The Middle Cerebral Artery is, phylogenetically, the youngest of all cerebral vessels (see dedicated Neurovascular Evolution and Vascular Neuroembryology for more detail). This situation reflects relatively recent development of the neocortex in “higher species” Briefly, the MCA arises from a phylogenetically far older anterior cerebral artery (older because it vascularizes, among other things, the more primitive olfactory lobe). A collection of lenticulostriate-like vessels, such as the future artery of Heubner, serve the basal ganglia homologs of fishes / reptiles. As the 6-layer neocortex emerges and enlarges, the lenticulostriate system is recruited to supply it. Thus, MCA is a hypertrophied lenticulostriate vessel that captures cortical territory of the cerebrum. Simple as that. It is both a branch of the anterior cerebral artery and a branch of the lenticulostriate system — not vice versa.
This perhaps initially strange theory is powerful and useful in that it allows us to conceptualize the many variations of MCA and to understand the lenticulostriate system as a whole. For example, a duplicated / accessory MCA is nothing more than a disposition of two “perforator” vessels remaining in balance instead of selection of a dominant single trunk, similar to duplicated superior cerebellar or AICAs arising from pontine perforators that capture cerebellar territory. A Heubner is not some unique vessel just because it is an eponym – it is simply a medial-most lenticulostrite vessel, in balance with the rest of the lenticulostriate system.
In terms of cortical branching patterns, there is no law or order. Anything goes. The M1 can be nonexistent (accessory MCA), short, long, or infinite –with no true bifurcation. The classic story of superior division always supplying frontal lobe, inferior division going to temporal lobe, with either division achieving dominance by annexing parietal lobe is a nice fairy tale. Minimal exposure to MCA thrombectomy will teach one to assume nothing and be ready for anything that might lie beyond the M1 stump. Trifurcations, pentafurcations, no bifurcation — real life laughs at manuscripts devoted to measurements of average lengths of this or that segment or percent estimates of this or that branching pattern. The only important thing is what territory is being supplied, not where the branch comes from.
Lenticulostriate system
Traditionally divided into medial and lateral groups, with further subdivision of the lateral group into lateral and medial subgroups. As you might guess, neuroangio does not hold this artificial subdivision in high opinion. We encourage you to think of the entire lenticulostriate family as a whole. There is a gaussian balance here, like in so many other vascular arrangements. There can be one, two, three, or more separate lenticulostraite trunks. A dominant “medial” lenticulostriate group means correspondingly smaller lateral family, and vice versa. The Heubner is simply the most medial perforator, usually from ACOM area. It is in balance with the rest. A big A1 perforator usually means no ACOM origin Heubner — unless you want to call the mid-A1 perforator by that eponym. A big Heubner can take over some “lateral” lenticulostriate territory, and cortical territory also as an “accessory MCA”. Which is why studies aimed at defining the Heubner territory are so unsatisfying. What matters is the individual patient disposition.
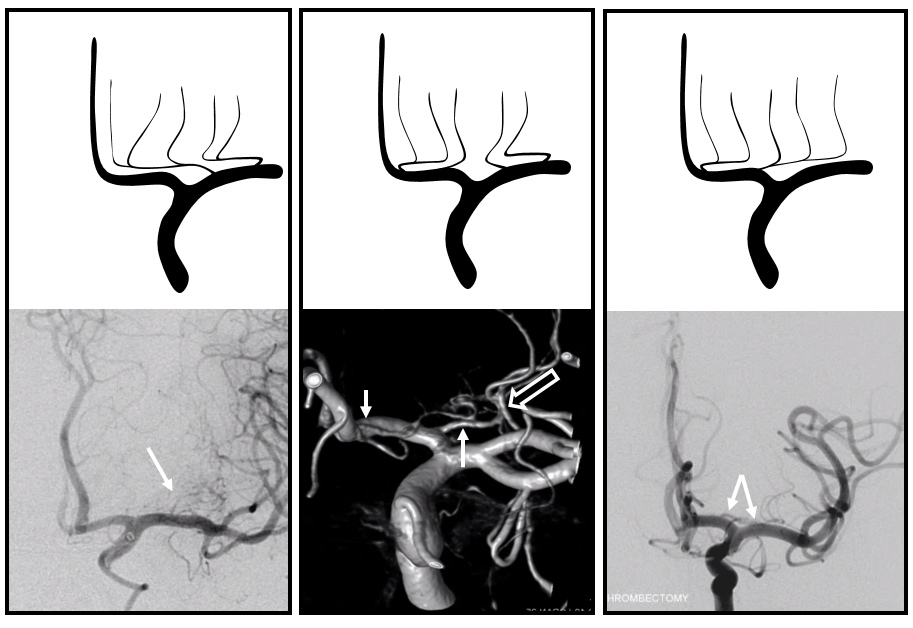
Look at the images below — on the left there is a dominant “lateral” system taking over the medial family. In the center there are two ACA lenticulostriates — a Heubner and a proximal A1 (white arrows) and a lateral one arising from the superior division, as it often does when the M1 segment is short. On the right there is an unusually highly dominant A1 segment family that is taking over the lateral lenticulostriate territory.

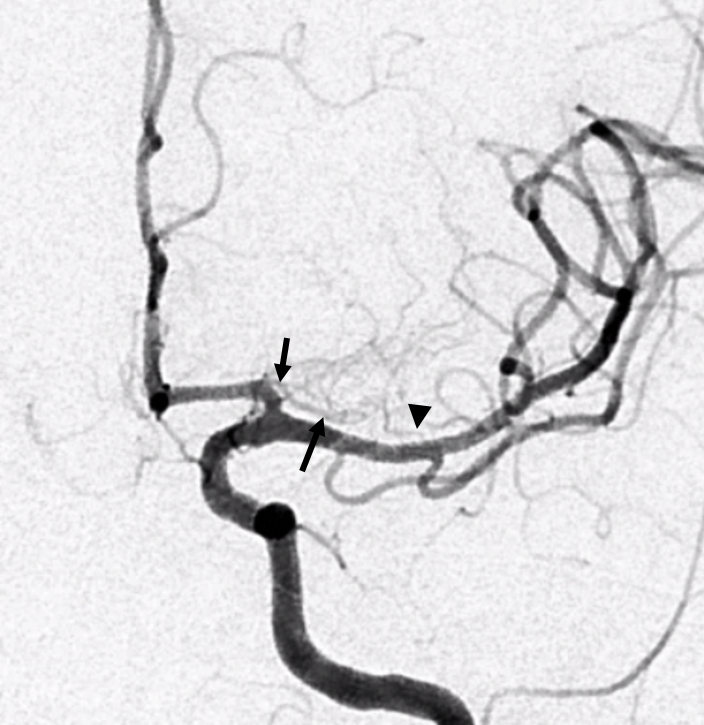
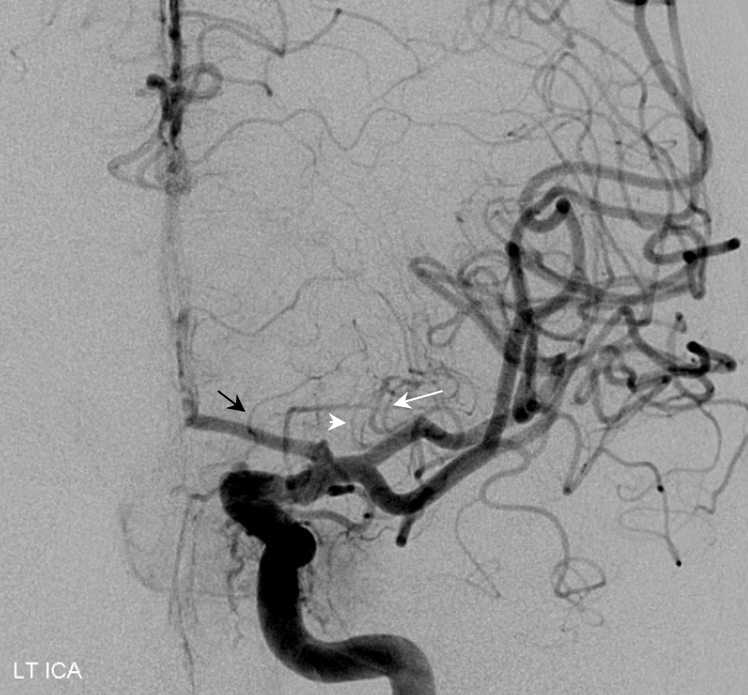
Below is another example of large lenticulostriate trunk arising from proximal A1 segment (black arrows), supplying most of the lateral lenticulostriate territory. A smaller one comes off the M1 (arrowhead)
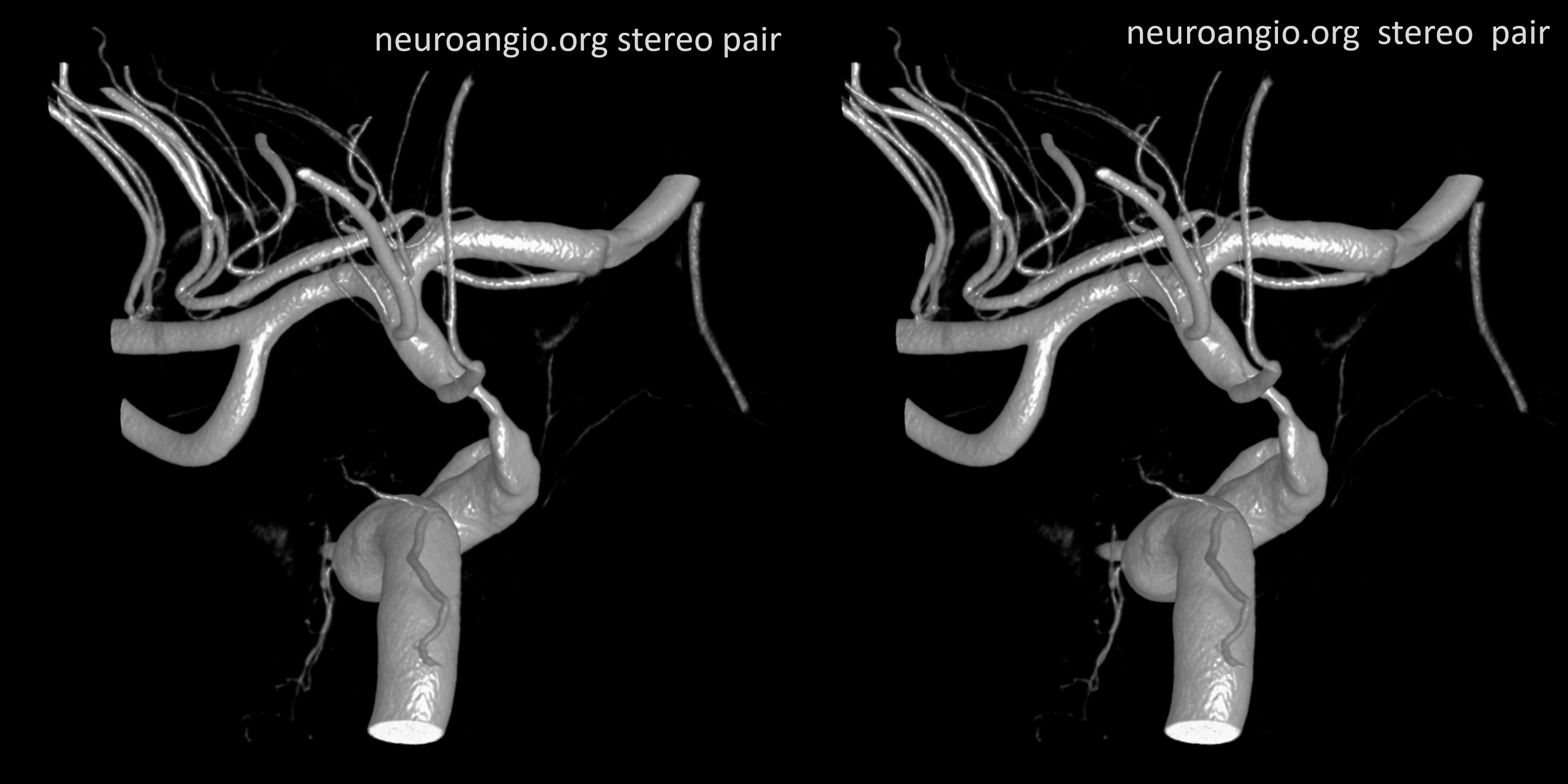
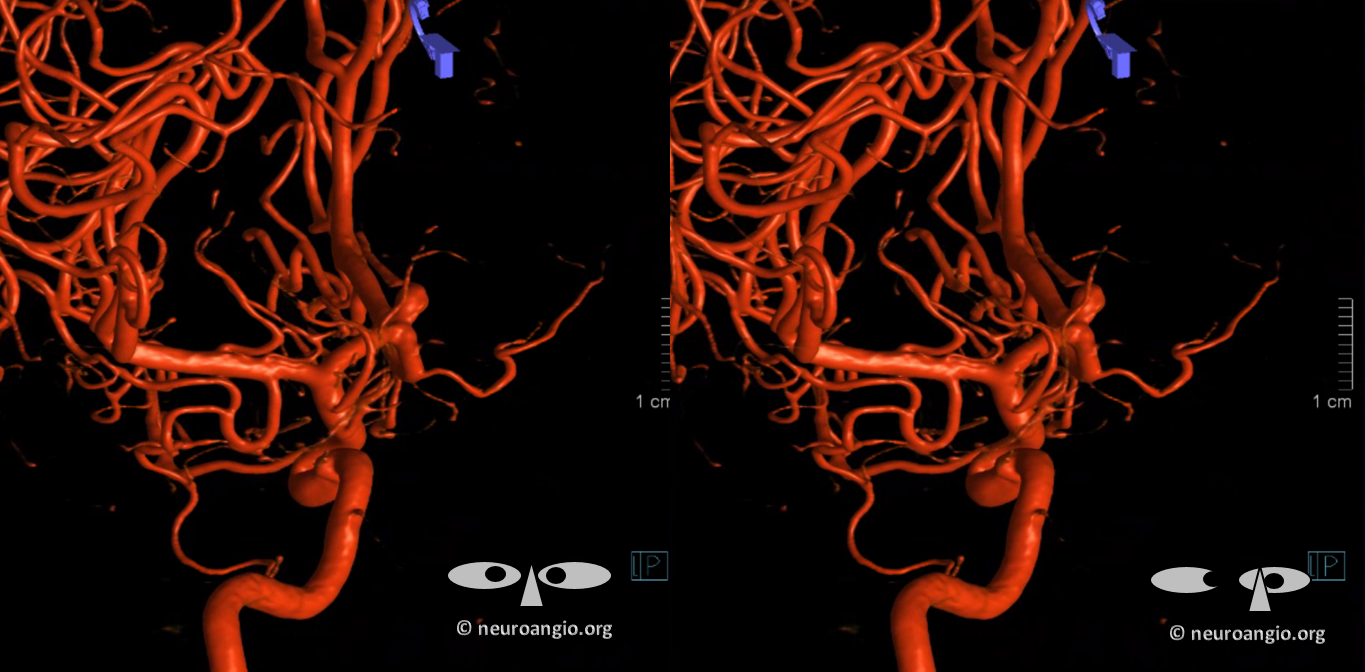
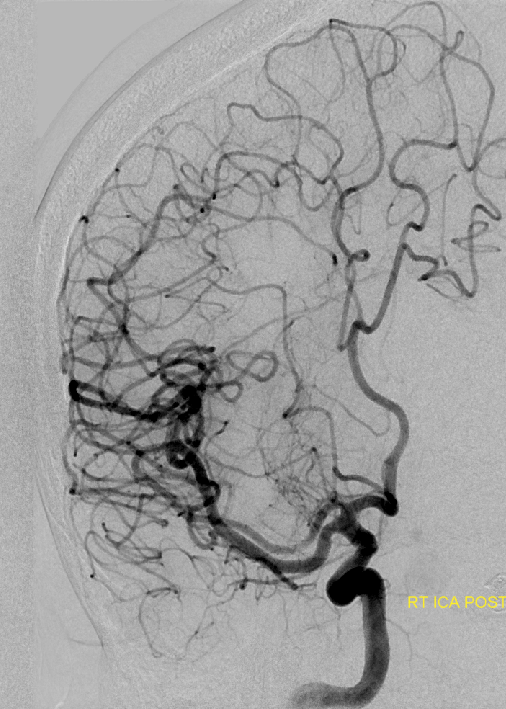
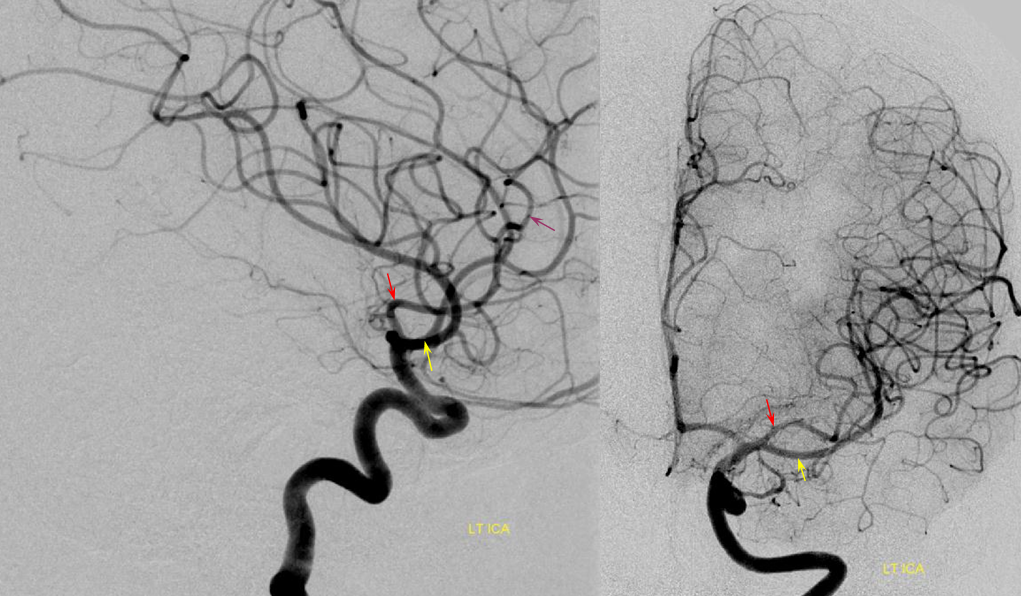
Here is a highly dominant lenticulostraite trunk arising from the proximal A1 segment, in patient with supraclinoid athero. This is not a Heubner, since it does not supply cortical territory. Stereo view from the back.
Heubner, speaking of
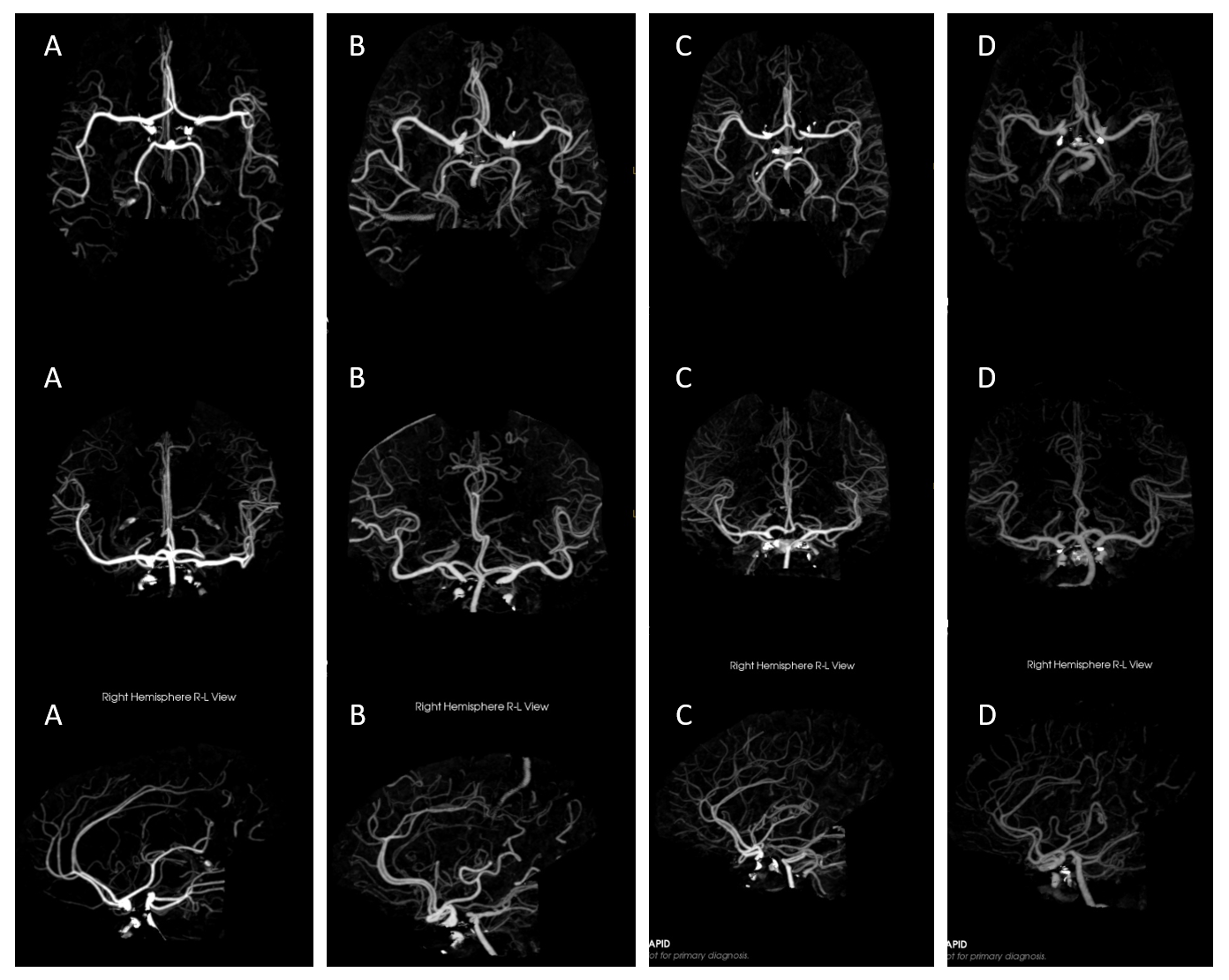
Here are some images. Modern 3T MRA can often visualize a Heubner. This is a very clear demonstration that Heubner and medial perforators off the MCA serve the same function. In this patient, a large Heubner on the left serves the same function as the medial M1 perforators on the right, supplying the putamen, caudate, and parts of the anterior limb of the internal capsule. Short M1 segment on the left is associated with typical superior division M2 origin of lateral perforators
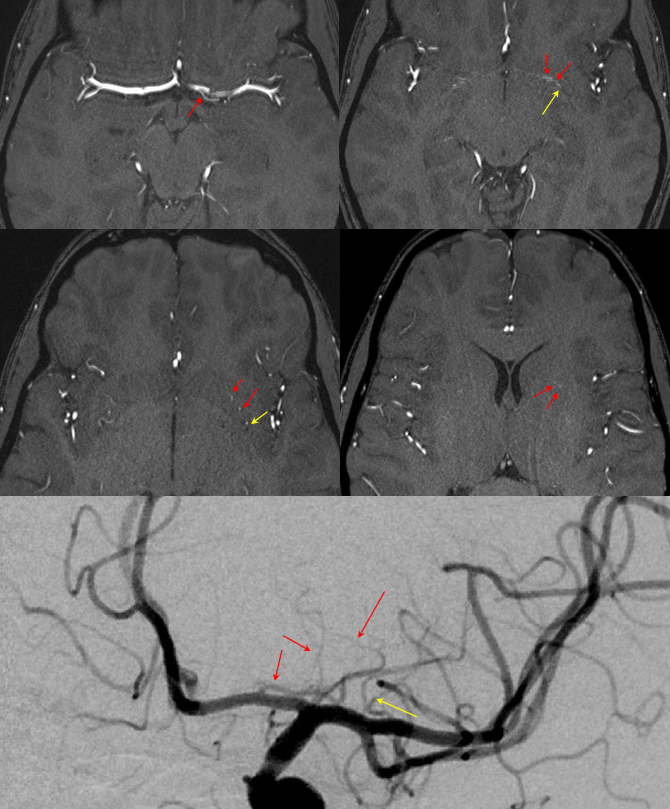
Right MCA, same patient
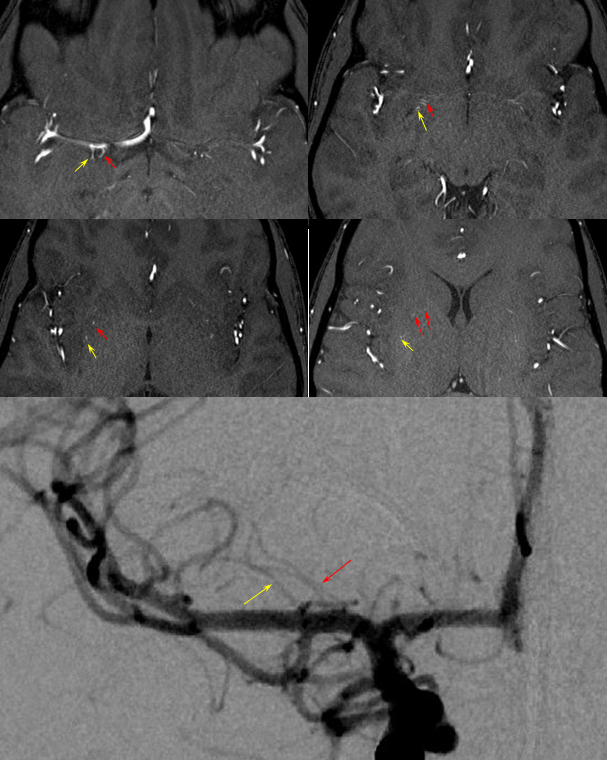
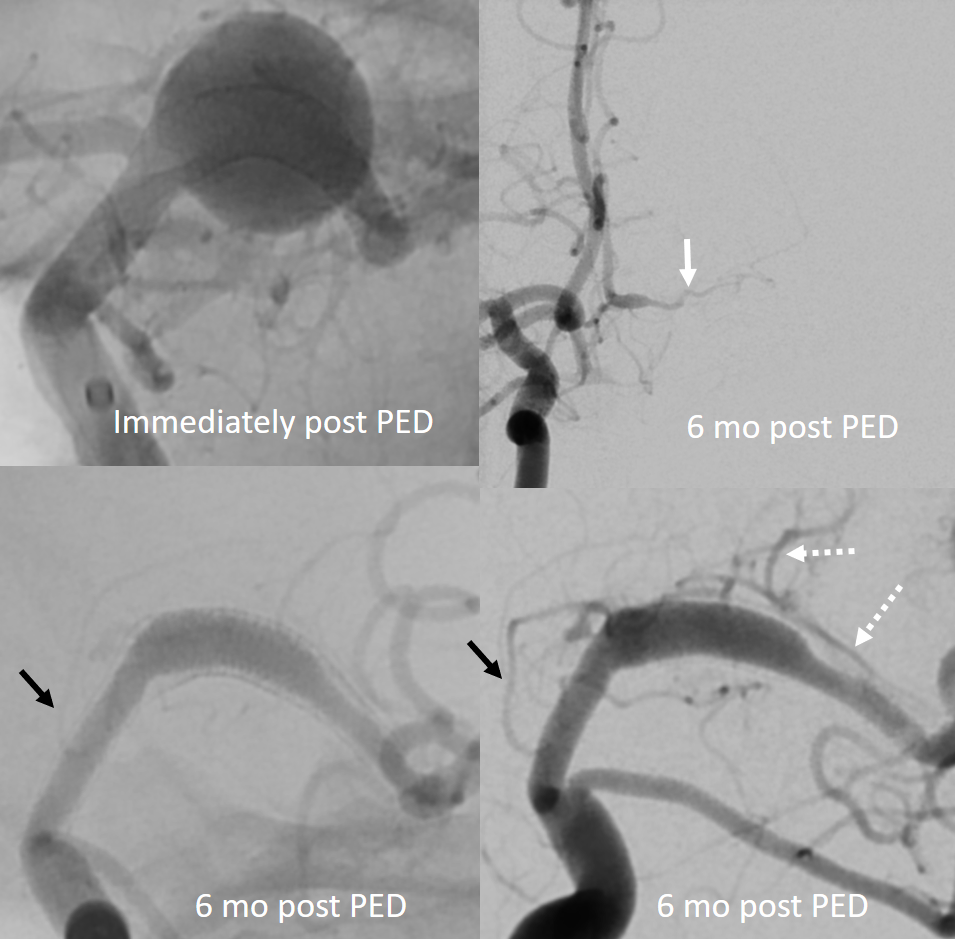
Whats an MCA page without an aneurysm? Here is a fusiform M1, just post and 6 mo after Pipeline embolization. Cure of aneurysm has been associated with expected post-pipeline hemodynamic rearrangements, including transfer of left ACA territory to the right A1 and ACOM. A large Heubner (white arrow) is present. The lateral lenticulostriates arise disal to the aneurysm (dashed arrows). Anterior choroidal is black arrow. None were found to arise from the aneurysmal segment. Case courtesy Dr. Daniel Sahlein.
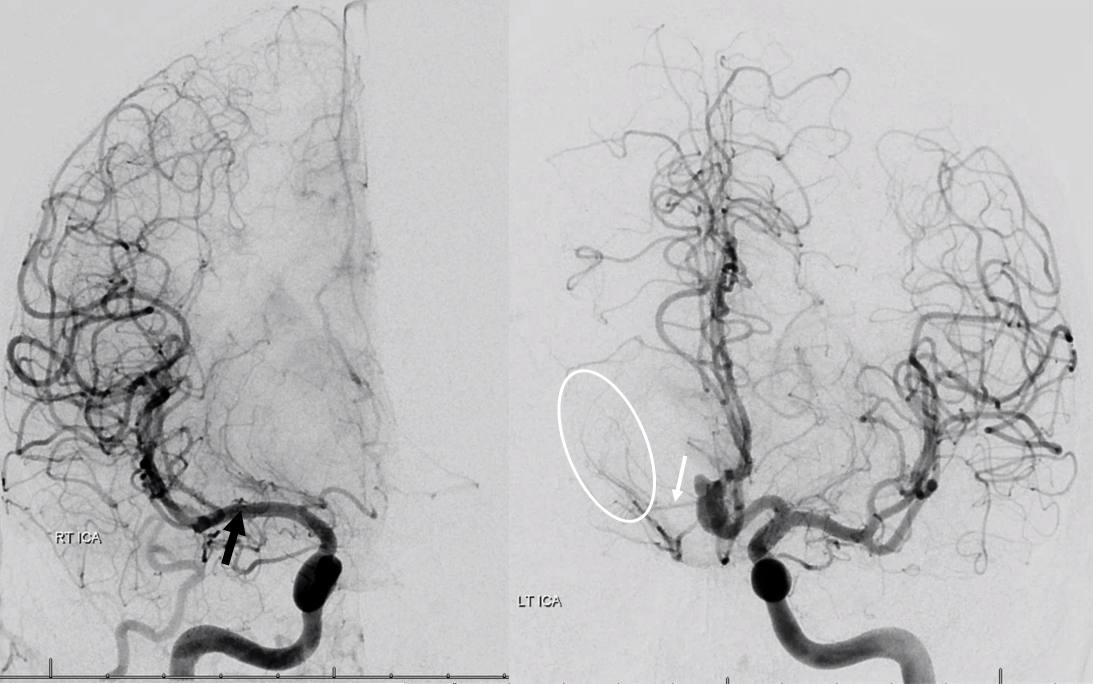
Just as the Heubner is usually best seen from contralateral injections, so are the medial lenticulostriate arteries associated with it. Here is an example of an aneurysm case. The relatively large Heubner (white arrow) is associated with well-developed medial lenticulostriate vessels that also supply part of the traditional “lateral lenticulostriate” territory (white oval). On the right, developmentally hypoplastic A1, some proximal A1 lenticulostriates (not marked) and distal M1 lateral lenticulostraite origin (black arrow)
Early arterial phase (left) and late arterial phase (right) oblique views, showing the Heubner (white arrow) and reflux into the right A1 segment (dashed arrows)
Duplicated/Accessory MCA
How do we conceptualize that? Very simple. Any place that “gives rise” to lenticulostriate vessels can be a source of MCA, according to above dicussion of MCA evolution. Sometimes, two co-dominant cortical vessels exist instead of one. Extremely rarely, there can be three. They can be equal or unequal in size depending on what they supply. Same can be said of the M1 segment. It can be infinitely long when there is no M1 bifurcation. A progressively short M1 segment blends into a duplicated MCA. So does an an anterior temporal branch which can arise from the M1 trunk as well as from the carotid terminus or below. Its all about how you see it.
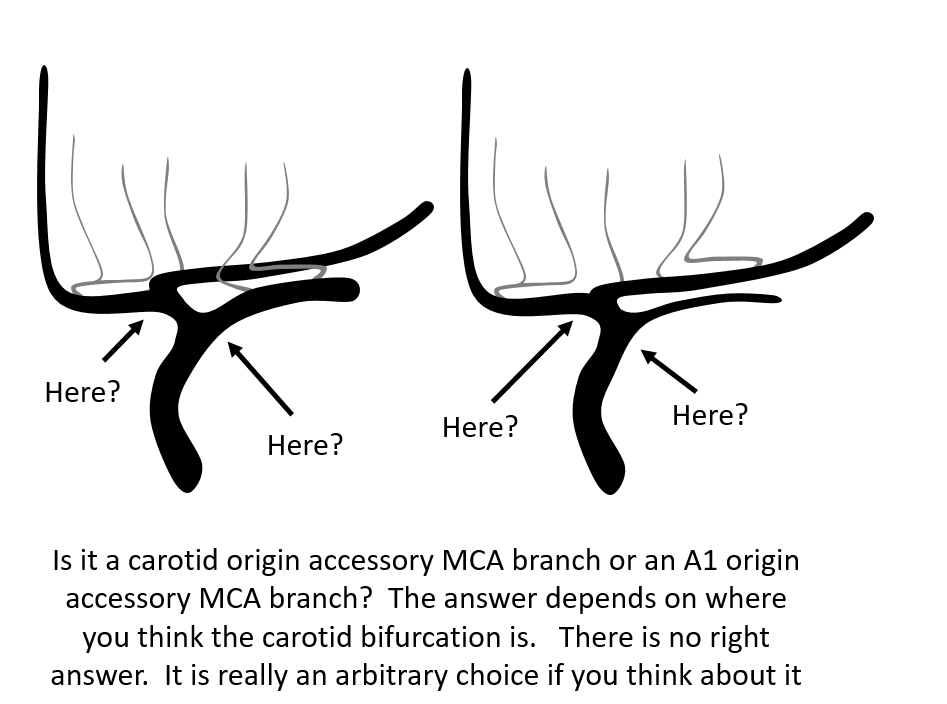
Look at the figure below. The whole duplicated vs. accessory MCA nomenclature is based on an arbitrary decision of where the carotid bifurcation is. Sure, we can just say that the biggest branch is the “true” bifurcation. But this has no embryological or functional significance. Embryologically, all of MCA or MCAs come from ACA. Functionally, the only things that matter are which territory each trunk supplies and where do the lenticulostriates come from.
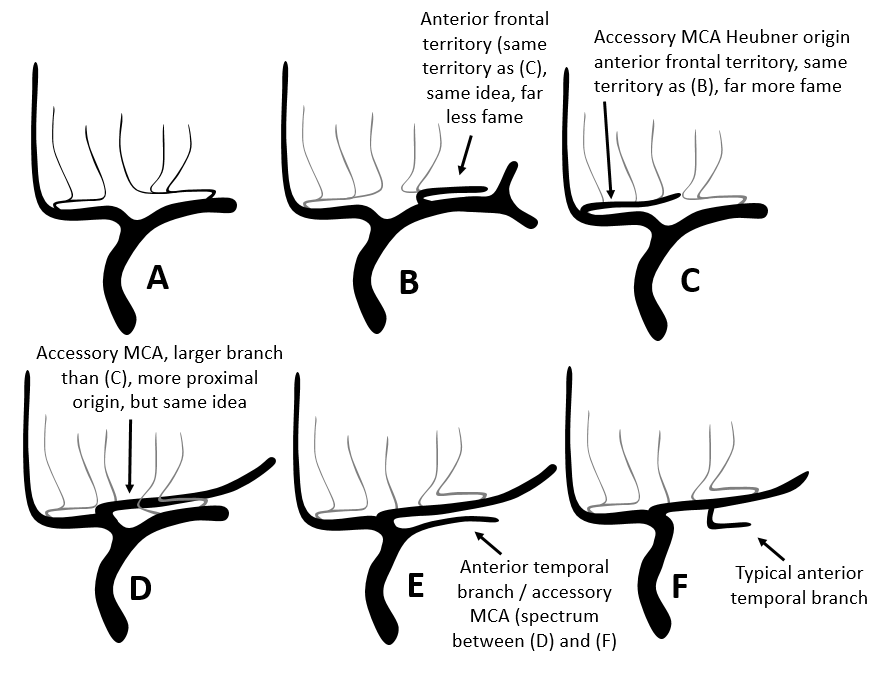
So, back to the above figure. A is usual. B shows a rarely appreciated but not uncommon “early origin” anterior frontal branch arising from the lateral lenticulostriate trunk. This is really no different than (C) when the same anterior frontal territory branch happens to arise from the ACOM region — except here it is called a Manelfe Type III accessory MCA. Another “accessory MCA” arises from the A1 segment (D) — or does it? Where again is the carotid bifurcation — perhaps it is the inferior division that “arises” from below the carotid terminus? How about (E) — the anterior temporal branch, instead of its usual origin (F) has a more proximal location separate from the main MCA trunk. All of these are part of a continuous spectrum. If you want to get even more philosophical, vasculature is like an Edison light bulb — a continuous spectrum. Not like discrete bands of LED or halogen bulbs.

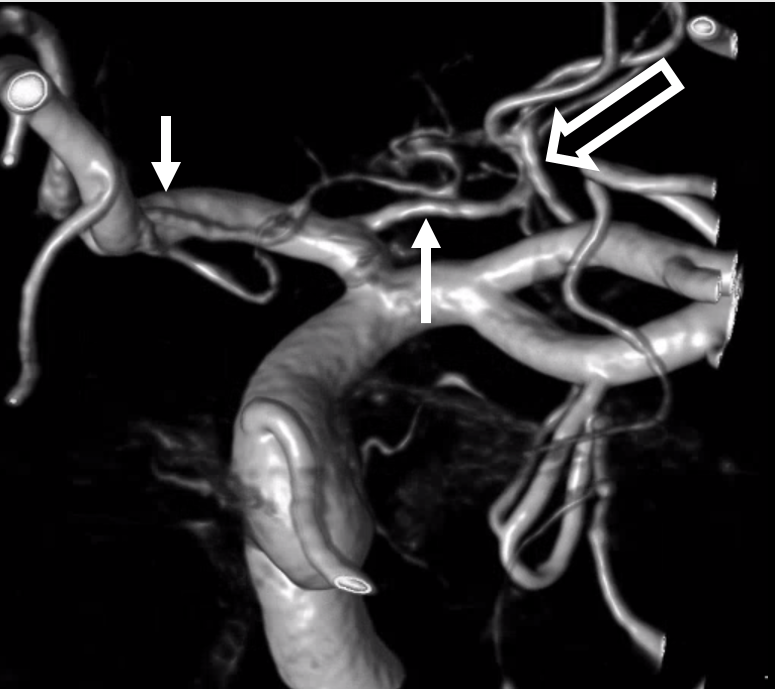
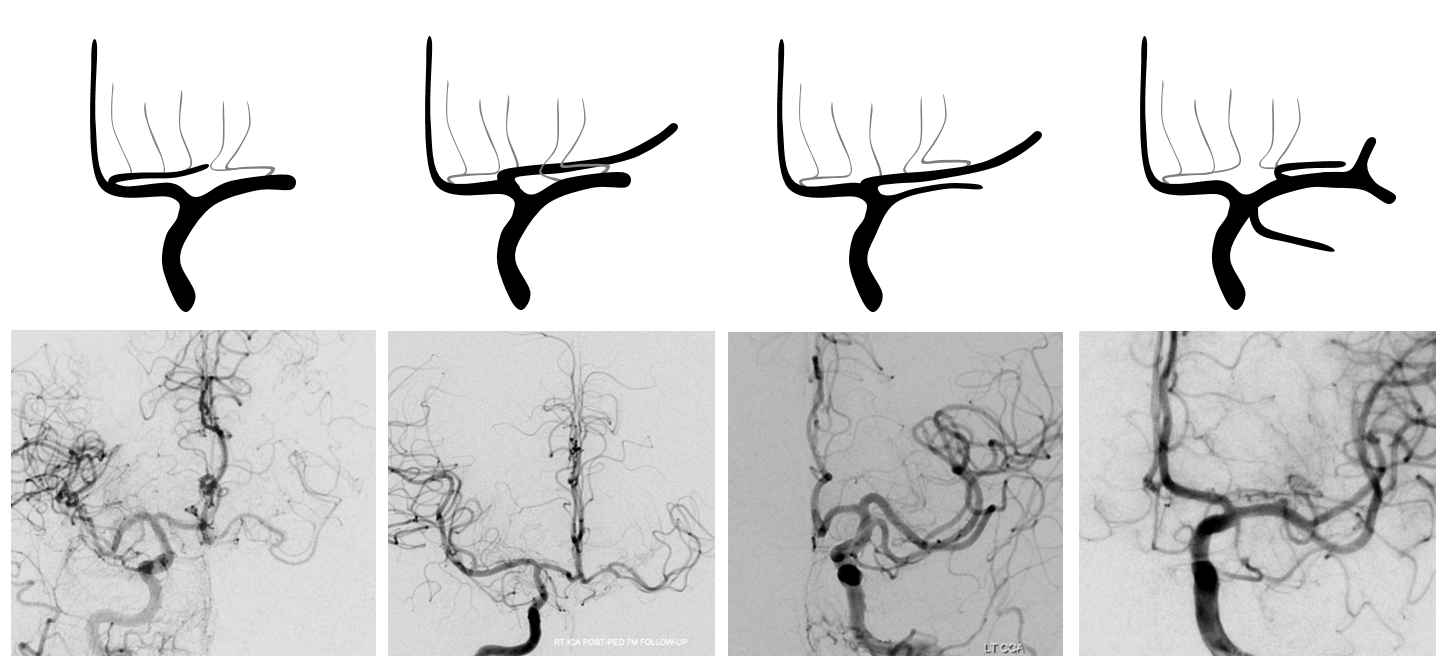
How about some real examples? Look at a spectrum of accessory / duplicated MCAs corresponding to each schematic
Here is a figure from our embryology page, with a “duplicated” MCA on the anatomical right (purple arrows) and a “normal” recurrent artery of Heubner, with a Heubner origin accessory MCA (aMCA) on the left
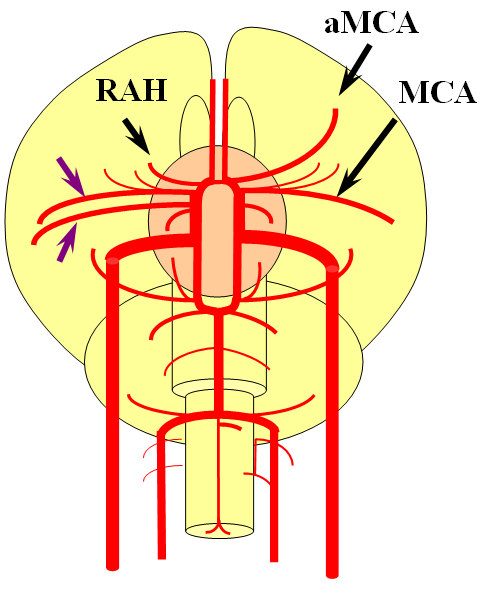
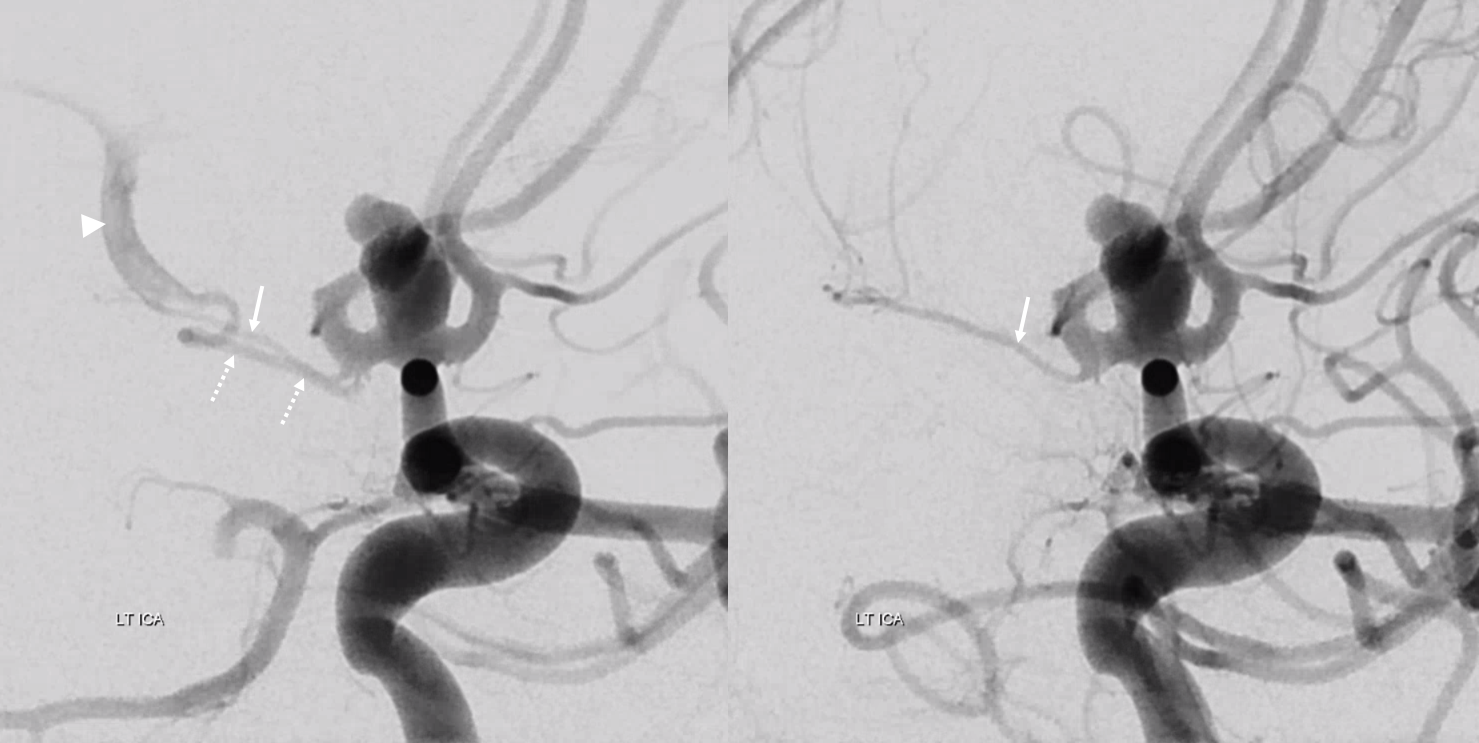
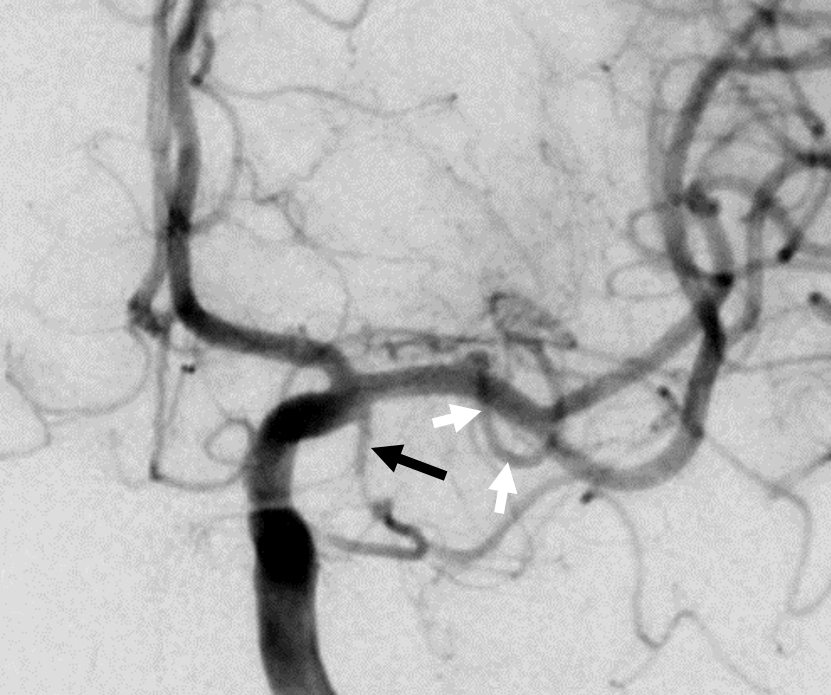
An angiographic example — usually the Heubner — and an accessory Heubner MCA — are better seen from the contralateral injection
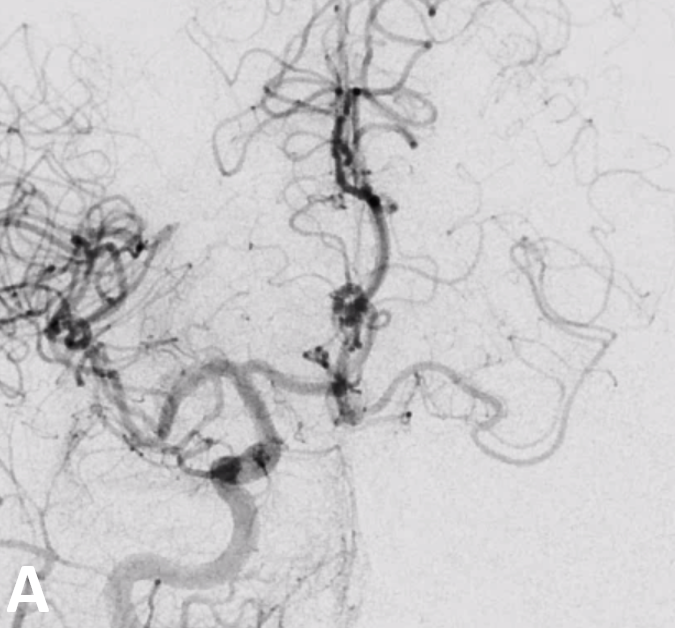
Here is a mid-A1 origin accessory MCA — all part of the spectrum. Same patient, right and left ICA injections
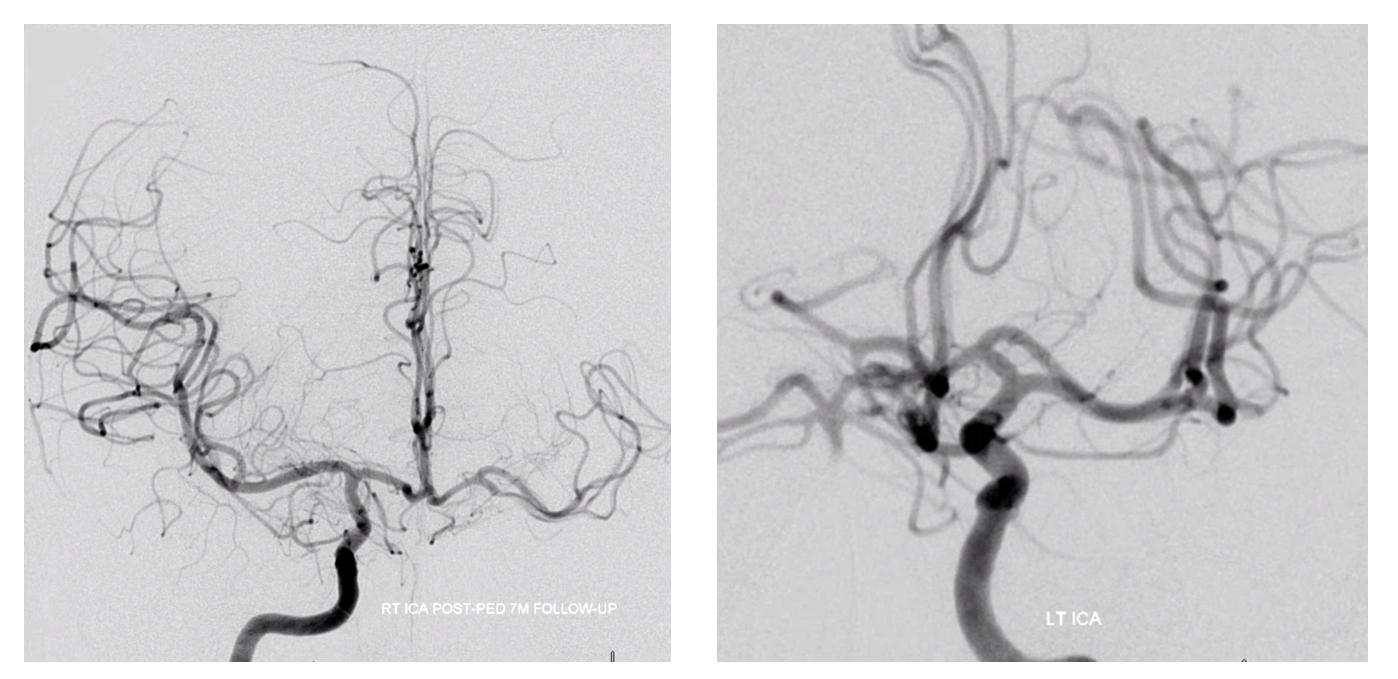
Angio and CTA of same patient
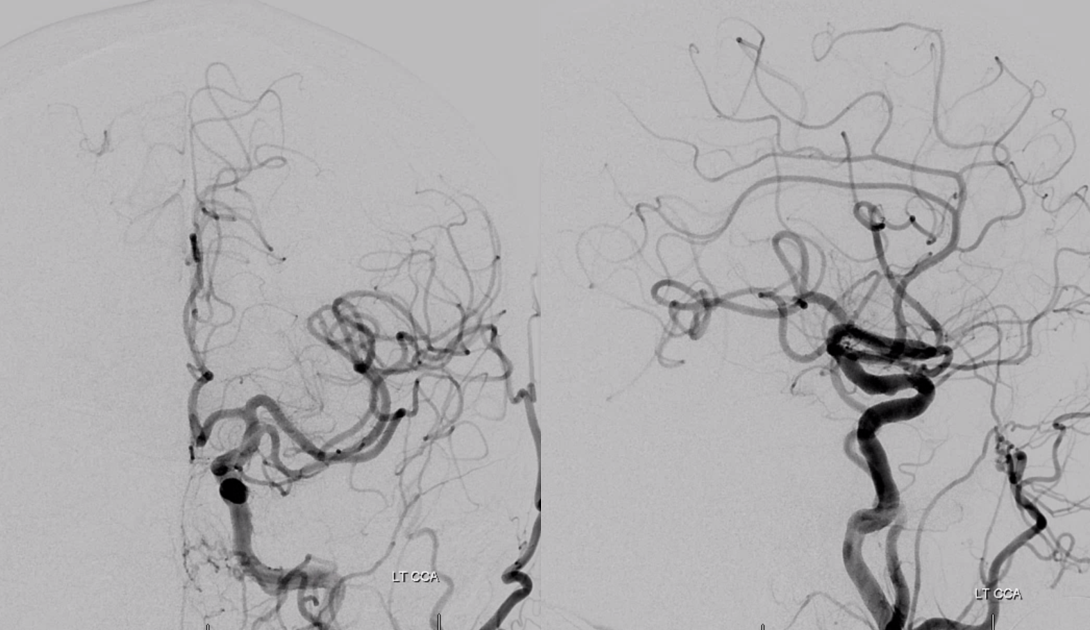
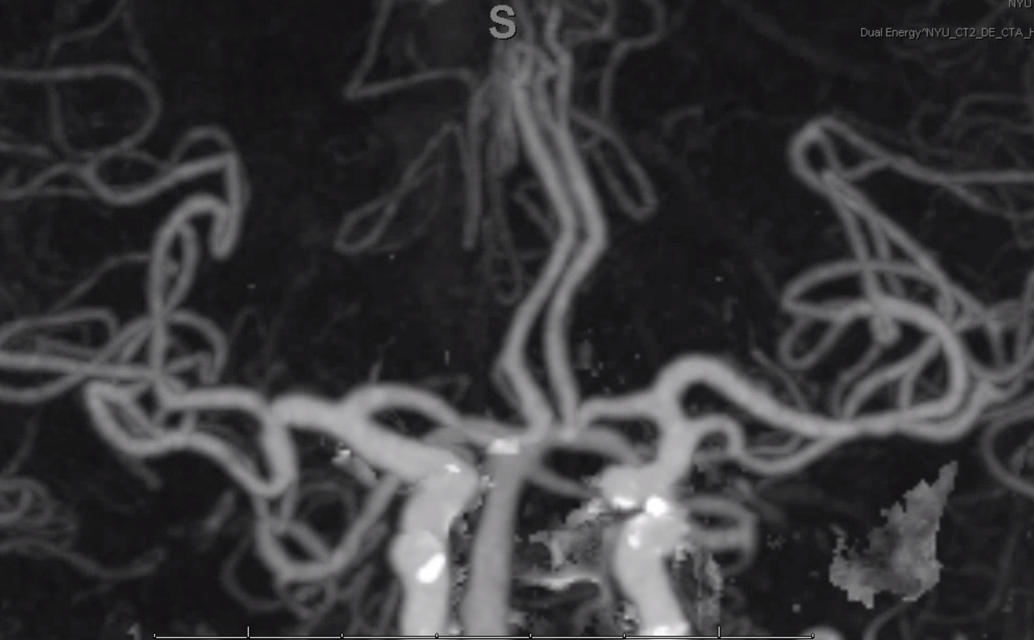
Stereo
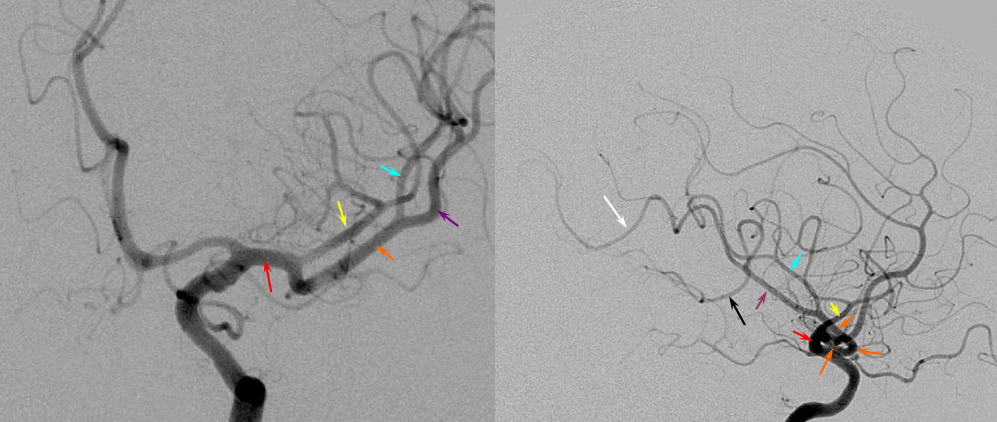
Case with both early origin anterior temporal branch (black arrow) and anterior frontal branch (white arrows). Also shown above. The anterior frontal branch is not at all recognized as being similar to a Heubner type aMCA, even though it is
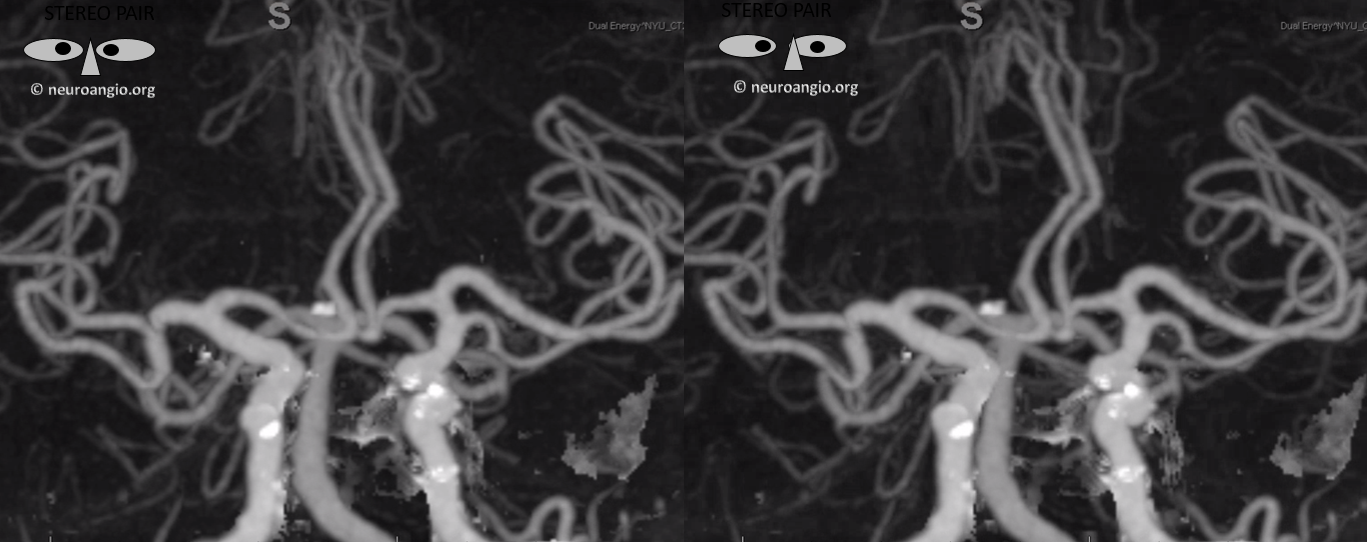
Anaglyph stereo of same
Accessory MCA DYNA CT, supplying substantial temporal territory (arrows)
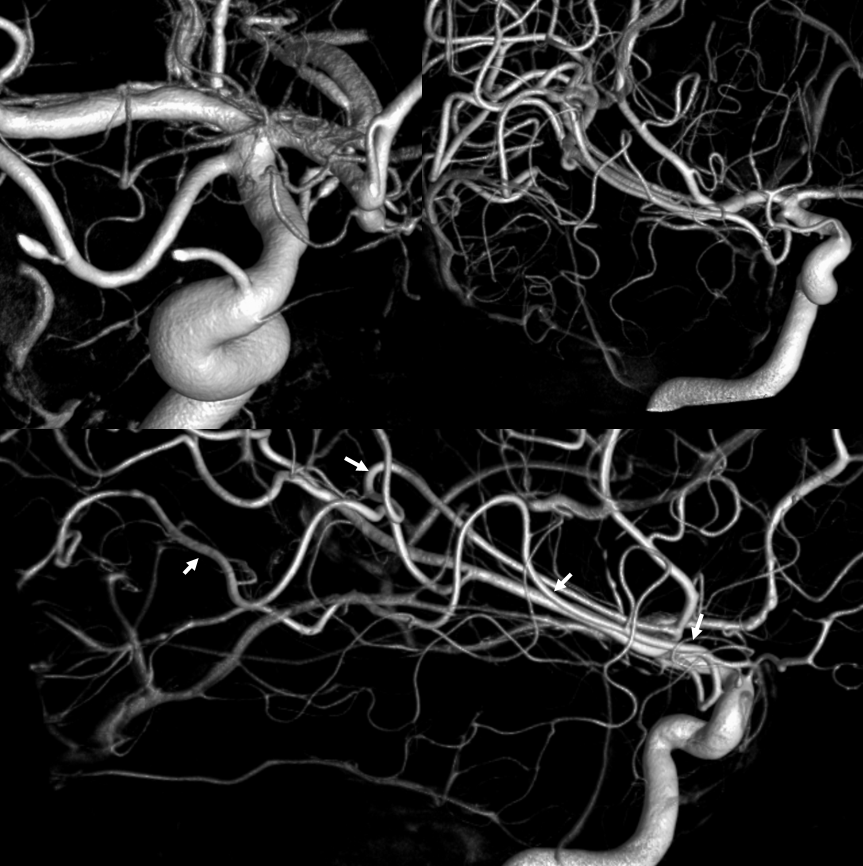
Clinical Relevance
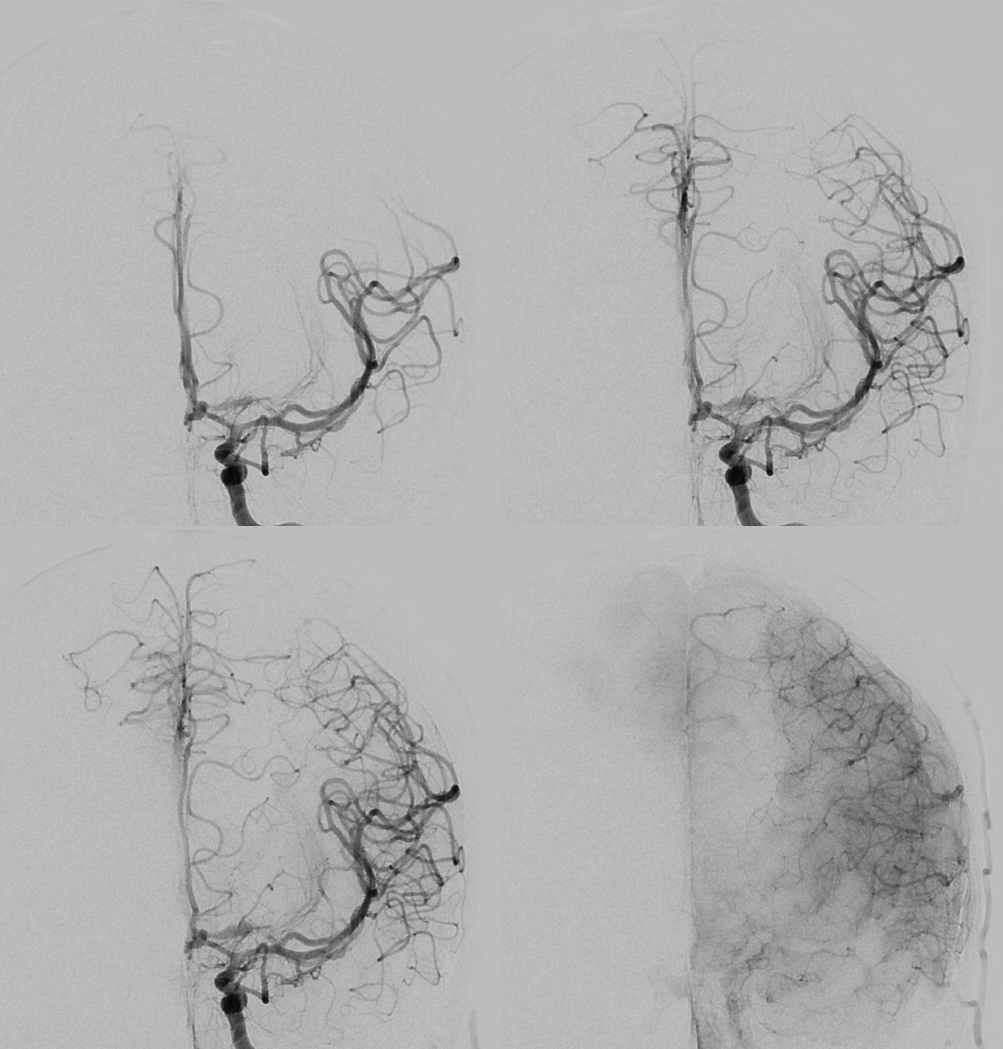
How about some strokes? Look at this case, courtesy Dr. Daniel Sahlein. An MCA occlusion before (left) and post revascularization (center). Another embolus is lodged in the superior division territory of accessory MCA (white arrow). Final post is on the right
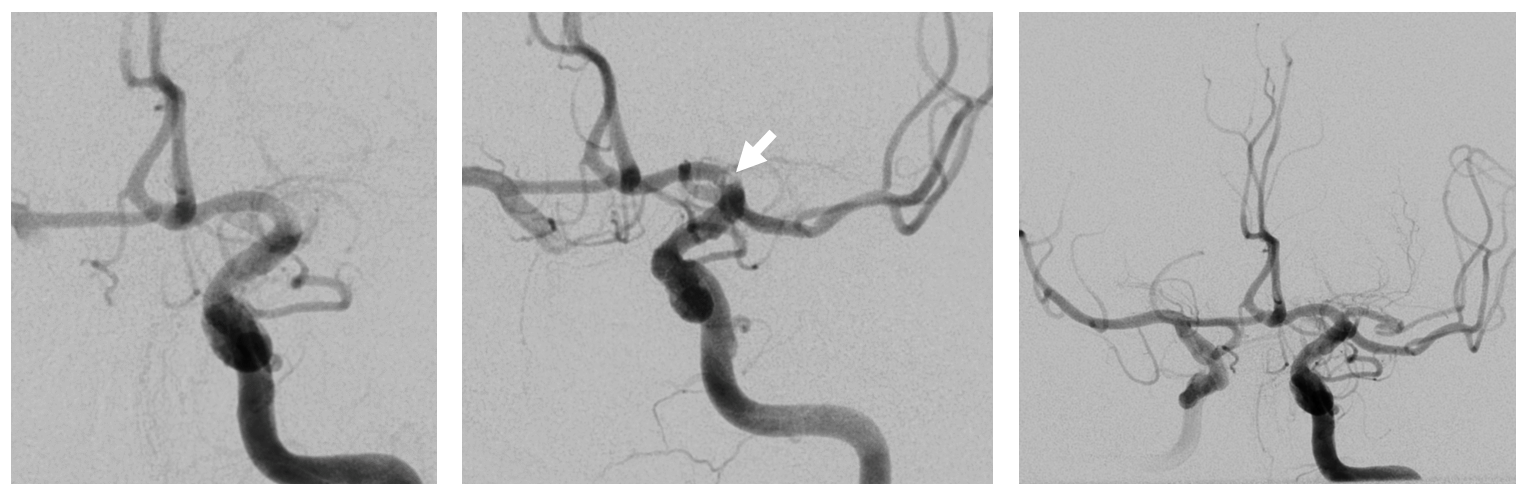
Another case — mid “M1” occlusion with a patent larger caliber anterior temporal branch — there is semantic continuum between anterior temporal branch and inferior division, depending on how much territory it supplies. This is about half of temporal lobe. Post thrombectomy is on right
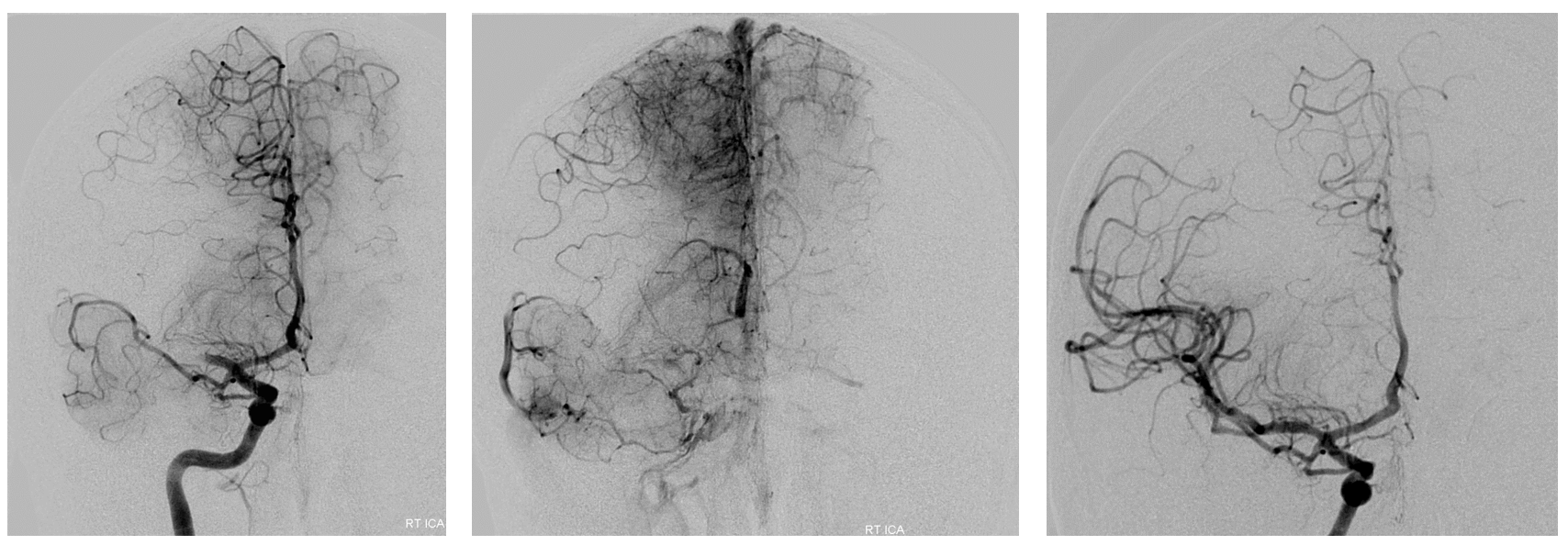
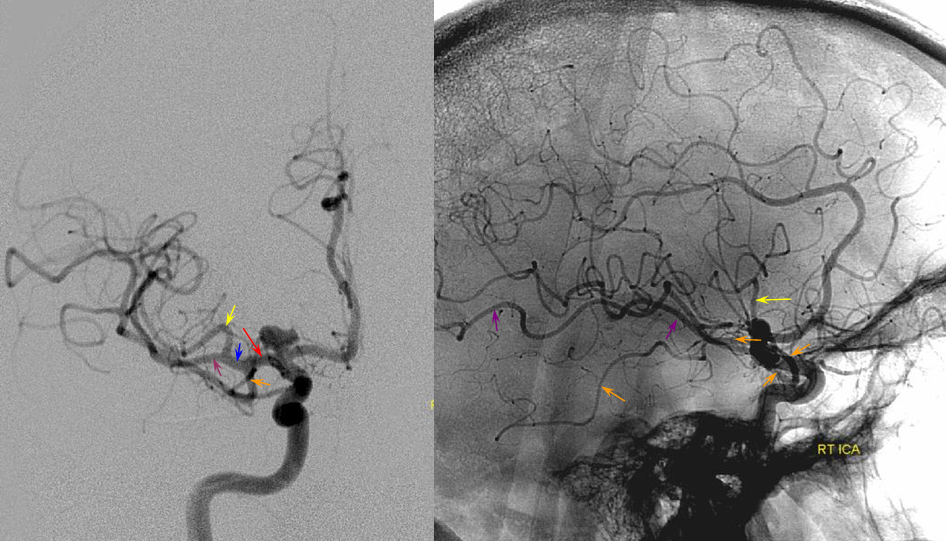
Third case — carotid terminus embolus. A Heubner (Manelfe III) accessory MCA remains patent (center). Lateral view of left ICA injection shows its frontal opercular territory — as usually the case, Heubner aMCAs go frontal.
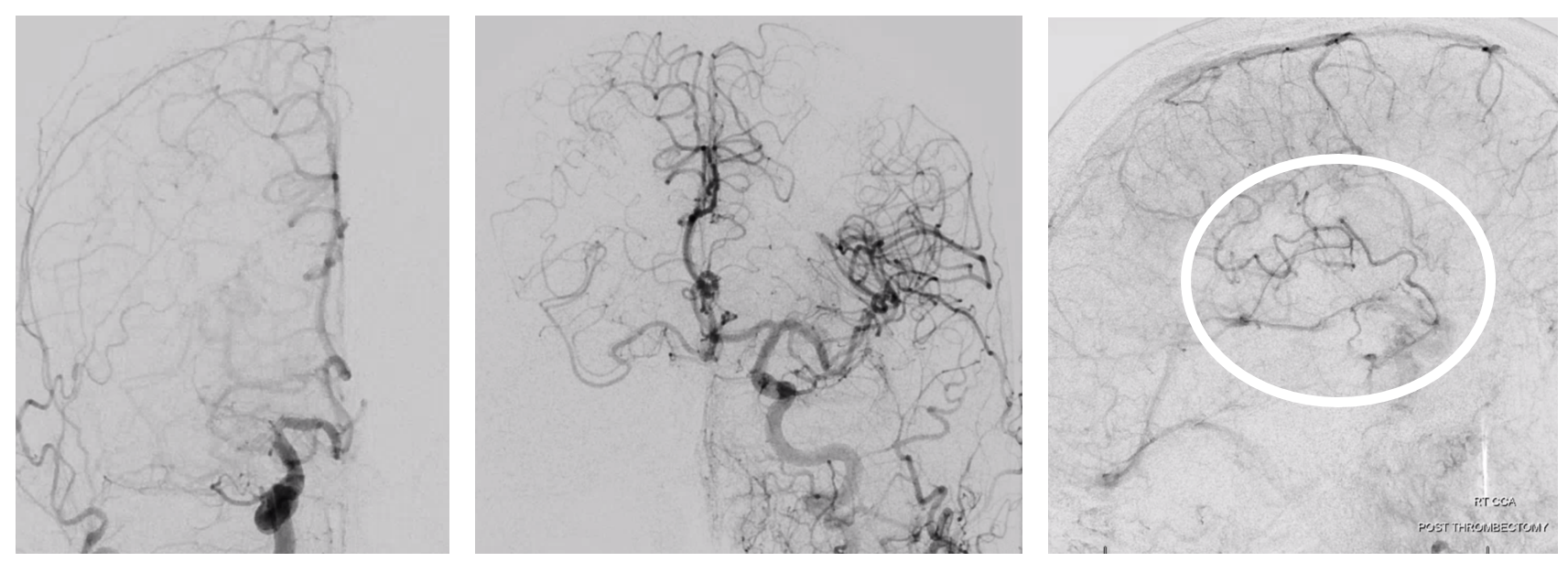
Accessory MCA Ruptured Dissecting Pseudoaneurysm
Case Courtesy Dr. Howard Riina. Presentation is SAH
CTA — pause and scroll through individual images
See how faint the accessory MCA trunk is just distal to the pseudoaneurysm — its the flattening of the dissecting lumen
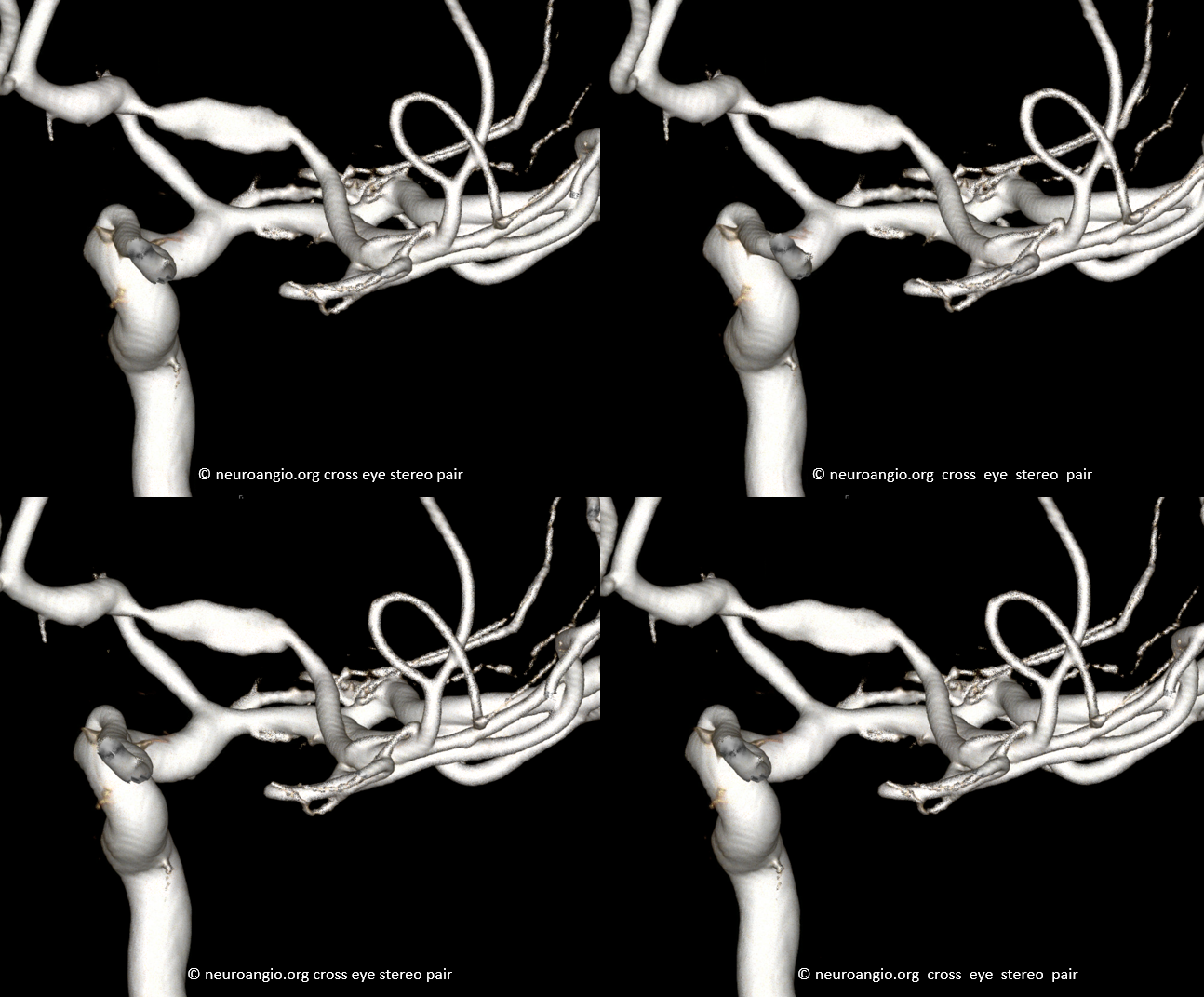
Stereo VRs — notice the typical narrowing at both sides of the pseudoaneurysmal segment. The mechanisms of spontaneous intracranial dissection remain unclear.
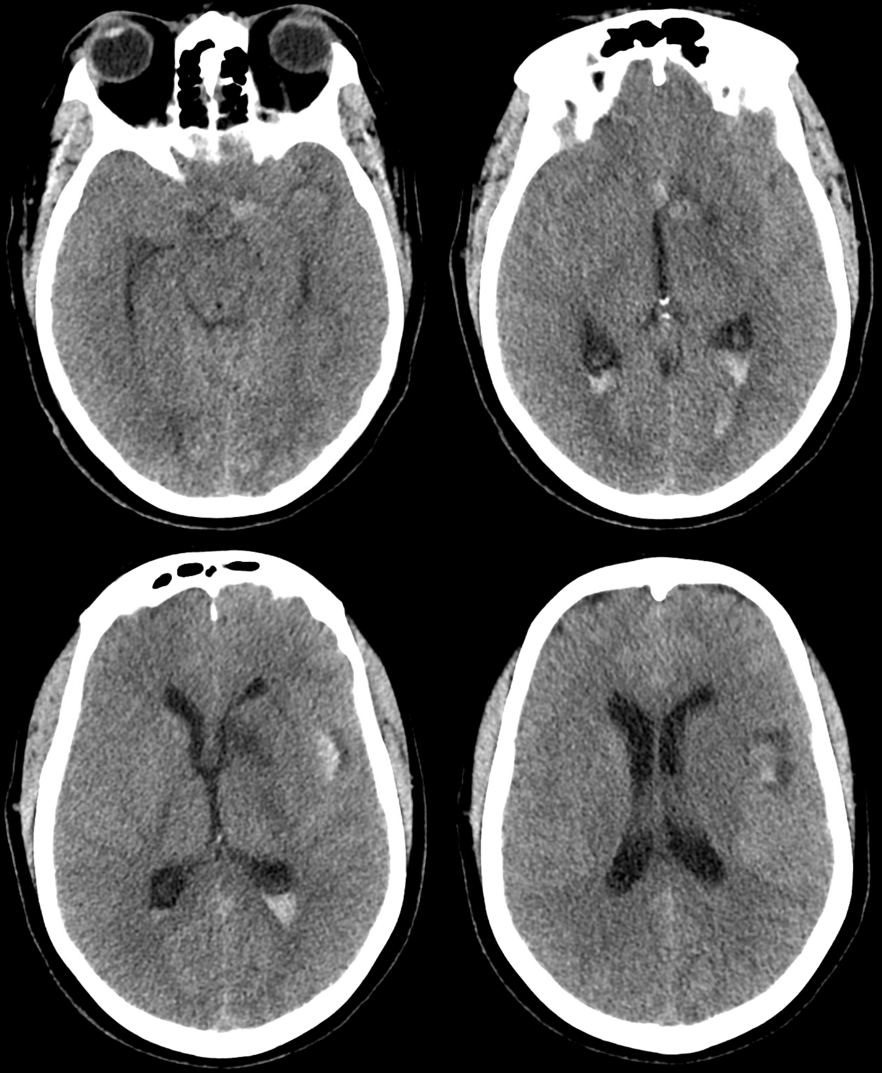
Later, there is telltale medical lenticulostriate infarct from a Heubner-like branch that must have gone down
Hemispheric Branching Pattern
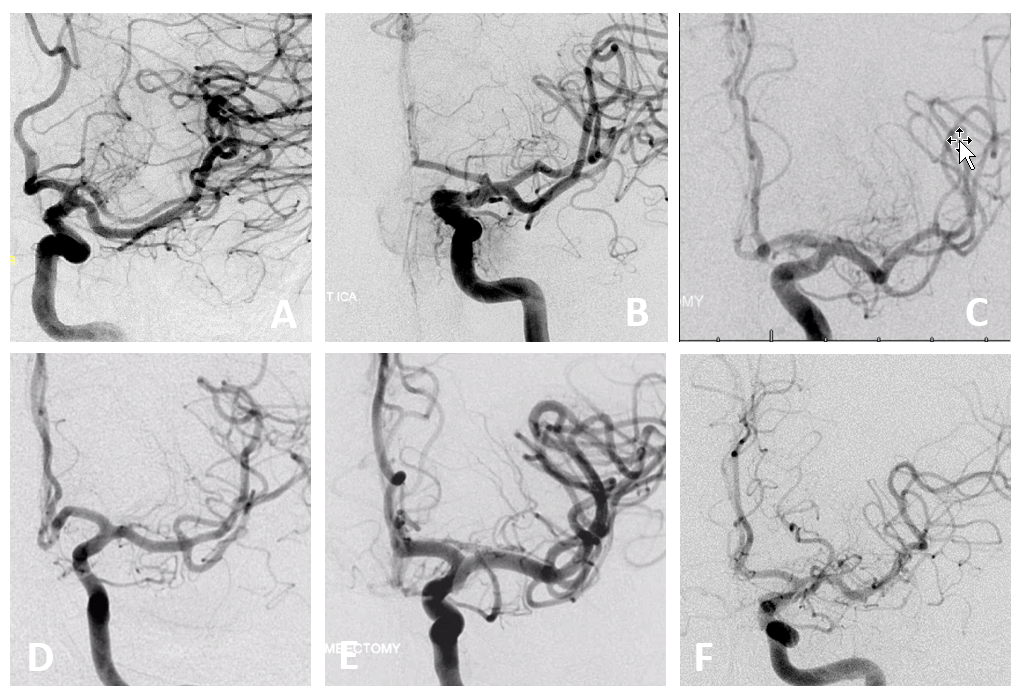
Very simple — no rules. Just look at these. A — “duplicated” MCA — on the spectrum with short M1 (B). Dominant superior division (C). Trifurcation (D). Pentafurcation (E). No bifurcation (F) — just individual origins of branches
More pretty examples — no bifurcation case in cross-eye stereo
Same case, anaglyph stereo
Lateral Anaglyph, so u dont think we are hiding the bifurcation here
CTA — Its not just angio — look at a selection of these RAPIDs below, that came thru over 2 days. A – on right there is no bifurcation at all — pine tree style; B — super long “M1” segment — the bifurcation is at the upper end of the vertical sylvian segment — which would be distal surgical M2… C — a right trifurcation. On left who knows where the bifurcation is. The first branch is either a large anterior temporal branch or a small inferior division. More distally there is another major bifurcation — for the superior division and what’s left of the inferior one. D — something “classic” for a change…
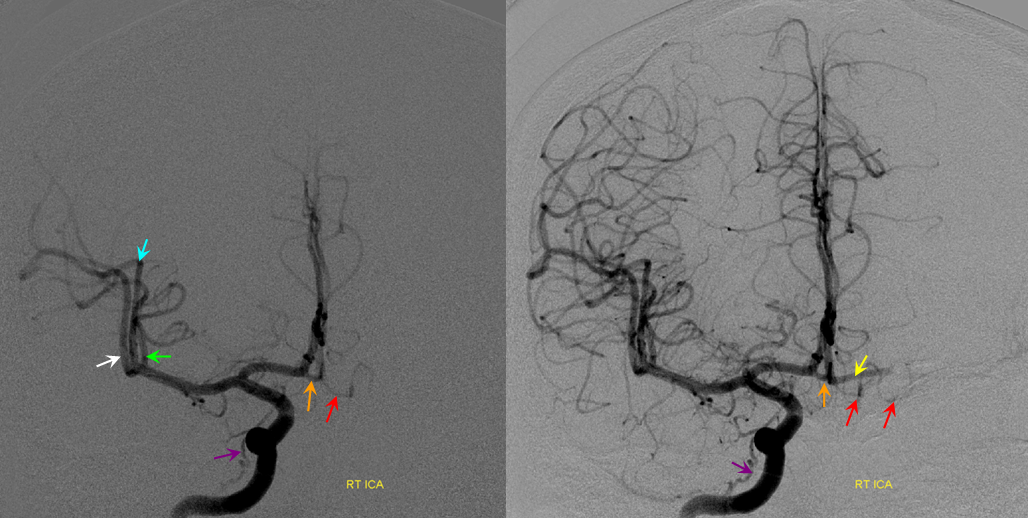
Below — dominant inferior division — short M1 segment (red) with smaller superior division (yellow) supplying the frontal convexity, and larger inferior division (orange) ointo the the temporal lobe (purple, subdividing into black anterior and white posterior temporal and white parieto-occipital) and parietal lobe (blue) feeders.
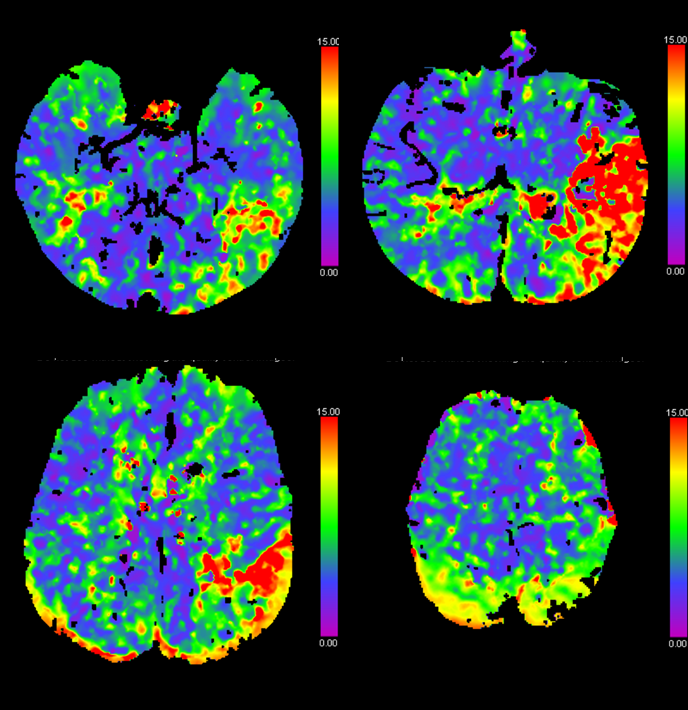
Dominant inferior division demonstrated by thromboembolic occlusion
No better way to define a territory than by its occlusion. Here is a very dominant inferior division with embolus in it (arrows). The patent superior division only supplies the anterior and mid-frontal lobes (white oval). The inferior division does posterior frontal, parietal, and temporal regions. Notice leptomeningeal parietal territory support in late venous phase (black oval)
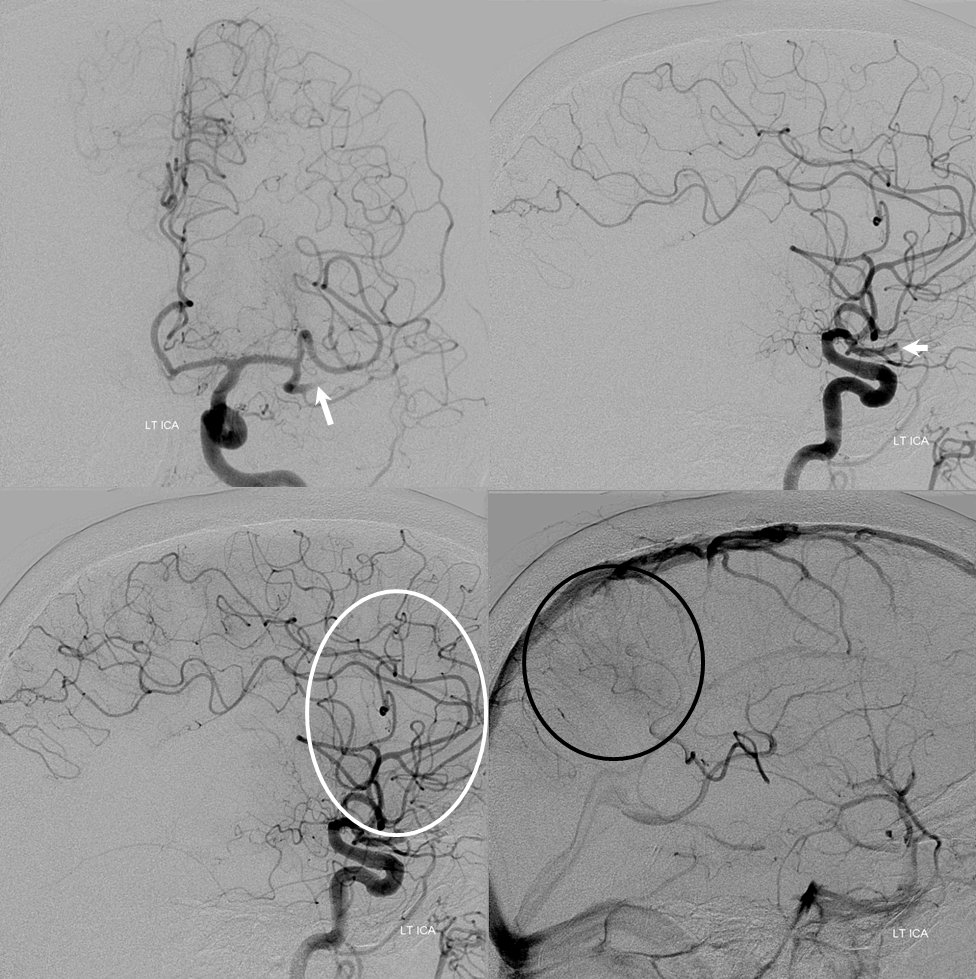
CTP of the same patient, Time to Drain. The inferior temporal lobe is well collateralized by the PCA, while the superior convexity collaterals come from the ACA, as seen in the angio above. The superior and mid-temporal gyri are not in such good shape. As you know, the embolus favors the largest branch with greatest flow.
How about this branching pattern — fit any mold? Of course not
A more classic situation of early bifurcation with dominant inferior division (yellow).
Trifurcation here
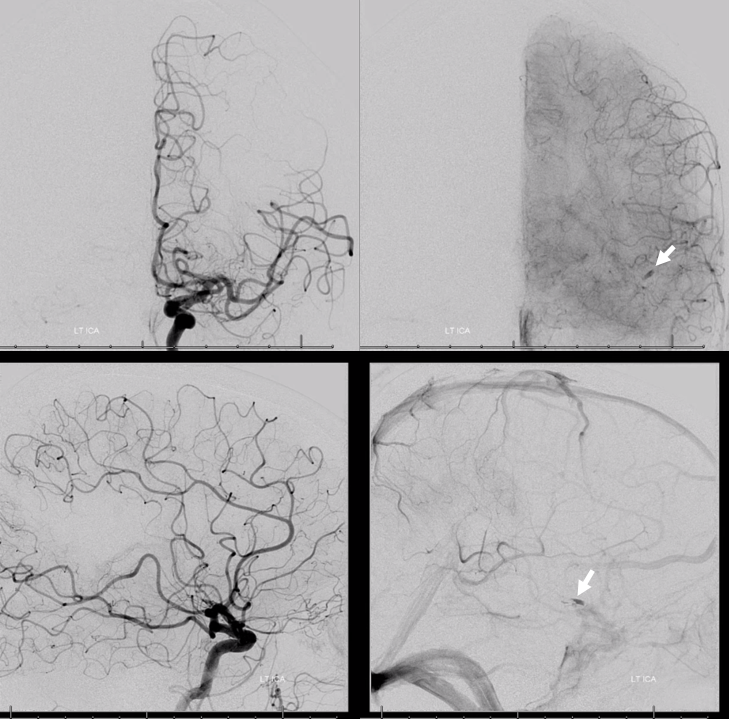
No MCA bifurcation thrombectomy
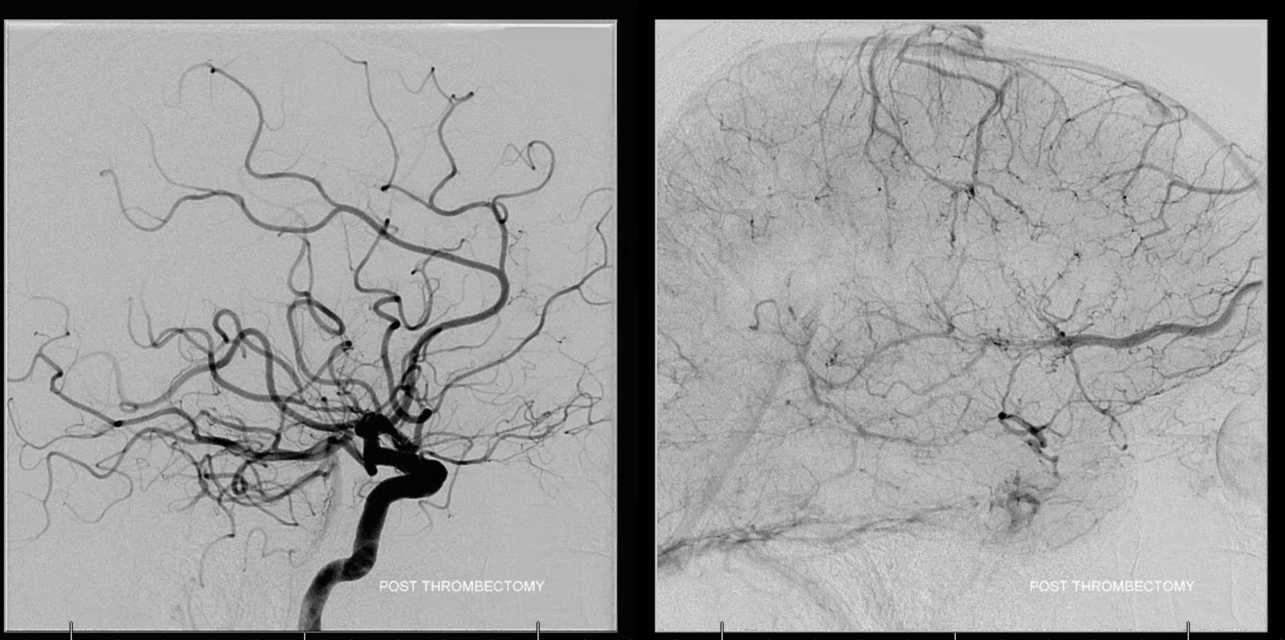
On the opposite end of the silliness spectrum is an MCA with no bifurcation. Not too common, but serves the same point — anything in MCA is possible. A single stem with individual branches, like a pine or a palm tree, depending on your mood — why not? Here is another stroke, with the embolus (white arrows) stuck in the stem of your tree of choice, somewhere in the proximal third — a few branches to the temporal and frontal lobes are proximal, the parietal territory is mostly distal to the occlusion. That hum-drum early anterior frontal branch is present again (purple), but no anterior temporal branch is seen. Again, anything goes. Bottom set of images after revascularization show persistent lack of bifurcation, with the reopened temporal and parietal destination trunk marked by yellow arrow.
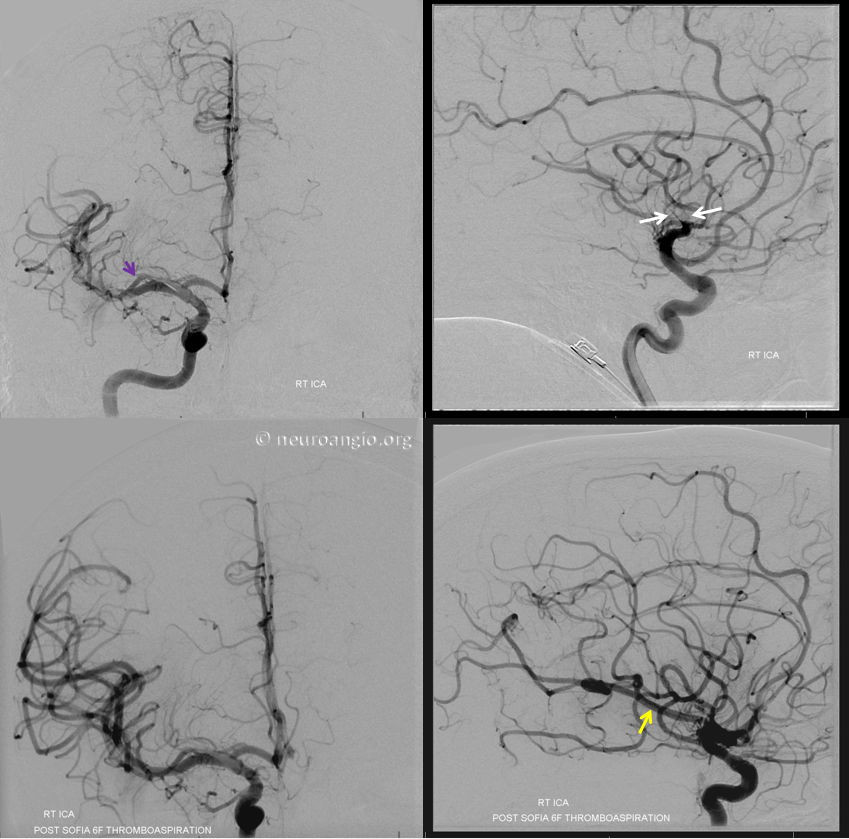
“Trifurcation thrombectomy”
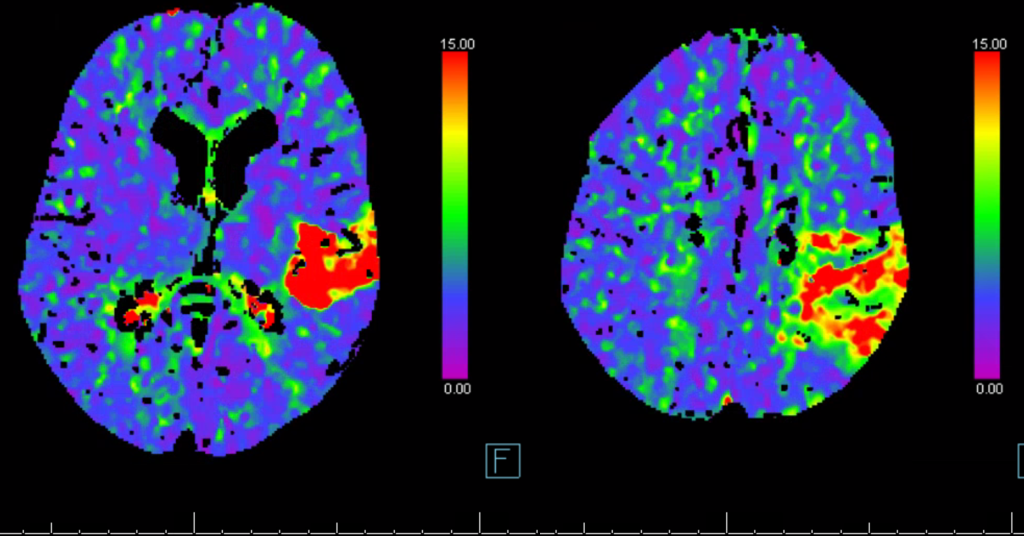
Again, life makes its own rules. Mean Transit Time maps of MCA branch occlusion, affecting posterior temporal, posterior frontal, and anterior parietal lobes — almost a full MCA syndrome
Angio shows an MCA trifurcation — which is supposed to mean that each branch takes its own frontal, parietal, and temporal lobes. Of course they don’t. The embolus (white arrows) lodged in the functionally critical middle branch, along vector of flow. Lateral views show its posterior temporal and frontoparietal territory. This is yet another example relatively medium volume occlusions producing out-sized clinical syndromes
Post-thrombectomy. A nice venous variant of large anterior frontal vein.
In the end, the thing that is most important is territory of supply. Look for territory at risk and make your decisions based on that.

MCA fenestration
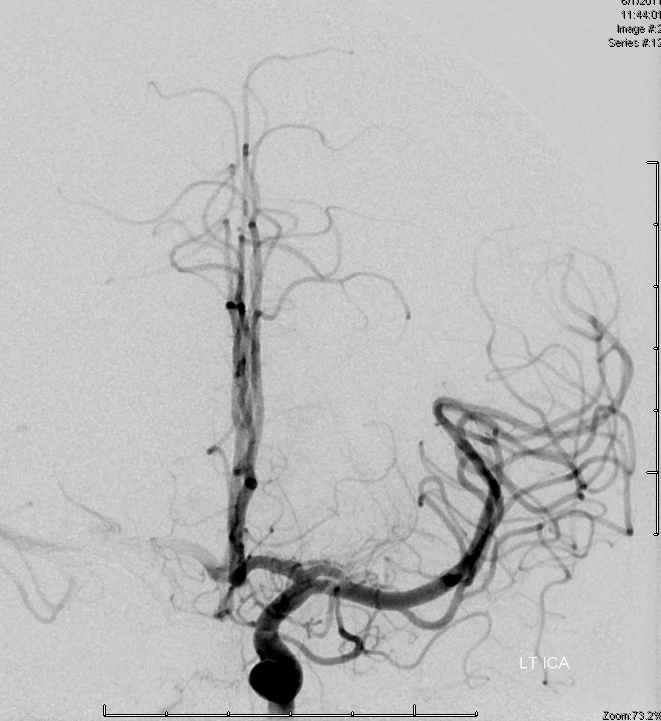
Here is another one, in a patient with contralateral AVM
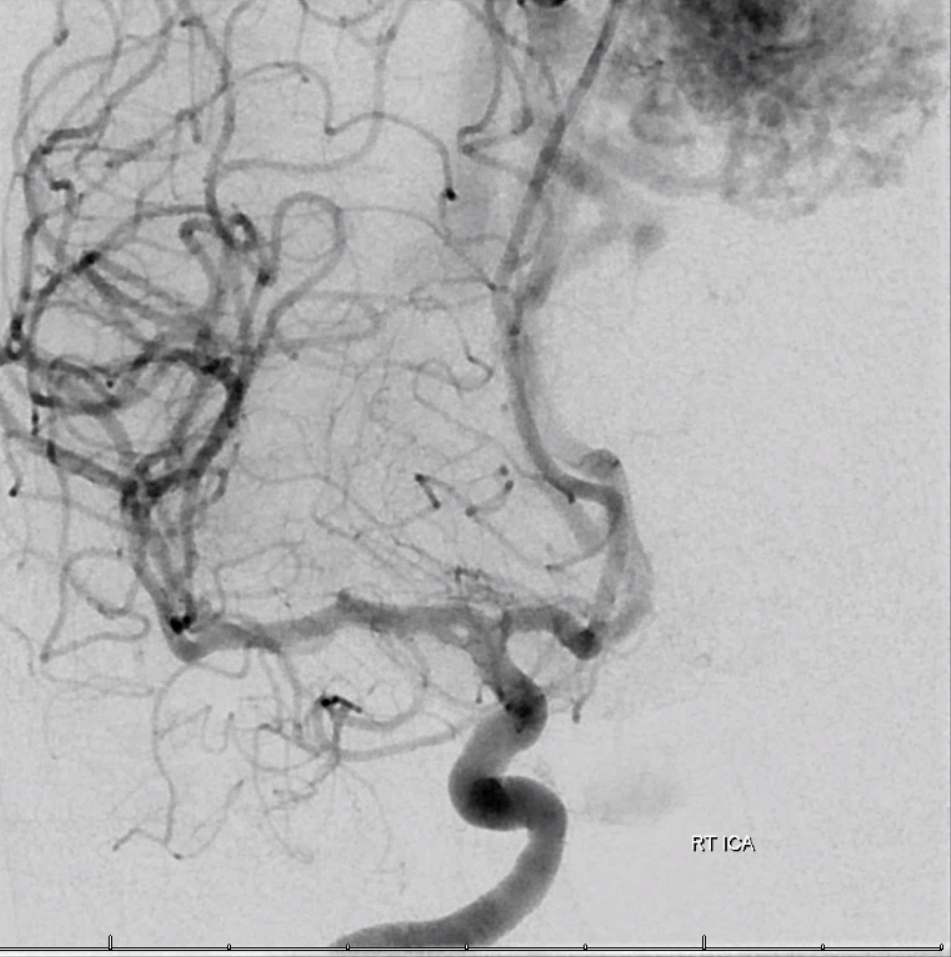
Another one, courtesy Dr. Raz — anterior temporal branch comes off inferior limb
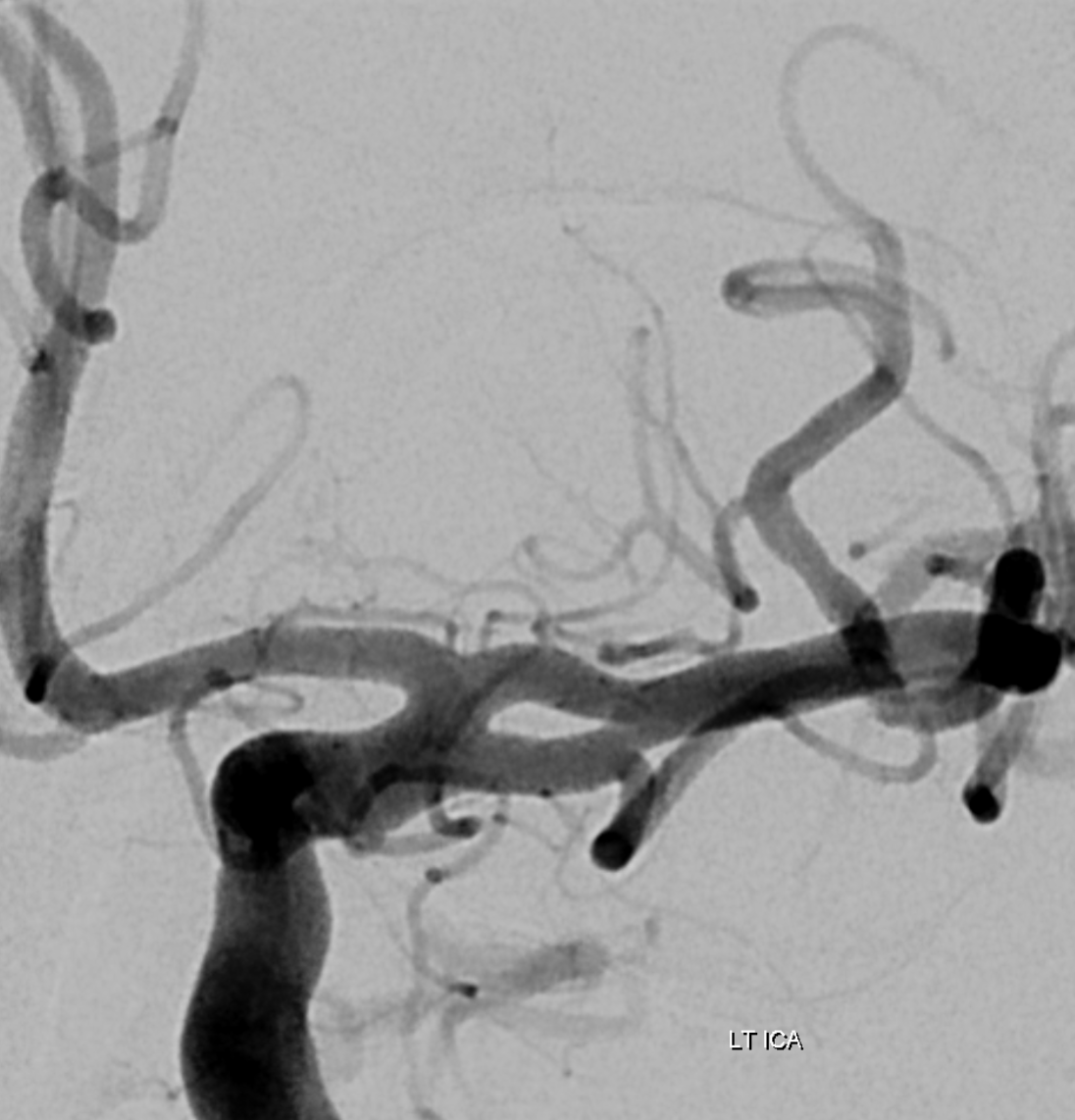
Yet another one — here the lenticulostriates are off the top one — something to keep in mind in a stroke case
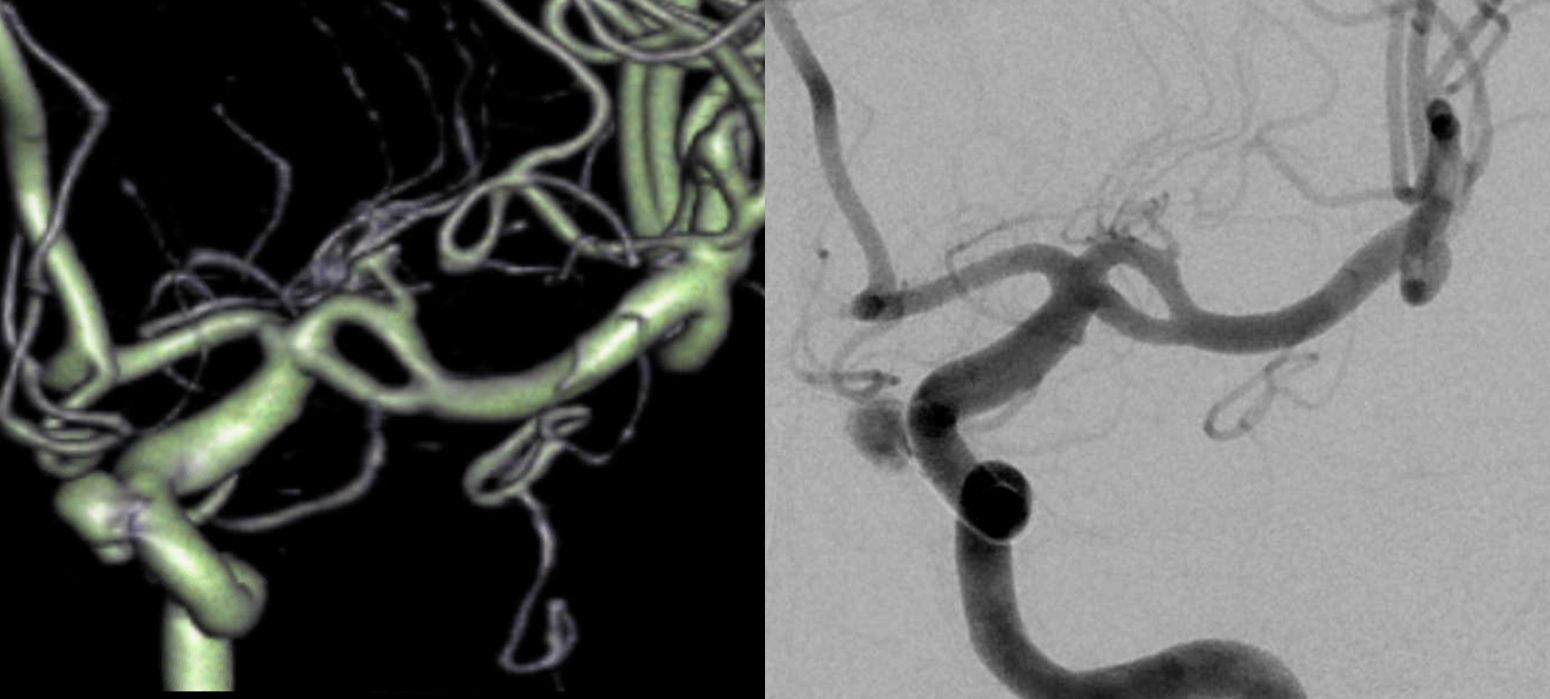
Finally, a fenestration on MRA. Fenestrations are some of the few things that are better seen on 3D reconstructions rather than source images. Small ones appear as dark lines or bars though the vessel (arrows). The VRT reconstruction is much more obvious
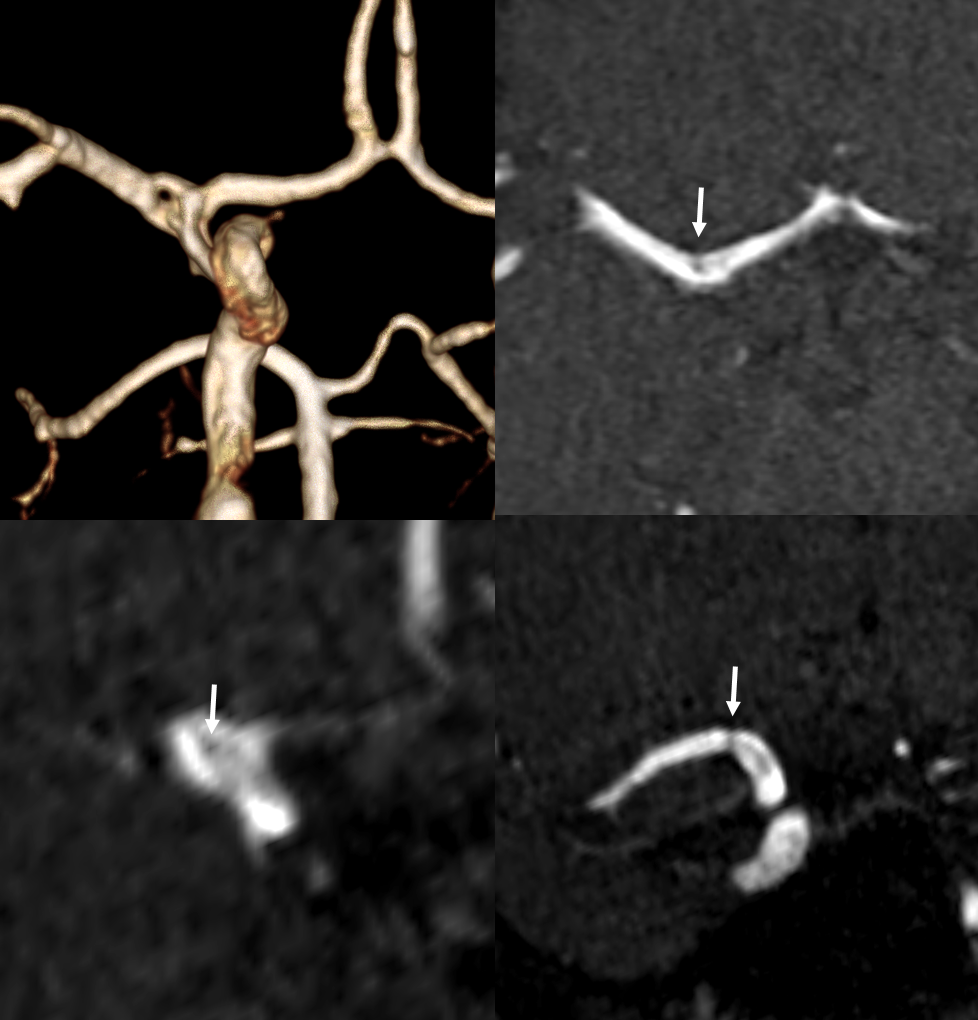
Here is another one — note medial lenticulostriate origin from A1 and M1 beyond the fenestration. Anything goes
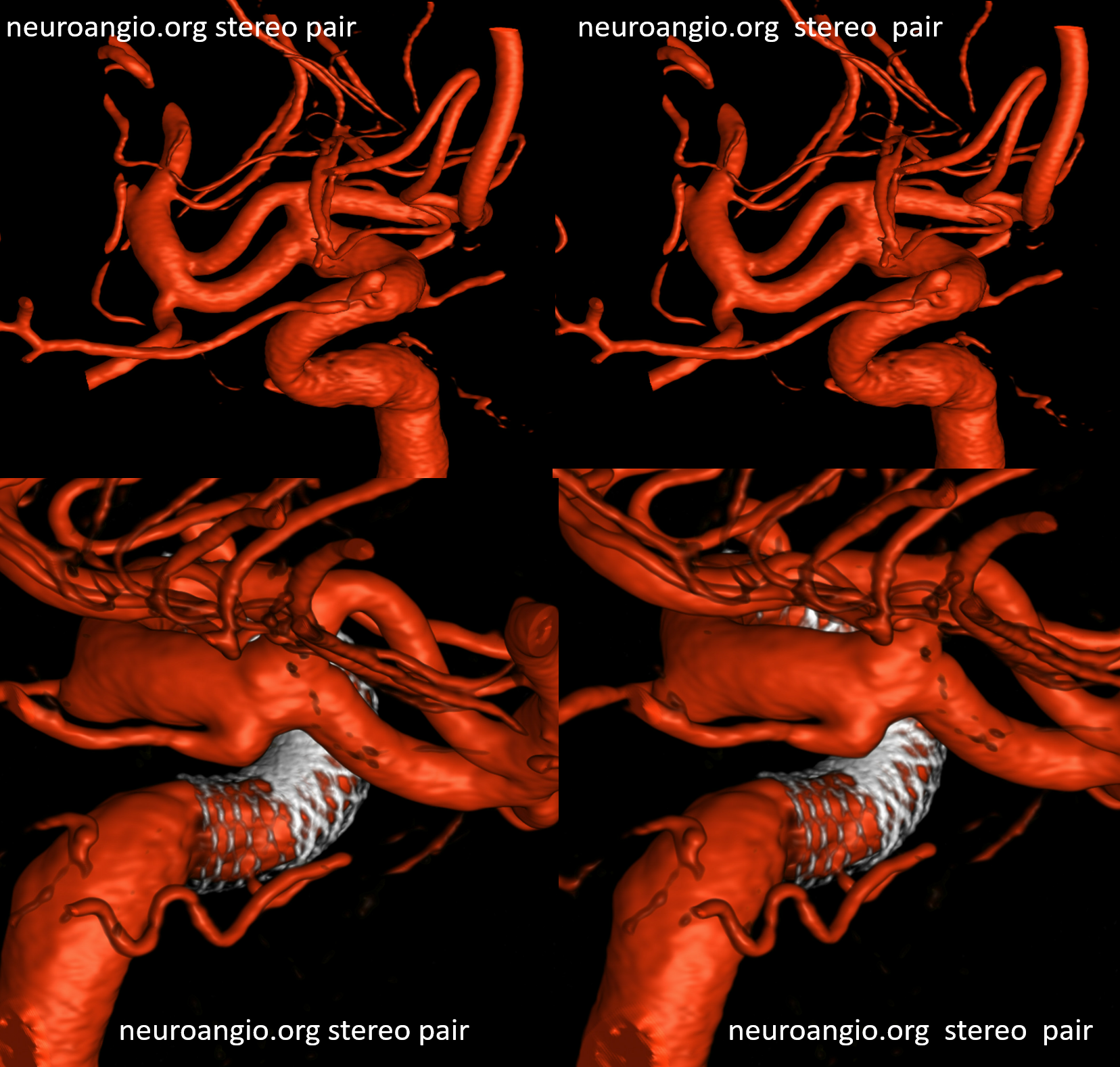
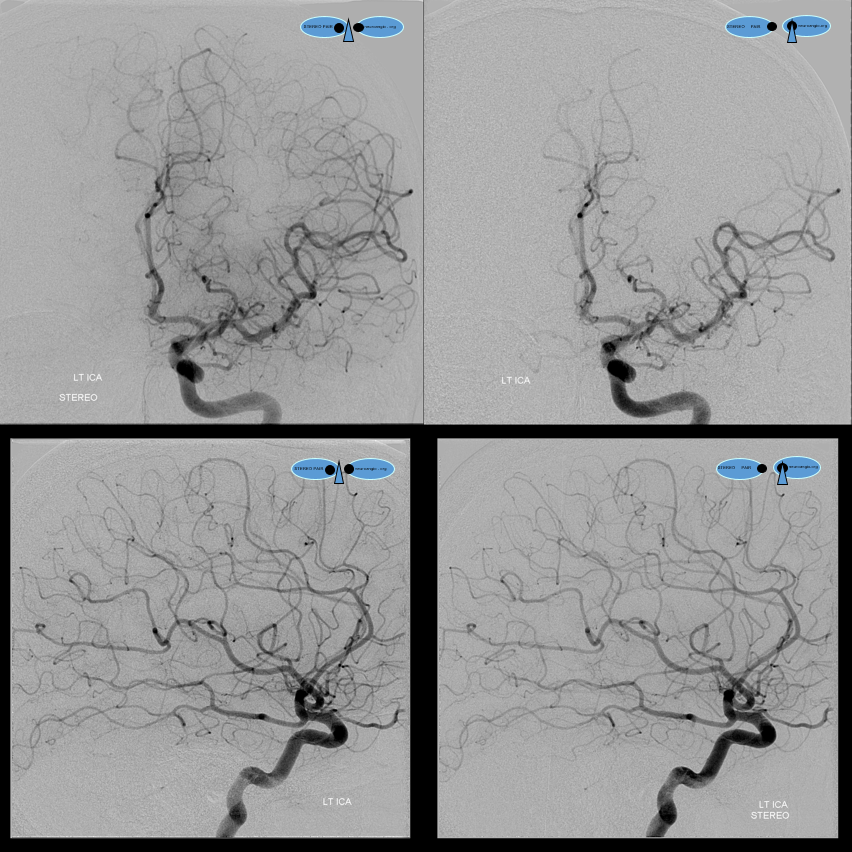
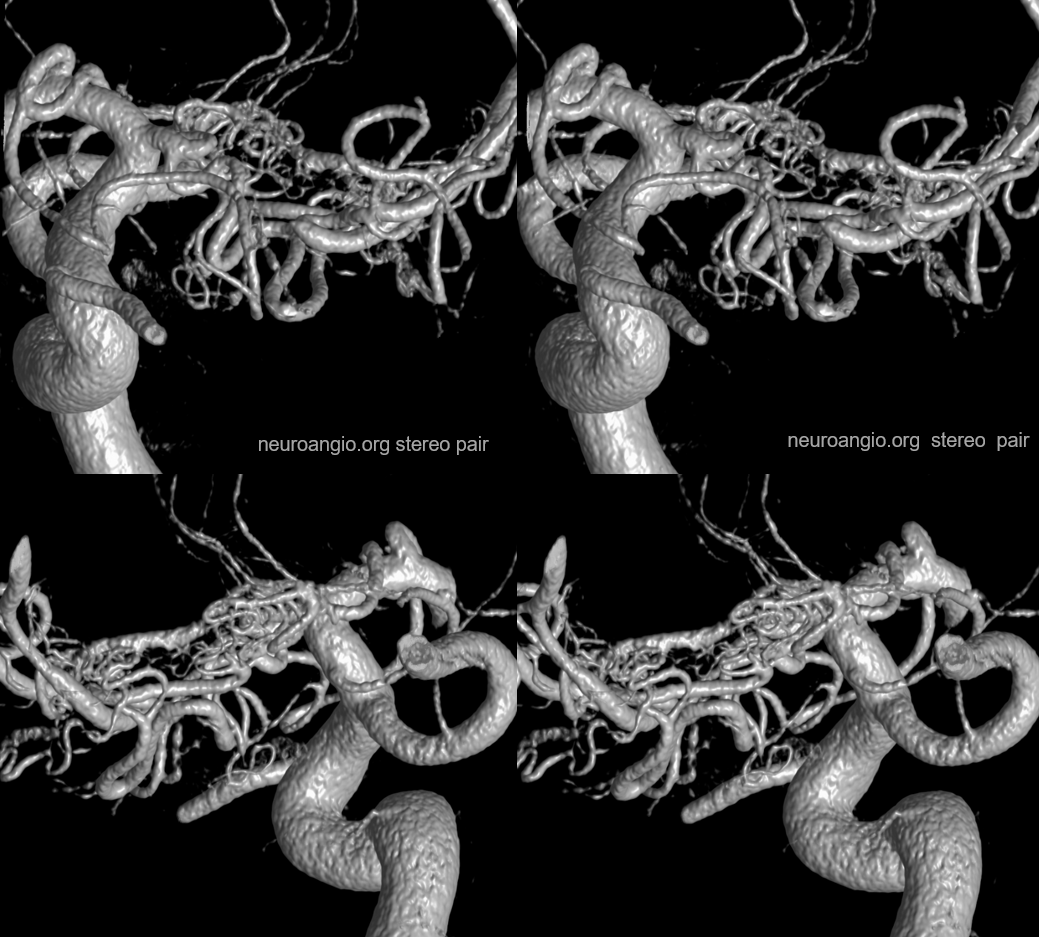
Occlusive Disease Imaging — Moya Moya, Intracranial Dissection, etc.
DYNA CT offers superb evaluation of Moya Moya / occlusive disease. Some might think this is a “twig MCA” — unilateral occlusive disease with MCA to MCA reconstitution. The ECA is not doing anything, and the leptomeningeal support is minimal given efficient “rete-type” reconstitution. Stereo pairs
Angio
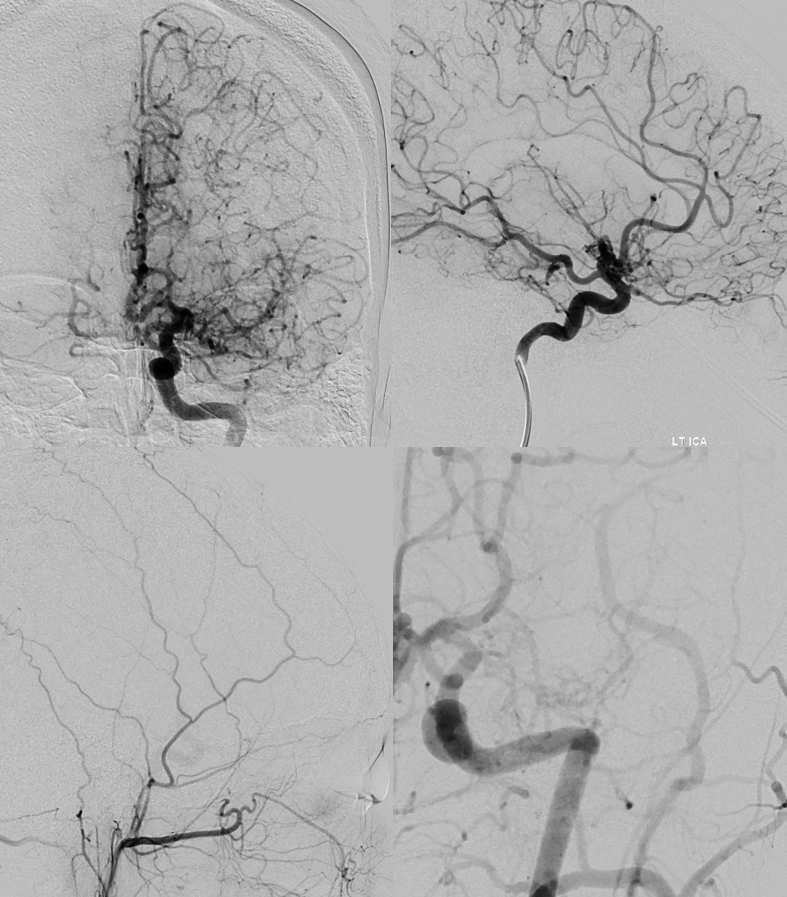
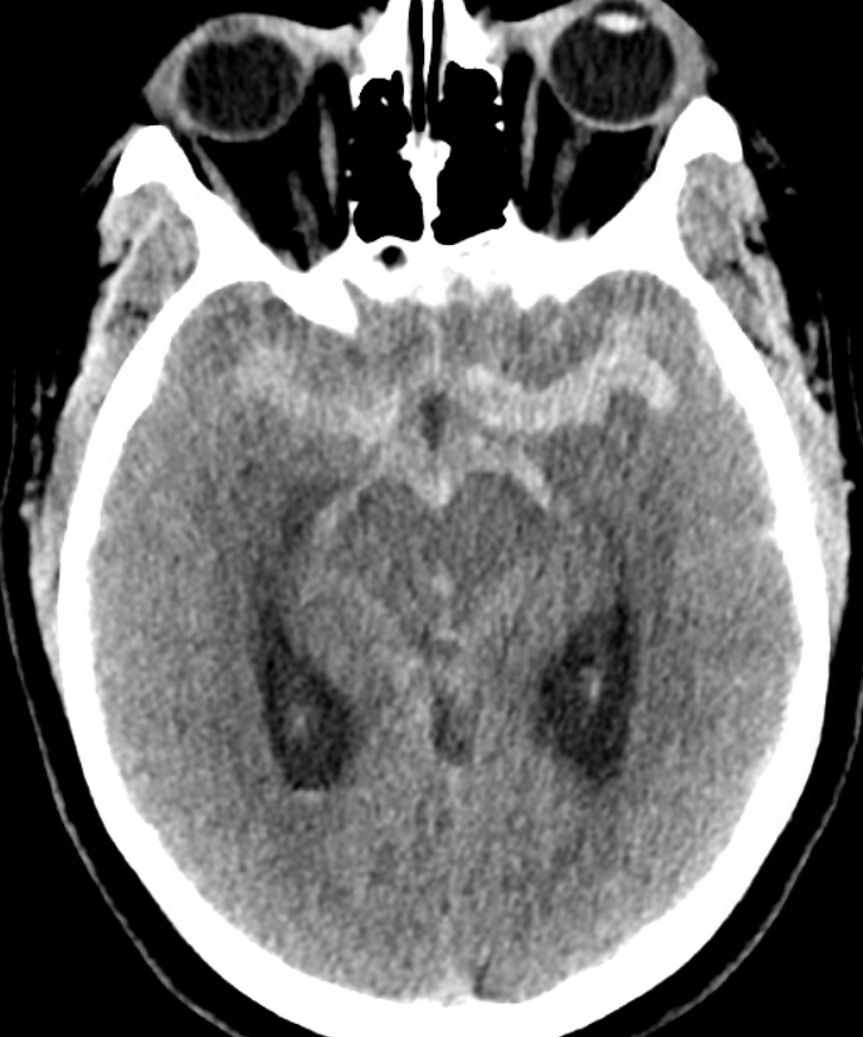
Parenchyma is normal
Had enough?
For discussions of leptomeningeal anastomoses between MCA, PCA, and ACA territories, see Collateral Circulation page
