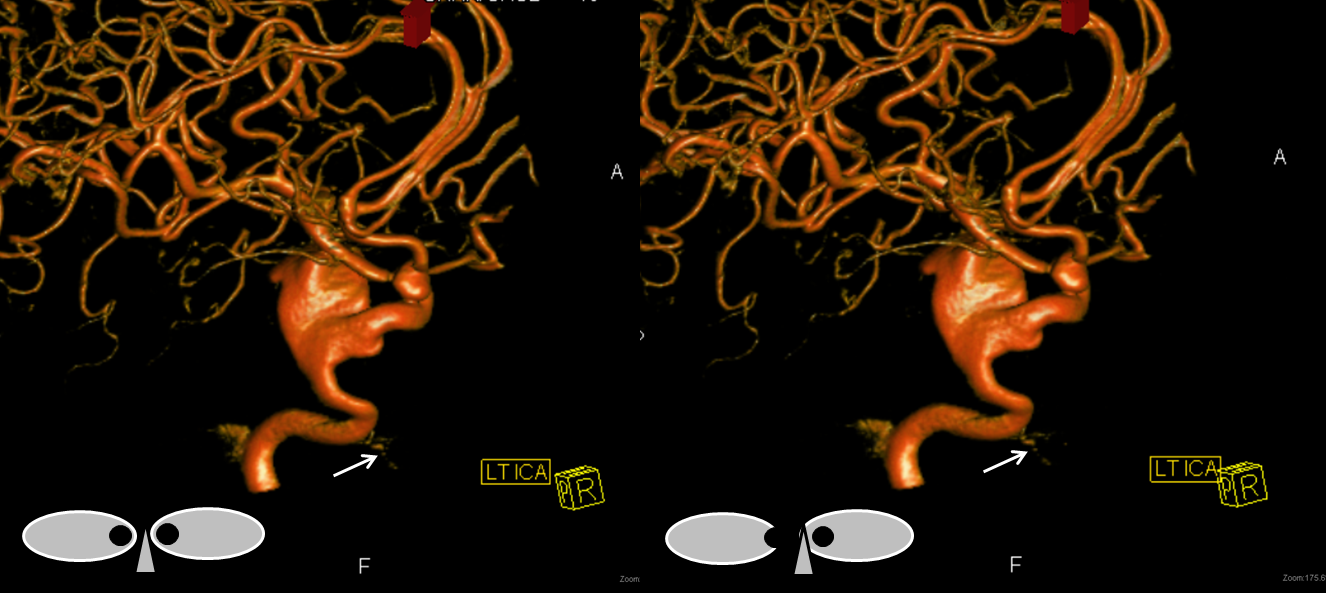
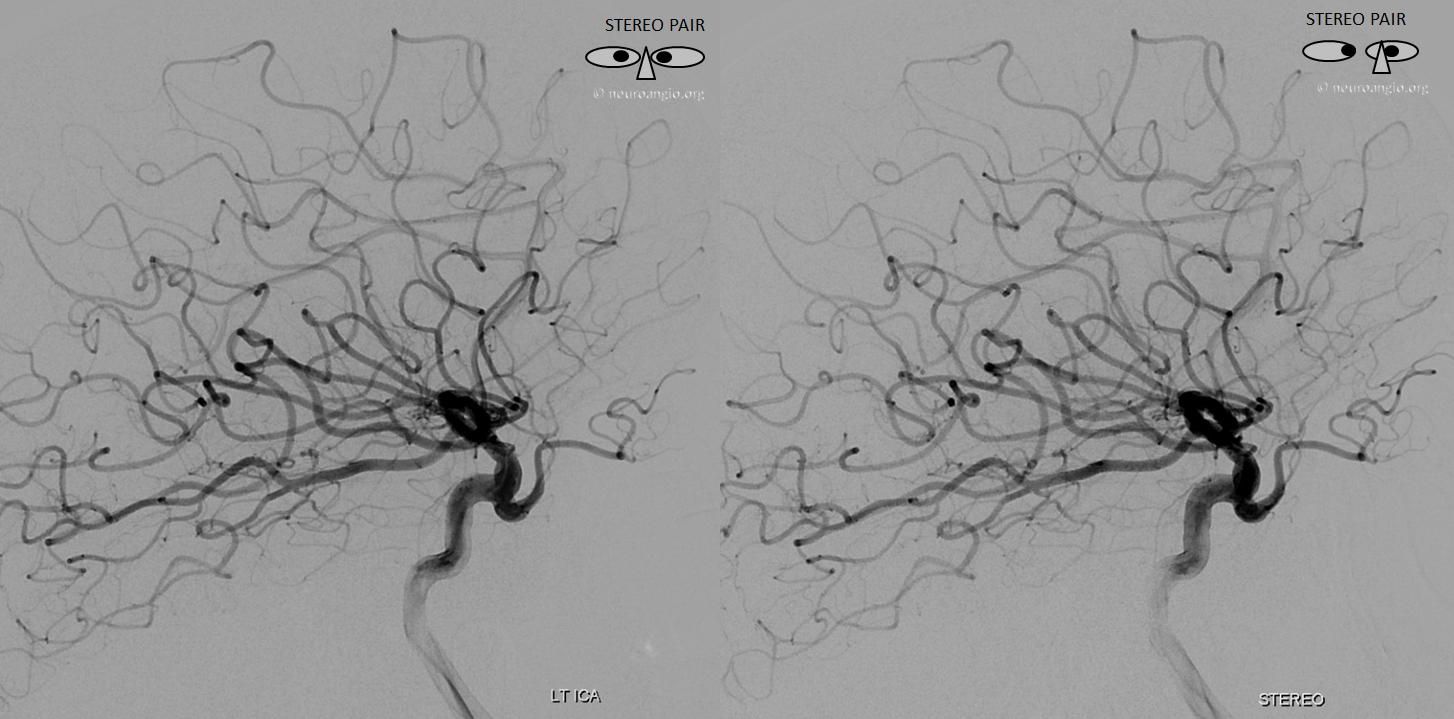
Stereo pair in frontal oblique position, demonstrating associated A1 segment fenestration (black arrows), an embryologic variant closely tied to the ventral ophthalmic artery — the infraoptic course of the A1
Beautiful stereo pairs
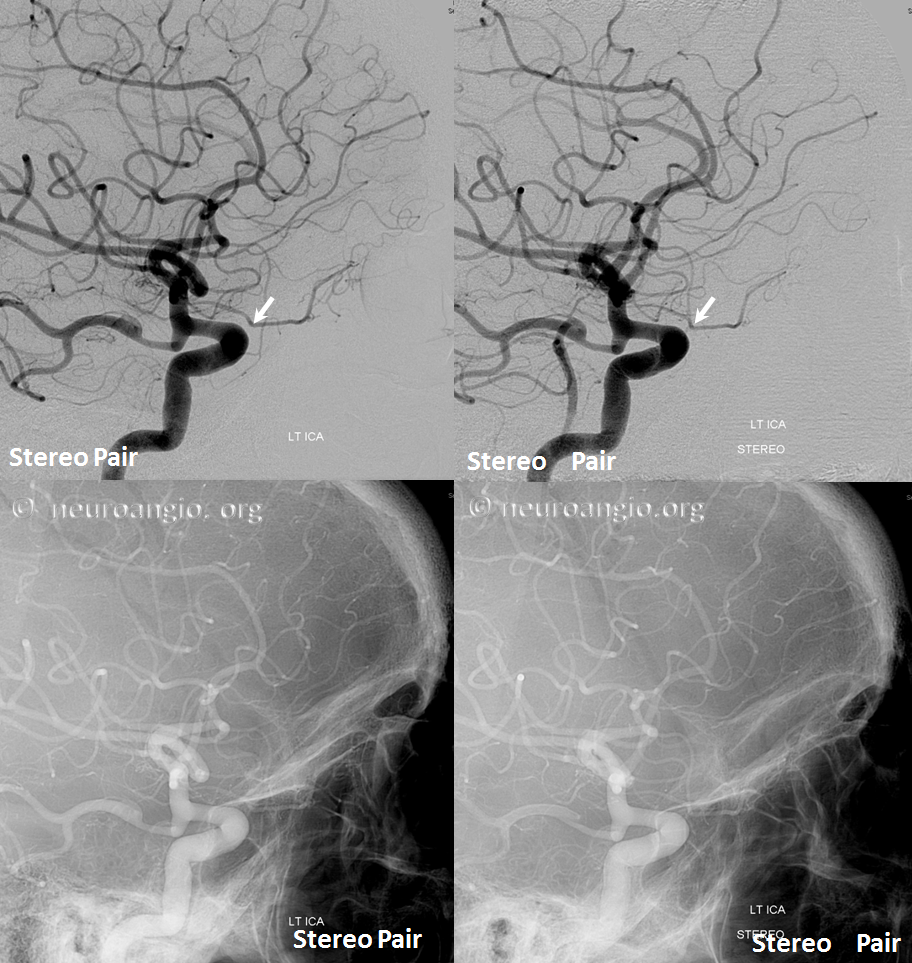
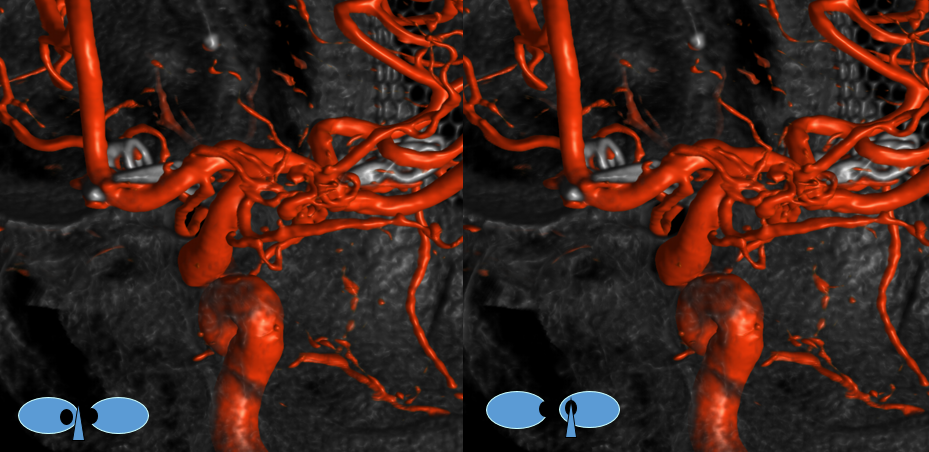
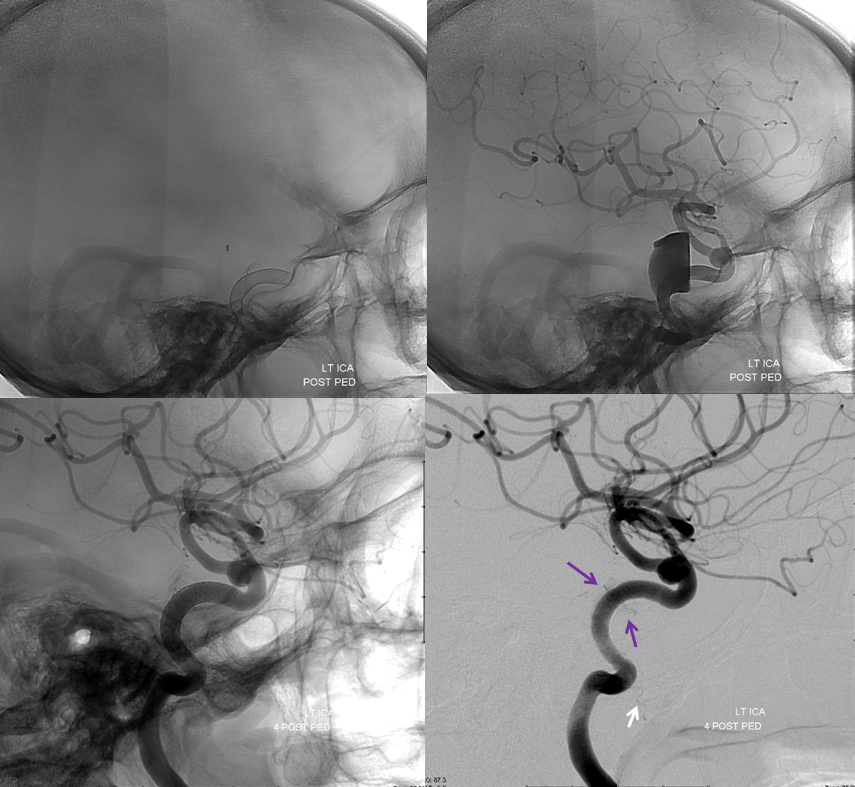
Another example, in setting of multiple aneurysms. Post clipping of ruptured ACOM and bystander MCA aneurysm, and subsequent Pipeline Shield Embolization of a small ACOM residual. Stereo images, VO origin from proximal A1. The Pipeline is placed distal to its origin, just in case.
Movie
MIPS — great anatomy of the superior hypophyseal, MHT, ILT as well
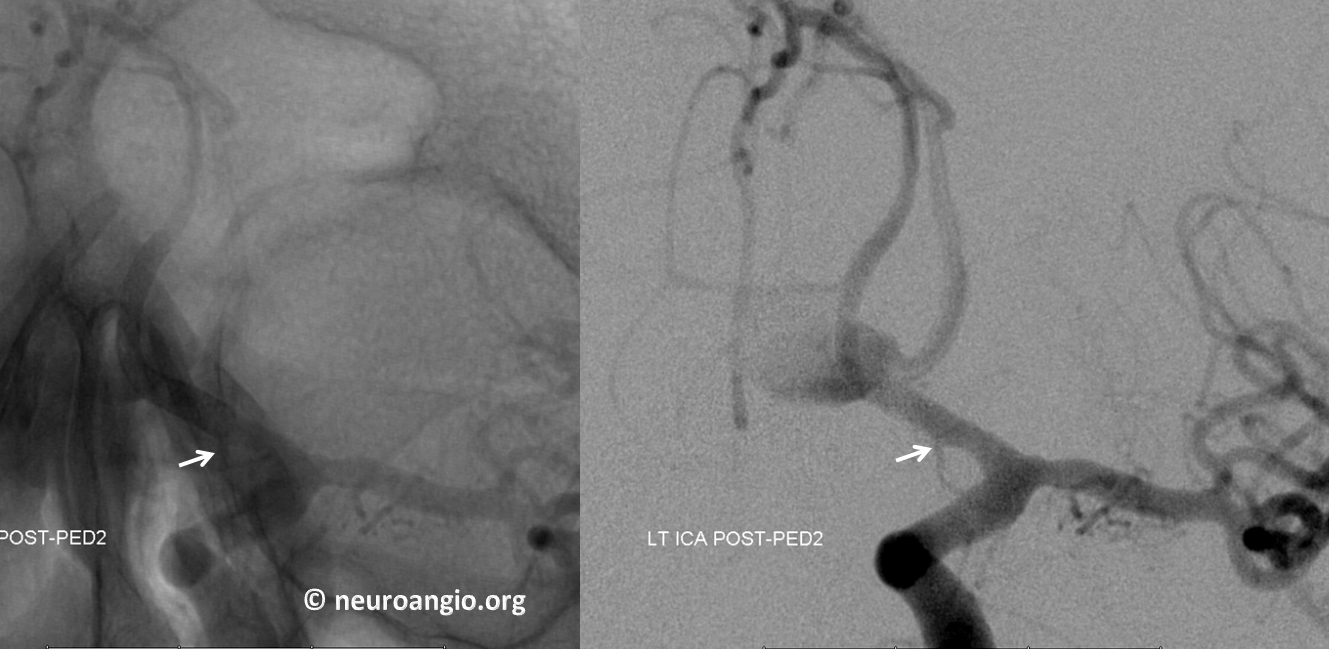
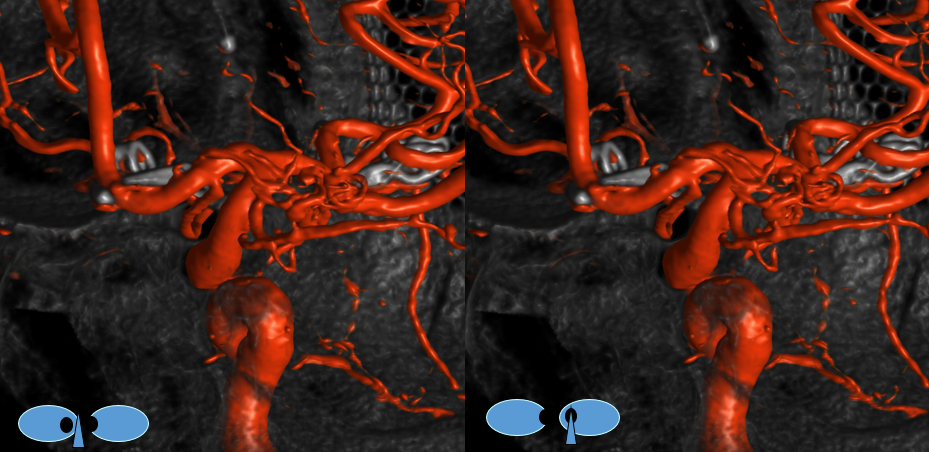
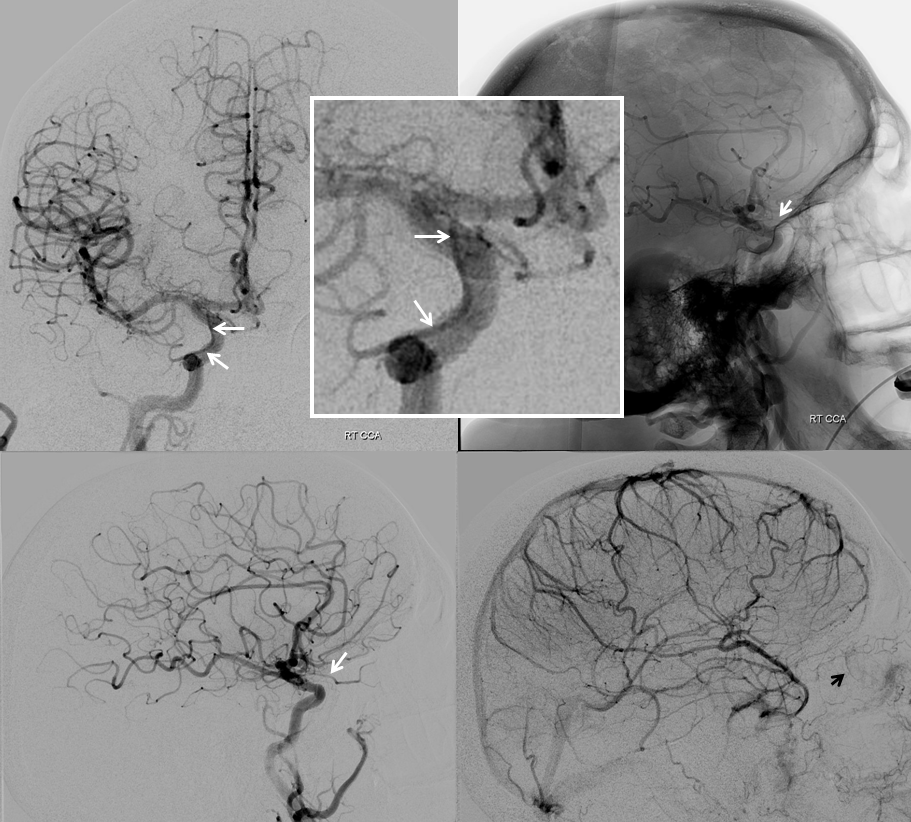
Another example of ventral ophthalmic artery (white arrows) in a patient with a giant, partially thrombosed ACOM aneurysm, courtesy Dr. Peter Kim Nelson The origin is also from proximal A1
Lateral phase images, with choroid globe blush in parenchymal phase (black arrows)
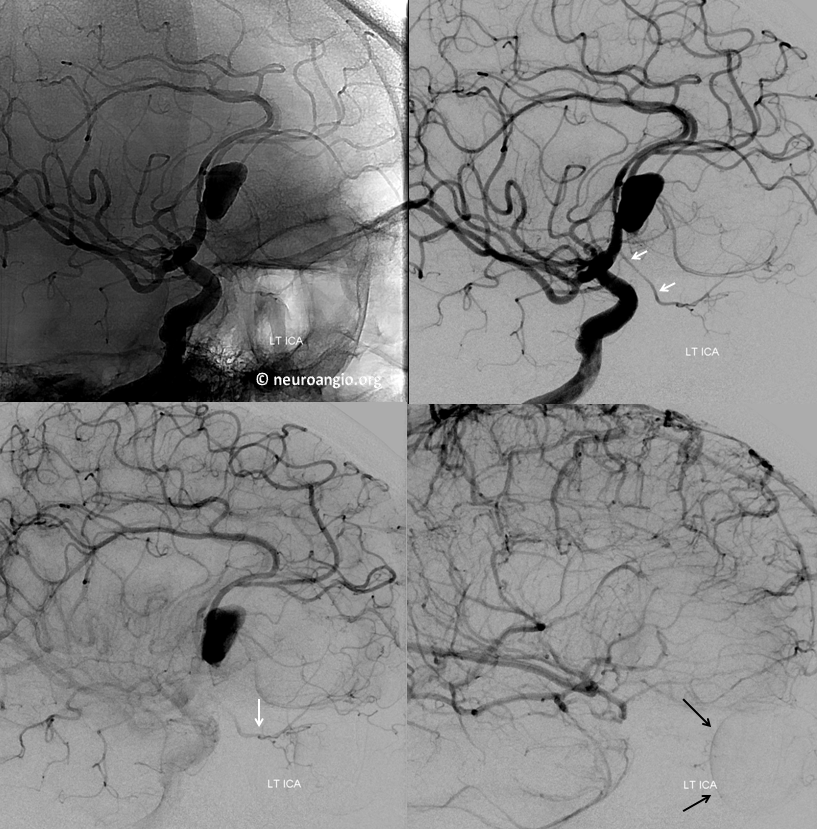
The branch remains uncovered post Pipeline aneurysm embolization
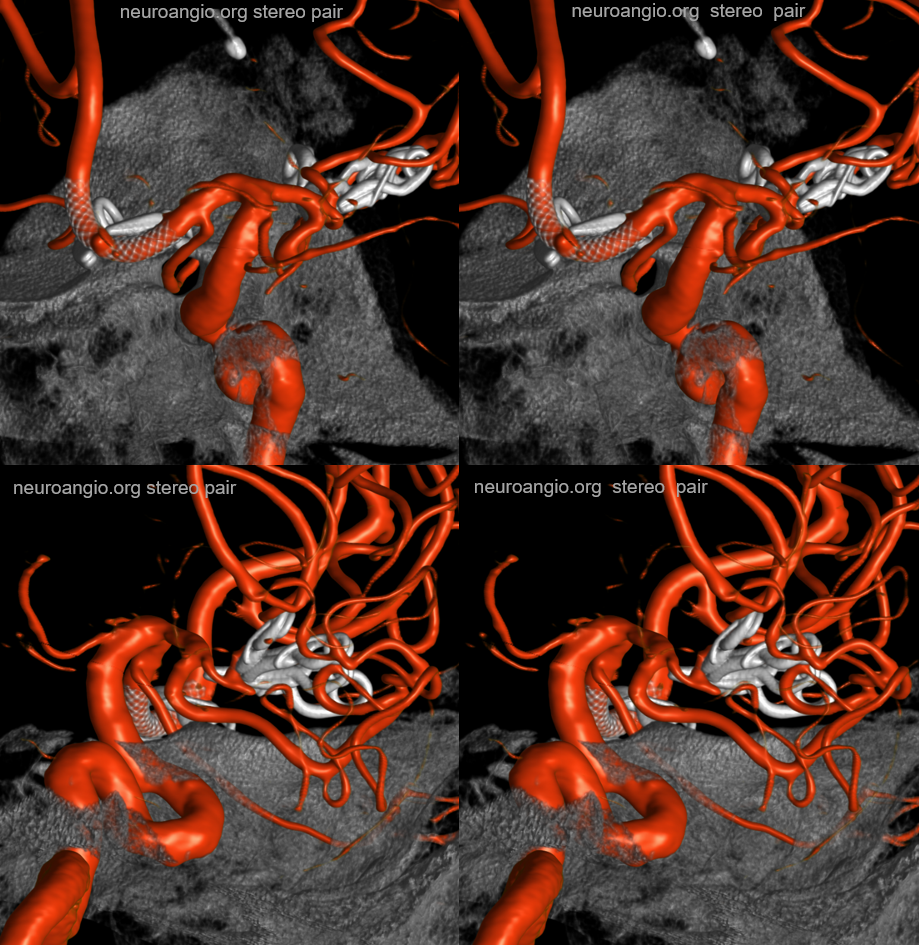
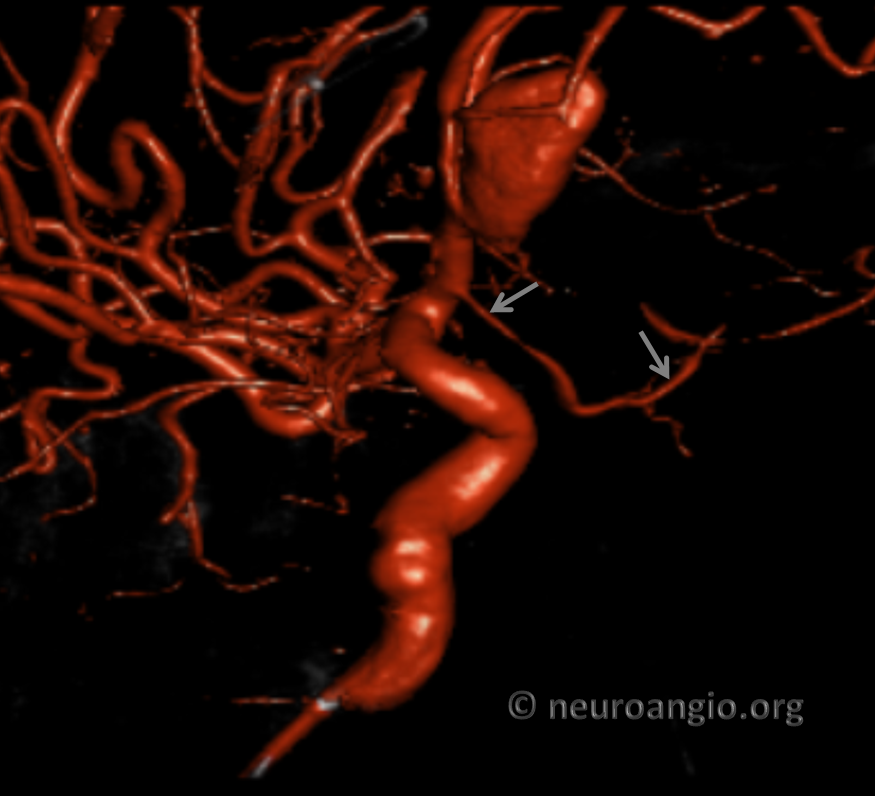
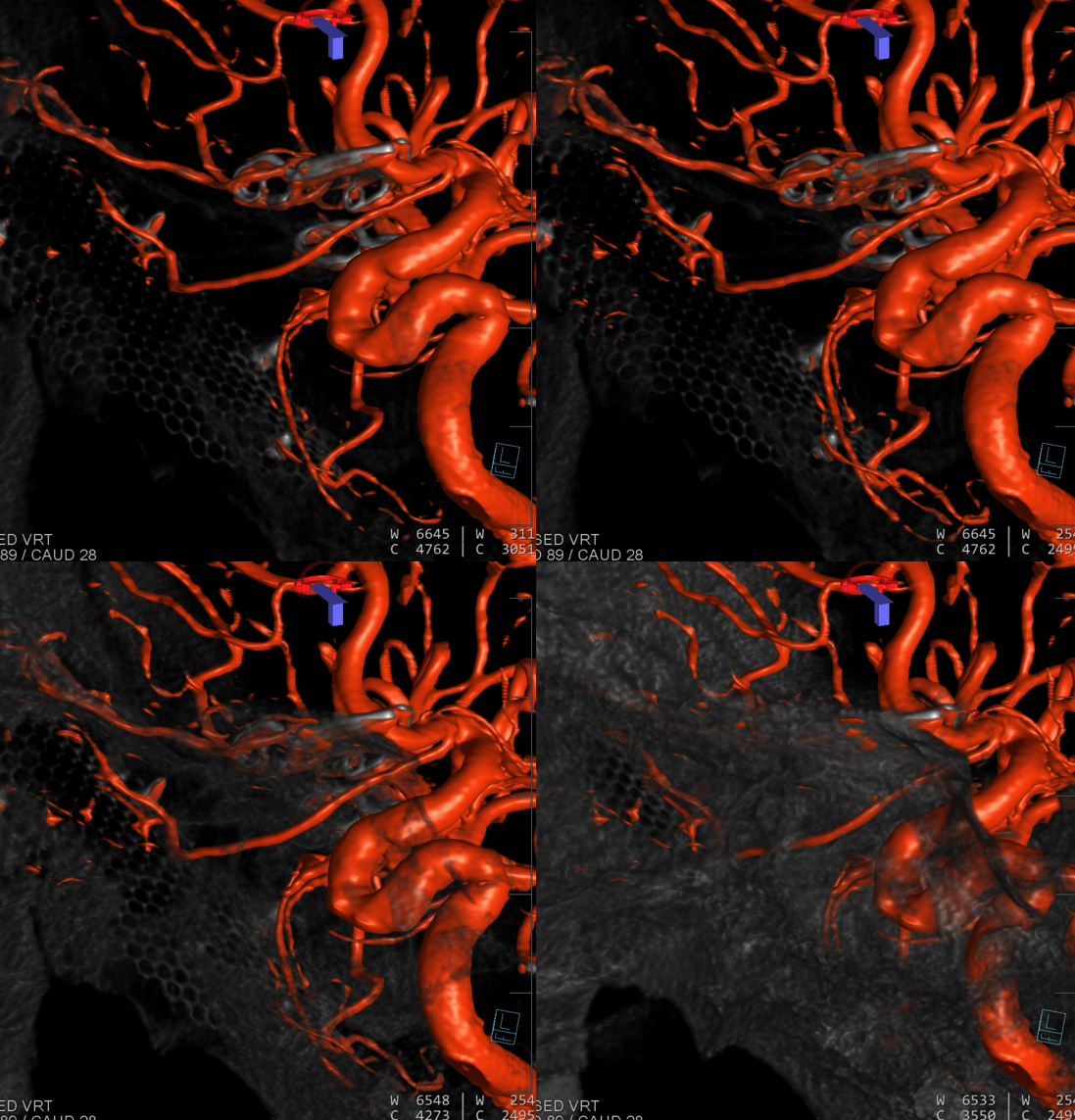
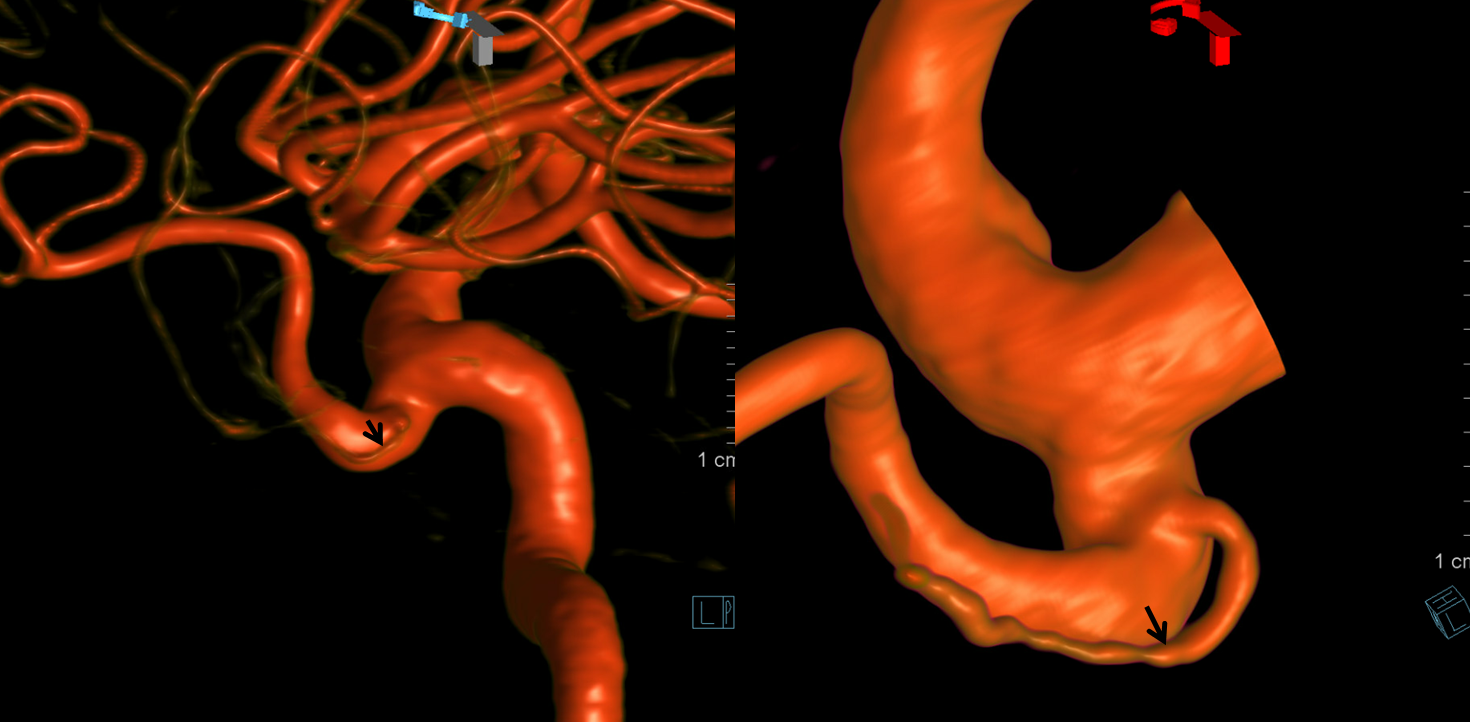
Volume Rendered
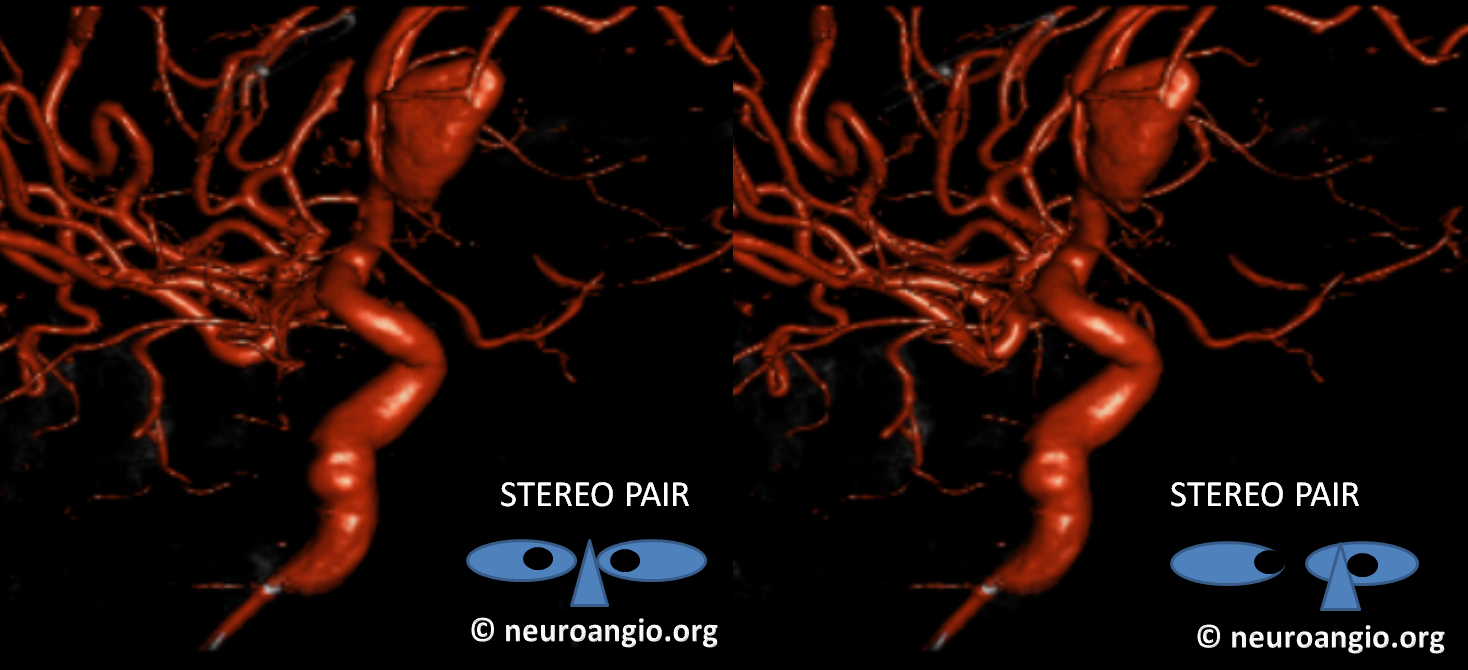
Stereo pairs
Another stereo with vessel inside orbit
Example 3
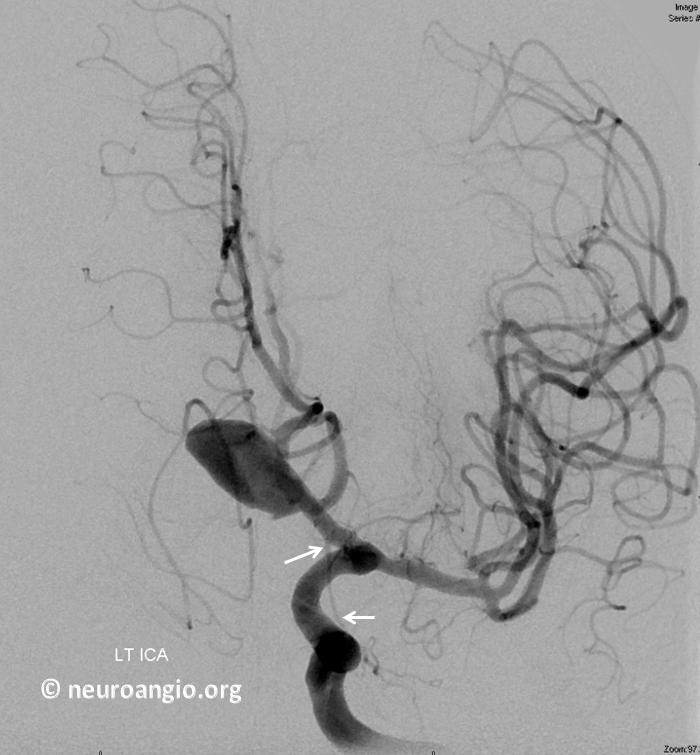
Accumulating these over years — this one is another mid-A1 origin, and also with aneurysms.
“Early Origin” ACA
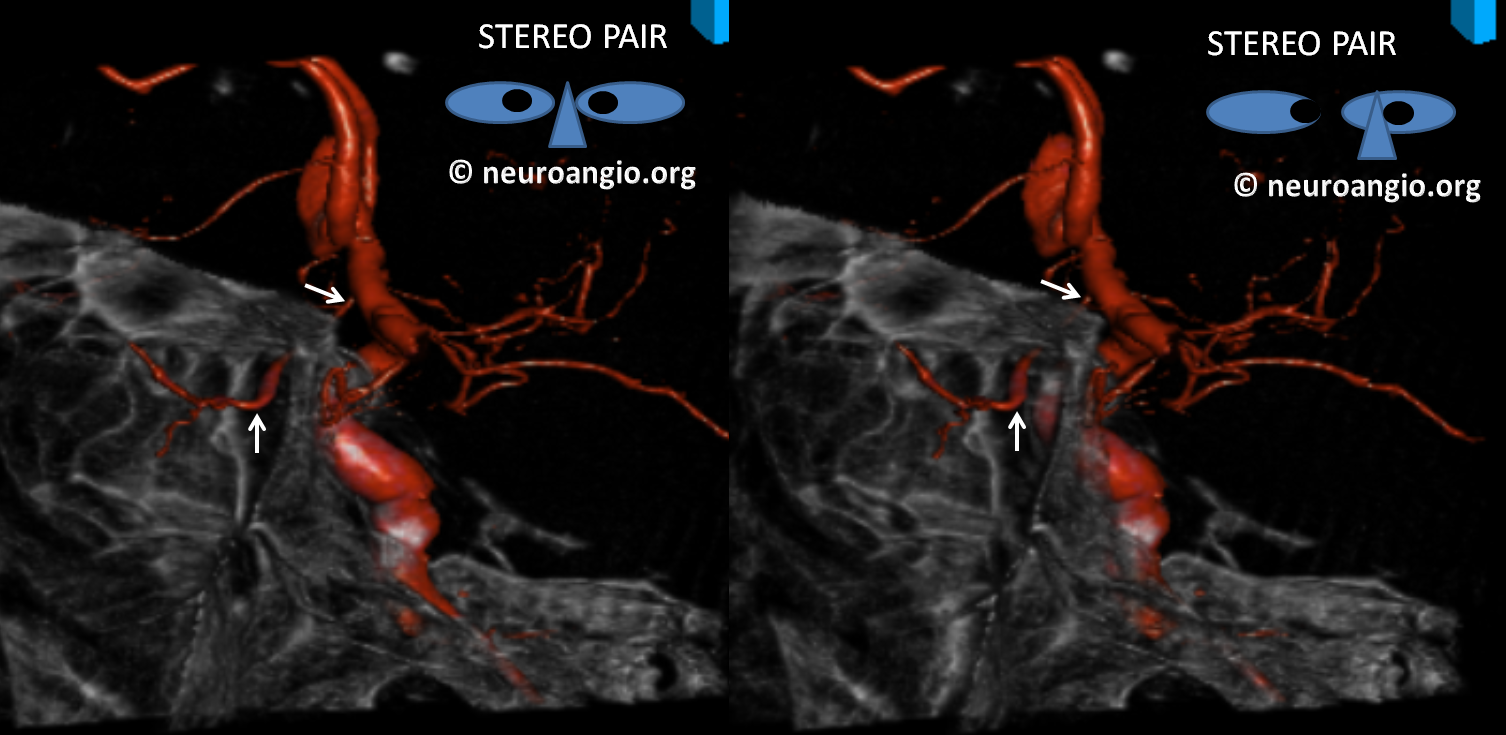
Because ventral ophthalmic represents a persistent communication between the ACOM complex and the ophthalmic artery / ophthalmic segment of the ICA, it also explains the so-called “infraoptic course” of the anterior cerebral artery A1 segment. The A1 which runs underneath, rather than over the optic nerve is nothing more than a segment of persistent ventral ophthalmic artery, in this case associated with “normal” origin of the ophthalmic artery itself. Here is an example of this variant, in a patient with a giant cavernous aneurysm — another testament to aberrant development. The infraoptic A1 / ventral ophthalmic is labeled wit the yellow arrow. The ophthalmic artery arises from the same same segment as the “A1” — as should be the case if both represent a vestige of the ventral ophthalmic artery. Notice infraoptic course on T2 MRI, and developmental ectasia of the contralateral right A1 (white arrow)
Stereoscopic view of 3D-DSA, from medial to lateral, demonstrating preferential lateral aneurysm expansion and ophthalmic segment anatomy, and the infraoptic ventral ophthalmic / A1 segment. There is also a well-seen mandibulovidian artery (white arrow)
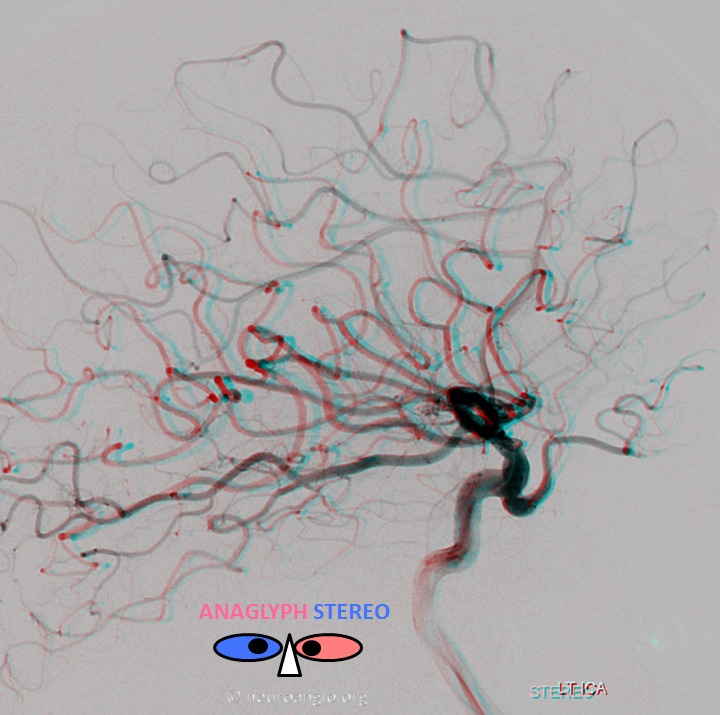
Anaglyph stereo
Lateral projection views immediately post Pipeline embolization (top), and 4 months later (bottom). Notice branches of the MHT (purple arrows) which remain open, and are now visible after the aneurysm has disappeared. The mandibulovidian artery is again marked with white arrow.
Here is a great case of ventral ophthalmic artery on the right and dorsal ophthalmic artery on the left in the same patient. How about that for embryology. Case courtesy Dr. Eytan Raz. White arrows point to ventral ophthalmic. Black to choroid blush
Left side shows a very large dorsal ophthalmic artery — embryologically this is the same as the anteromedial branch of the ILT.
Cross-eye stereos
VRs, showing a another branch of the ILT/dorsal ophthalmic (black arrows)
Dyna CT axial views trace the dorsal ophthalmic (white arrows) through the superior orbital fissure. The smaller branch (black) also traverses the superior orbital fissure to enter the orbit more laterally — perhaps a lacrimal branch
Anaglyph VR stereo
See Ophthalmic Artery Page for more info
